Key Insights
The automated sterility testing market is experiencing robust growth, projected to reach \$1.43 billion in 2025 and maintain a Compound Annual Growth Rate (CAGR) of 11.10% from 2025 to 2033. This expansion is driven by several key factors. Increasing regulatory scrutiny across pharmaceutical and medical device manufacturing necessitates rigorous sterility assurance, fueling demand for automated solutions that offer enhanced accuracy, efficiency, and traceability compared to traditional manual methods. Furthermore, the rising prevalence of contamination incidents and the need to minimize the risk of product recalls are significantly impacting market growth. The pharmaceutical and biological manufacturing segment currently dominates the application landscape, owing to stringent quality control standards. However, growth within medical device manufacturing is expected to accelerate, driven by similar regulatory pressures and the increasing complexity of medical devices. Technological advancements, including the development of more sophisticated instrumentation and rapid microbial detection methods, are further contributing to market expansion. The market is segmented by test type (membrane filtration, direct inoculation, other sterility tests), application (pharmaceutical and biological manufacturing, medical device manufacturing, other applications), and product type (instruments, kits and reagents, services). Competition is fierce, with key players including Charles River Laboratories, Merck KGaA, and Sartorius AG continuously innovating and expanding their product portfolios to meet growing market demands.
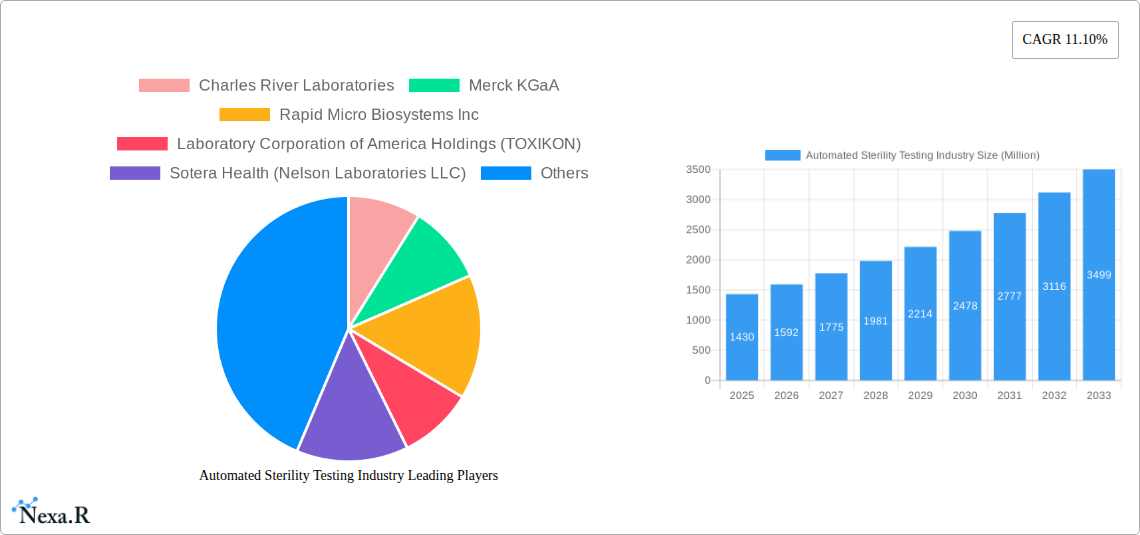
Automated Sterility Testing Industry Market Size (In Billion)

Geographic distribution reveals a strong presence across North America and Europe, reflecting the established regulatory frameworks and advanced healthcare infrastructure in these regions. However, the Asia-Pacific region is anticipated to witness the fastest growth rate in the coming years, fueled by increasing pharmaceutical manufacturing activity, rising disposable incomes, and the adoption of advanced technologies. The Middle East and Africa, and South America are also exhibiting significant growth potential, though from a smaller base, driven by investments in healthcare infrastructure and rising awareness of sterility assurance. The market's future trajectory is strongly tied to ongoing regulatory developments, technological advancements in detection technologies, and the increasing adoption of automated systems across diverse industries demanding sterile products. This suggests continued strong growth, with further market segmentation and niche players emerging to cater to specific industry requirements.
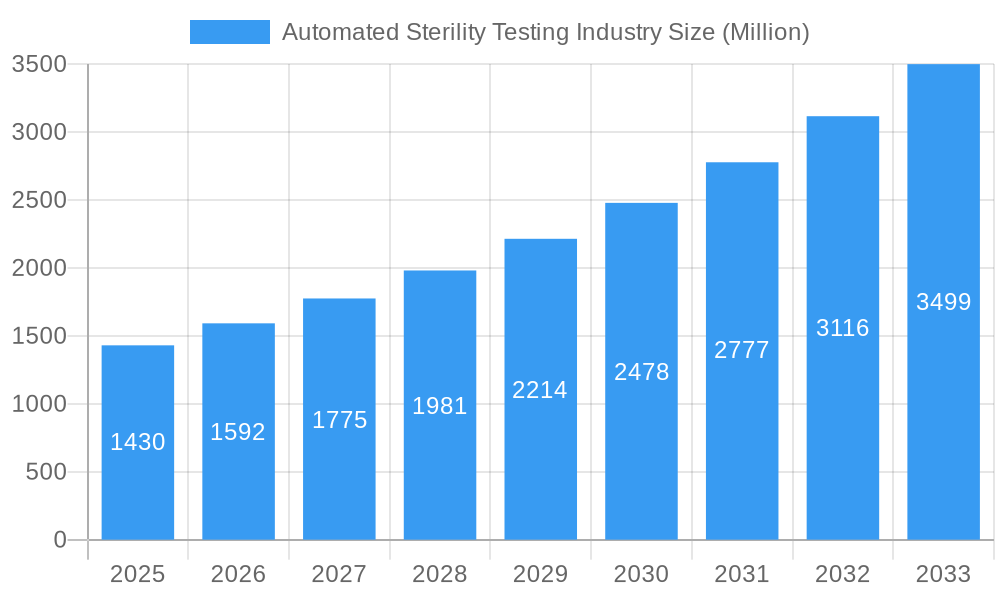
Automated Sterility Testing Industry Company Market Share

Automated Sterility Testing Industry Market Report: 2019-2033
This comprehensive report provides an in-depth analysis of the Automated Sterility Testing market, encompassing market dynamics, growth trends, regional dominance, product landscape, challenges, opportunities, and key players. The report covers the period from 2019 to 2033, with a focus on the forecast period 2025-2033 and a base year of 2025. The analysis considers the parent market of In-Vitro Diagnostics (IVD) and the child market of Sterility Testing. This report is invaluable for industry professionals, investors, and stakeholders seeking a comprehensive understanding of this rapidly evolving market. Projected market values are presented in Million USD.
Automated Sterility Testing Industry Market Dynamics & Structure
The automated sterility testing market is characterized by a moderate yet evolving concentration. Leading global players such as Charles River Laboratories, Merck KGaA, and Thermo Fisher Scientific continue to command significant market share, driving innovation and setting industry standards. A primary catalyst for market growth is continuous technological innovation, with a strong emphasis on advanced automation, robotics, and the development of rapid microbial detection methods. These advancements are crucial for streamlining processes, reducing turnaround times, and enhancing accuracy. Stringent regulatory frameworks, including guidelines from bodies like the FDA, EMA, and compliance with ISO standards, profoundly shape market practices and product development, emphasizing the critical need for validated and reliable testing solutions. While traditional manual sterility testing methods remain a competitive substitute, automated systems offer undeniable advantages in terms of speed, precision, data integrity, and ultimately, cost-effectiveness, particularly for high-volume testing. The end-user landscape is dominated by pharmaceutical and medical device manufacturers, with a notable and increasing contribution from contract research organizations (CROs) and contract development and manufacturing organizations (CDMOs) who rely on these advanced solutions for their diverse client portfolios. Mergers and acquisitions (M&A) activity has been a moderate but strategic force, with a number of targeted deals recorded between 2019 and 2024, reflecting a healthy trend of consolidation aimed at expanding capabilities and market reach within the sector.
- Market Concentration: Moderately concentrated, with the top 5 players estimated to hold approximately 50-60% of the global market share in 2024.
- Technological Innovation: Rapid advancements in automation, robotics, artificial intelligence (AI), and cutting-edge detection technologies are pivotal drivers of market growth and differentiation.
- Regulatory Landscape: Stringent and evolving regulations, including Good Manufacturing Practices (GMP), ISO 11137, and pharmacopoeial requirements, significantly influence market practices, product development, and validation protocols.
- Competitive Landscape: The market features intense competition among established global players, specialized solution providers, and emerging companies focused on niche technologies.
- M&A Activity: A moderate but strategic level of mergers and acquisitions, with approximately 10-15 significant deals recorded between 2019 and 2024, aimed at portfolio expansion and market consolidation.
Automated Sterility Testing Industry Growth Trends & Insights
The automated sterility testing market is currently experiencing robust and sustained growth, propelled by an escalating global demand for faster, more accurate, and highly reproducible sterility testing methods. The market size was impressively valued at approximately USD 1.5 billion in 2024 and is projected to expand significantly, reaching an estimated USD 3.5 billion by 2033, demonstrating a compelling Compound Annual Growth Rate (CAGR) of around 9-10% during the forecast period. This remarkable growth trajectory is primarily fueled by the accelerated adoption of automated sterility testing systems within the pharmaceutical and medical device manufacturing sectors, a growing trend of outsourcing sterile product testing to specialized CROs and CDMOs, and an unwavering focus on stringent regulatory compliance and quality assurance. Technological disruptions, most notably the emergence and refinement of rapid microbiology methods (RMMs) and the integration of AI-powered analytics for enhanced data interpretation and predictive insights, are further accelerating market expansion and innovation. Shifting consumer expectations toward higher product quality and faster turnaround times for life-saving therapeutics and medical devices also play a crucial role. Despite its rapid growth, the overall market penetration of fully automated systems remains relatively moderate in certain segments, presenting substantial opportunities for increased adoption across a wider range of manufacturers and laboratories in the coming years.
Dominant Regions, Countries, or Segments in Automated Sterility Testing Industry
North America currently dominates the automated sterility testing market, holding approximately xx% market share in 2024, followed by Europe and Asia Pacific. This dominance is attributed to the presence of major pharmaceutical and medical device manufacturers, advanced healthcare infrastructure, and stringent regulatory frameworks. Within the market segments, Pharmaceutical and Biological Manufacturing holds the largest share, followed by Medical Device Manufacturing. The Membrane Filtration test type accounts for the largest proportion of the market share, driven by its widespread adoption in various applications.
- North America: Strong presence of major players, advanced healthcare infrastructure, and high regulatory standards.
- Europe: Significant market size and growth potential, driven by increasing adoption of automated technologies.
- Asia Pacific: Rapid growth potential, fueled by expanding pharmaceutical and medical device industries.
- By Test Type: Membrane Filtration holds the largest market share, followed by Direct Inoculation.
- By Application: Pharmaceutical and Biological Manufacturing represents the largest segment.
Automated Sterility Testing Industry Product Landscape
The automated sterility testing market offers a wide range of products, including automated instruments, kits and reagents, and testing services. These products incorporate advanced technologies like robotics, image analysis, and rapid microbial detection methods, improving accuracy, speed, and efficiency. Key selling propositions include reduced testing time, improved accuracy, and enhanced regulatory compliance. Recent technological advancements have focused on miniaturization, integration with laboratory information management systems (LIMS), and the development of automated sample preparation techniques.
Key Drivers, Barriers & Challenges in Automated Sterility Testing Industry
Key Drivers:
- Increasing demand for rapid and accurate sterility testing.
- Stringent regulatory requirements demanding high accuracy and traceability.
- Rising adoption of automation in pharmaceutical and medical device manufacturing.
- Technological advancements such as automated sample preparation and rapid detection methods.
Challenges & Restraints:
- High initial investment costs associated with automated systems.
- Lack of skilled personnel to operate and maintain sophisticated equipment.
- Potential integration challenges with existing laboratory workflows.
- Regulatory hurdles and variations across different regions.
Emerging Opportunities in Automated Sterility Testing Industry
The automated sterility testing market is ripe with emerging opportunities, particularly in untapped and rapidly developing markets across Asia-Pacific, Latin America, and Africa, where the adoption of advanced automated sterility testing solutions is still in its nascent stages. Significant potential also lies in the research and development of innovative point-of-care testing devices and integrated platforms that bring sterility assurance closer to the manufacturing line or even directly to clinical settings. The integration of sophisticated AI and machine learning algorithms for advanced data analysis, predictive quality control, and proactive identification of potential contamination risks presents a transformative opportunity for improved efficiency and risk management. Furthermore, the expanding landscape of personalized medicine, advanced cell and gene therapies, and biopharmaceuticals necessitates the development of specialized, highly sensitive, and flexible sterility testing solutions. The growing demand for cost-effective, scalable, and user-friendly automated systems tailored for smaller-scale manufacturers, academic research laboratories, and niche biotechnology companies also represents a significant and promising area for market growth and innovation.
Growth Accelerators in the Automated Sterility Testing Industry
Technological breakthroughs in miniaturization, improved detection methods, and AI-powered analysis are key growth accelerators. Strategic partnerships between instrument manufacturers, reagent suppliers, and CROs enhance market penetration. Market expansion strategies, including entering emerging economies and developing tailored solutions for specific applications, fuel market growth. Increased regulatory emphasis on automation is accelerating wider adoption.
Key Players Shaping the Automated Sterility Testing Industry Market
- Charles River Laboratories
- Merck KGaA (including MilliporeSigma)
- Rapid Micro Biosystems Inc
- Laboratory Corporation of America Holdings (TOXIKON)
- Sotera Health (Nelson Laboratories LLC)
- Sartorius AG
- SGS SA
- bioMérieux SA
- WuXi AppTec
- Pace Analytical
- Thermo Fisher Scientific
- STERIS
- BD (Becton, Dickinson and Company)
- Novartis AG
Notable Milestones in Automated Sterility Testing Industry Sector
- June 2022: STEMart launched its comprehensive microbiology and sterility testing services, specifically tailored for sterile, non-pyrogenic medical devices, enhancing product safety for manufacturers.
- June 2022: Berkshire Sterile Manufacturing inaugurated a state-of-the-art sterility testing isolator, enabling on-site GMP-compliant batch testing and significantly reducing lead times for their clients.
- October 2023: Rapid Micro Biosystems announced the successful validation of its Growth Direct™ System with new media formulations, further expanding its applicability and efficiency for pharmaceutical sterility testing.
- January 2024: Sartorius AG expanded its portfolio with the acquisition of Cytiva, a move expected to bolster its offerings in bioprocessing and sterile drug manufacturing solutions, including advanced sterility assurance.
- March 2024: Merck KGaA unveiled its new automated sterility testing platform, designed to offer enhanced throughput and advanced data management capabilities, addressing the growing needs of large-scale pharmaceutical production.
In-Depth Automated Sterility Testing Industry Market Outlook
The automated sterility testing market is poised for continued strong growth, driven by technological advancements, increasing regulatory scrutiny, and the rising demand for efficient and accurate sterility testing. Strategic partnerships, expansion into emerging markets, and the development of innovative solutions will shape the future market landscape. Opportunities exist for companies that can offer cost-effective, user-friendly, and highly accurate automated systems tailored to the specific needs of various market segments. The integration of AI and advanced data analytics will further enhance the efficiency and effectiveness of automated sterility testing, creating new opportunities for growth and innovation.
Automated Sterility Testing Industry Segmentation
-
1. Product Type
- 1.1. Instruments
- 1.2. Kits and Reagents
- 1.3. Services
-
2. Test Type
- 2.1. Membrane Filtration
- 2.2. Direct Inoculation
- 2.3. Other Sterility Tests
-
3. Application
- 3.1. Pharmaceutical and Biological Manufacturing
- 3.2. Medical Device Manufacturing
- 3.3. Other Applications
Automated Sterility Testing Industry Segmentation By Geography
-
1. North America
- 1.1. United States
- 1.2. Canada
- 1.3. Mexico
-
2. Europe
- 2.1. Germany
- 2.2. United Kingdom
- 2.3. France
- 2.4. Italy
- 2.5. Spain
- 2.6. Rest of Europe
-
3. Asia Pacific
- 3.1. China
- 3.2. Japan
- 3.3. India
- 3.4. Australia
- 3.5. South Korea
- 3.6. Rest of Asia Pacific
-
4. Middle East and Africa
- 4.1. GCC
- 4.2. South Africa
- 4.3. Rest of Middle East and Africa
-
5. South America
- 5.1. Brazil
- 5.2. Argentina
- 5.3. Rest of South America
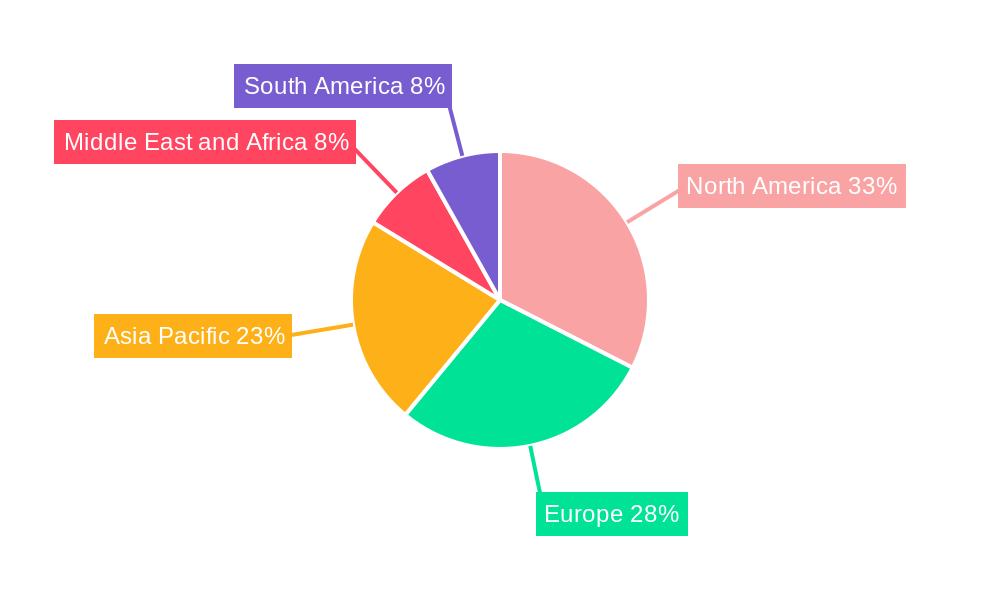
Automated Sterility Testing Industry Regional Market Share

Geographic Coverage of Automated Sterility Testing Industry
Automated Sterility Testing Industry REPORT HIGHLIGHTS
| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 11.10% from 2020-2034 |
| Segmentation |
|
Table of Contents
- 1. Introduction
- 1.1. Research Scope
- 1.2. Market Segmentation
- 1.3. Research Methodology
- 1.4. Definitions and Assumptions
- 2. Executive Summary
- 2.1. Introduction
- 3. Market Dynamics
- 3.1. Introduction
- 3.2. Market Drivers
- 3.2.1. Increasing Research and Development in Life Sciences; Increasing Number of Drug Launches; Rise in Demand for Sterilized Products
- 3.3. Market Restrains
- 3.3.1. Stringent Regulatory Framework
- 3.4. Market Trends
- 3.4.1. Pharmaceutical and Biological Manufacturing Segment Expected to Grow Over the Forecast Period
- 4. Market Factor Analysis
- 4.1. Porters Five Forces
- 4.2. Supply/Value Chain
- 4.3. PESTEL analysis
- 4.4. Market Entropy
- 4.5. Patent/Trademark Analysis
- 5. Global Automated Sterility Testing Industry Analysis, Insights and Forecast, 2020-2032
- 5.1. Market Analysis, Insights and Forecast - by Product Type
- 5.1.1. Instruments
- 5.1.2. Kits and Reagents
- 5.1.3. Services
- 5.2. Market Analysis, Insights and Forecast - by Test Type
- 5.2.1. Membrane Filtration
- 5.2.2. Direct Inoculation
- 5.2.3. Other Sterility Tests
- 5.3. Market Analysis, Insights and Forecast - by Application
- 5.3.1. Pharmaceutical and Biological Manufacturing
- 5.3.2. Medical Device Manufacturing
- 5.3.3. Other Applications
- 5.4. Market Analysis, Insights and Forecast - by Region
- 5.4.1. North America
- 5.4.2. Europe
- 5.4.3. Asia Pacific
- 5.4.4. Middle East and Africa
- 5.4.5. South America
- 5.1. Market Analysis, Insights and Forecast - by Product Type
- 6. North America Automated Sterility Testing Industry Analysis, Insights and Forecast, 2020-2032
- 6.1. Market Analysis, Insights and Forecast - by Product Type
- 6.1.1. Instruments
- 6.1.2. Kits and Reagents
- 6.1.3. Services
- 6.2. Market Analysis, Insights and Forecast - by Test Type
- 6.2.1. Membrane Filtration
- 6.2.2. Direct Inoculation
- 6.2.3. Other Sterility Tests
- 6.3. Market Analysis, Insights and Forecast - by Application
- 6.3.1. Pharmaceutical and Biological Manufacturing
- 6.3.2. Medical Device Manufacturing
- 6.3.3. Other Applications
- 6.1. Market Analysis, Insights and Forecast - by Product Type
- 7. Europe Automated Sterility Testing Industry Analysis, Insights and Forecast, 2020-2032
- 7.1. Market Analysis, Insights and Forecast - by Product Type
- 7.1.1. Instruments
- 7.1.2. Kits and Reagents
- 7.1.3. Services
- 7.2. Market Analysis, Insights and Forecast - by Test Type
- 7.2.1. Membrane Filtration
- 7.2.2. Direct Inoculation
- 7.2.3. Other Sterility Tests
- 7.3. Market Analysis, Insights and Forecast - by Application
- 7.3.1. Pharmaceutical and Biological Manufacturing
- 7.3.2. Medical Device Manufacturing
- 7.3.3. Other Applications
- 7.1. Market Analysis, Insights and Forecast - by Product Type
- 8. Asia Pacific Automated Sterility Testing Industry Analysis, Insights and Forecast, 2020-2032
- 8.1. Market Analysis, Insights and Forecast - by Product Type
- 8.1.1. Instruments
- 8.1.2. Kits and Reagents
- 8.1.3. Services
- 8.2. Market Analysis, Insights and Forecast - by Test Type
- 8.2.1. Membrane Filtration
- 8.2.2. Direct Inoculation
- 8.2.3. Other Sterility Tests
- 8.3. Market Analysis, Insights and Forecast - by Application
- 8.3.1. Pharmaceutical and Biological Manufacturing
- 8.3.2. Medical Device Manufacturing
- 8.3.3. Other Applications
- 8.1. Market Analysis, Insights and Forecast - by Product Type
- 9. Middle East and Africa Automated Sterility Testing Industry Analysis, Insights and Forecast, 2020-2032
- 9.1. Market Analysis, Insights and Forecast - by Product Type
- 9.1.1. Instruments
- 9.1.2. Kits and Reagents
- 9.1.3. Services
- 9.2. Market Analysis, Insights and Forecast - by Test Type
- 9.2.1. Membrane Filtration
- 9.2.2. Direct Inoculation
- 9.2.3. Other Sterility Tests
- 9.3. Market Analysis, Insights and Forecast - by Application
- 9.3.1. Pharmaceutical and Biological Manufacturing
- 9.3.2. Medical Device Manufacturing
- 9.3.3. Other Applications
- 9.1. Market Analysis, Insights and Forecast - by Product Type
- 10. South America Automated Sterility Testing Industry Analysis, Insights and Forecast, 2020-2032
- 10.1. Market Analysis, Insights and Forecast - by Product Type
- 10.1.1. Instruments
- 10.1.2. Kits and Reagents
- 10.1.3. Services
- 10.2. Market Analysis, Insights and Forecast - by Test Type
- 10.2.1. Membrane Filtration
- 10.2.2. Direct Inoculation
- 10.2.3. Other Sterility Tests
- 10.3. Market Analysis, Insights and Forecast - by Application
- 10.3.1. Pharmaceutical and Biological Manufacturing
- 10.3.2. Medical Device Manufacturing
- 10.3.3. Other Applications
- 10.1. Market Analysis, Insights and Forecast - by Product Type
- 11. Competitive Analysis
- 11.1. Global Market Share Analysis 2025
- 11.2. Company Profiles
- 11.2.1 Charles River Laboratories
- 11.2.1.1. Overview
- 11.2.1.2. Products
- 11.2.1.3. SWOT Analysis
- 11.2.1.4. Recent Developments
- 11.2.1.5. Financials (Based on Availability)
- 11.2.2 Merck KGaA
- 11.2.2.1. Overview
- 11.2.2.2. Products
- 11.2.2.3. SWOT Analysis
- 11.2.2.4. Recent Developments
- 11.2.2.5. Financials (Based on Availability)
- 11.2.3 Rapid Micro Biosystems Inc
- 11.2.3.1. Overview
- 11.2.3.2. Products
- 11.2.3.3. SWOT Analysis
- 11.2.3.4. Recent Developments
- 11.2.3.5. Financials (Based on Availability)
- 11.2.4 Laboratory Corporation of America Holdings (TOXIKON)
- 11.2.4.1. Overview
- 11.2.4.2. Products
- 11.2.4.3. SWOT Analysis
- 11.2.4.4. Recent Developments
- 11.2.4.5. Financials (Based on Availability)
- 11.2.5 Sotera Health (Nelson Laboratories LLC)
- 11.2.5.1. Overview
- 11.2.5.2. Products
- 11.2.5.3. SWOT Analysis
- 11.2.5.4. Recent Developments
- 11.2.5.5. Financials (Based on Availability)
- 11.2.6 Sartorius AG
- 11.2.6.1. Overview
- 11.2.6.2. Products
- 11.2.6.3. SWOT Analysis
- 11.2.6.4. Recent Developments
- 11.2.6.5. Financials (Based on Availability)
- 11.2.7 SGS SA
- 11.2.7.1. Overview
- 11.2.7.2. Products
- 11.2.7.3. SWOT Analysis
- 11.2.7.4. Recent Developments
- 11.2.7.5. Financials (Based on Availability)
- 11.2.8 bioMerieux SA
- 11.2.8.1. Overview
- 11.2.8.2. Products
- 11.2.8.3. SWOT Analysis
- 11.2.8.4. Recent Developments
- 11.2.8.5. Financials (Based on Availability)
- 11.2.9 WuXi AppTec
- 11.2.9.1. Overview
- 11.2.9.2. Products
- 11.2.9.3. SWOT Analysis
- 11.2.9.4. Recent Developments
- 11.2.9.5. Financials (Based on Availability)
- 11.2.10 Pace Analytical
- 11.2.10.1. Overview
- 11.2.10.2. Products
- 11.2.10.3. SWOT Analysis
- 11.2.10.4. Recent Developments
- 11.2.10.5. Financials (Based on Availability)
- 11.2.11 Thermo Fisher Scientific
- 11.2.11.1. Overview
- 11.2.11.2. Products
- 11.2.11.3. SWOT Analysis
- 11.2.11.4. Recent Developments
- 11.2.11.5. Financials (Based on Availability)
- 11.2.12 STERIS
- 11.2.12.1. Overview
- 11.2.12.2. Products
- 11.2.12.3. SWOT Analysis
- 11.2.12.4. Recent Developments
- 11.2.12.5. Financials (Based on Availability)
- 11.2.1 Charles River Laboratories
List of Figures
- Figure 1: Global Automated Sterility Testing Industry Revenue Breakdown (Million, %) by Region 2025 & 2033
- Figure 2: North America Automated Sterility Testing Industry Revenue (Million), by Product Type 2025 & 2033
- Figure 3: North America Automated Sterility Testing Industry Revenue Share (%), by Product Type 2025 & 2033
- Figure 4: North America Automated Sterility Testing Industry Revenue (Million), by Test Type 2025 & 2033
- Figure 5: North America Automated Sterility Testing Industry Revenue Share (%), by Test Type 2025 & 2033
- Figure 6: North America Automated Sterility Testing Industry Revenue (Million), by Application 2025 & 2033
- Figure 7: North America Automated Sterility Testing Industry Revenue Share (%), by Application 2025 & 2033
- Figure 8: North America Automated Sterility Testing Industry Revenue (Million), by Country 2025 & 2033
- Figure 9: North America Automated Sterility Testing Industry Revenue Share (%), by Country 2025 & 2033
- Figure 10: Europe Automated Sterility Testing Industry Revenue (Million), by Product Type 2025 & 2033
- Figure 11: Europe Automated Sterility Testing Industry Revenue Share (%), by Product Type 2025 & 2033
- Figure 12: Europe Automated Sterility Testing Industry Revenue (Million), by Test Type 2025 & 2033
- Figure 13: Europe Automated Sterility Testing Industry Revenue Share (%), by Test Type 2025 & 2033
- Figure 14: Europe Automated Sterility Testing Industry Revenue (Million), by Application 2025 & 2033
- Figure 15: Europe Automated Sterility Testing Industry Revenue Share (%), by Application 2025 & 2033
- Figure 16: Europe Automated Sterility Testing Industry Revenue (Million), by Country 2025 & 2033
- Figure 17: Europe Automated Sterility Testing Industry Revenue Share (%), by Country 2025 & 2033
- Figure 18: Asia Pacific Automated Sterility Testing Industry Revenue (Million), by Product Type 2025 & 2033
- Figure 19: Asia Pacific Automated Sterility Testing Industry Revenue Share (%), by Product Type 2025 & 2033
- Figure 20: Asia Pacific Automated Sterility Testing Industry Revenue (Million), by Test Type 2025 & 2033
- Figure 21: Asia Pacific Automated Sterility Testing Industry Revenue Share (%), by Test Type 2025 & 2033
- Figure 22: Asia Pacific Automated Sterility Testing Industry Revenue (Million), by Application 2025 & 2033
- Figure 23: Asia Pacific Automated Sterility Testing Industry Revenue Share (%), by Application 2025 & 2033
- Figure 24: Asia Pacific Automated Sterility Testing Industry Revenue (Million), by Country 2025 & 2033
- Figure 25: Asia Pacific Automated Sterility Testing Industry Revenue Share (%), by Country 2025 & 2033
- Figure 26: Middle East and Africa Automated Sterility Testing Industry Revenue (Million), by Product Type 2025 & 2033
- Figure 27: Middle East and Africa Automated Sterility Testing Industry Revenue Share (%), by Product Type 2025 & 2033
- Figure 28: Middle East and Africa Automated Sterility Testing Industry Revenue (Million), by Test Type 2025 & 2033
- Figure 29: Middle East and Africa Automated Sterility Testing Industry Revenue Share (%), by Test Type 2025 & 2033
- Figure 30: Middle East and Africa Automated Sterility Testing Industry Revenue (Million), by Application 2025 & 2033
- Figure 31: Middle East and Africa Automated Sterility Testing Industry Revenue Share (%), by Application 2025 & 2033
- Figure 32: Middle East and Africa Automated Sterility Testing Industry Revenue (Million), by Country 2025 & 2033
- Figure 33: Middle East and Africa Automated Sterility Testing Industry Revenue Share (%), by Country 2025 & 2033
- Figure 34: South America Automated Sterility Testing Industry Revenue (Million), by Product Type 2025 & 2033
- Figure 35: South America Automated Sterility Testing Industry Revenue Share (%), by Product Type 2025 & 2033
- Figure 36: South America Automated Sterility Testing Industry Revenue (Million), by Test Type 2025 & 2033
- Figure 37: South America Automated Sterility Testing Industry Revenue Share (%), by Test Type 2025 & 2033
- Figure 38: South America Automated Sterility Testing Industry Revenue (Million), by Application 2025 & 2033
- Figure 39: South America Automated Sterility Testing Industry Revenue Share (%), by Application 2025 & 2033
- Figure 40: South America Automated Sterility Testing Industry Revenue (Million), by Country 2025 & 2033
- Figure 41: South America Automated Sterility Testing Industry Revenue Share (%), by Country 2025 & 2033
List of Tables
- Table 1: Global Automated Sterility Testing Industry Revenue Million Forecast, by Product Type 2020 & 2033
- Table 2: Global Automated Sterility Testing Industry Revenue Million Forecast, by Test Type 2020 & 2033
- Table 3: Global Automated Sterility Testing Industry Revenue Million Forecast, by Application 2020 & 2033
- Table 4: Global Automated Sterility Testing Industry Revenue Million Forecast, by Region 2020 & 2033
- Table 5: Global Automated Sterility Testing Industry Revenue Million Forecast, by Product Type 2020 & 2033
- Table 6: Global Automated Sterility Testing Industry Revenue Million Forecast, by Test Type 2020 & 2033
- Table 7: Global Automated Sterility Testing Industry Revenue Million Forecast, by Application 2020 & 2033
- Table 8: Global Automated Sterility Testing Industry Revenue Million Forecast, by Country 2020 & 2033
- Table 9: United States Automated Sterility Testing Industry Revenue (Million) Forecast, by Application 2020 & 2033
- Table 10: Canada Automated Sterility Testing Industry Revenue (Million) Forecast, by Application 2020 & 2033
- Table 11: Mexico Automated Sterility Testing Industry Revenue (Million) Forecast, by Application 2020 & 2033
- Table 12: Global Automated Sterility Testing Industry Revenue Million Forecast, by Product Type 2020 & 2033
- Table 13: Global Automated Sterility Testing Industry Revenue Million Forecast, by Test Type 2020 & 2033
- Table 14: Global Automated Sterility Testing Industry Revenue Million Forecast, by Application 2020 & 2033
- Table 15: Global Automated Sterility Testing Industry Revenue Million Forecast, by Country 2020 & 2033
- Table 16: Germany Automated Sterility Testing Industry Revenue (Million) Forecast, by Application 2020 & 2033
- Table 17: United Kingdom Automated Sterility Testing Industry Revenue (Million) Forecast, by Application 2020 & 2033
- Table 18: France Automated Sterility Testing Industry Revenue (Million) Forecast, by Application 2020 & 2033
- Table 19: Italy Automated Sterility Testing Industry Revenue (Million) Forecast, by Application 2020 & 2033
- Table 20: Spain Automated Sterility Testing Industry Revenue (Million) Forecast, by Application 2020 & 2033
- Table 21: Rest of Europe Automated Sterility Testing Industry Revenue (Million) Forecast, by Application 2020 & 2033
- Table 22: Global Automated Sterility Testing Industry Revenue Million Forecast, by Product Type 2020 & 2033
- Table 23: Global Automated Sterility Testing Industry Revenue Million Forecast, by Test Type 2020 & 2033
- Table 24: Global Automated Sterility Testing Industry Revenue Million Forecast, by Application 2020 & 2033
- Table 25: Global Automated Sterility Testing Industry Revenue Million Forecast, by Country 2020 & 2033
- Table 26: China Automated Sterility Testing Industry Revenue (Million) Forecast, by Application 2020 & 2033
- Table 27: Japan Automated Sterility Testing Industry Revenue (Million) Forecast, by Application 2020 & 2033
- Table 28: India Automated Sterility Testing Industry Revenue (Million) Forecast, by Application 2020 & 2033
- Table 29: Australia Automated Sterility Testing Industry Revenue (Million) Forecast, by Application 2020 & 2033
- Table 30: South Korea Automated Sterility Testing Industry Revenue (Million) Forecast, by Application 2020 & 2033
- Table 31: Rest of Asia Pacific Automated Sterility Testing Industry Revenue (Million) Forecast, by Application 2020 & 2033
- Table 32: Global Automated Sterility Testing Industry Revenue Million Forecast, by Product Type 2020 & 2033
- Table 33: Global Automated Sterility Testing Industry Revenue Million Forecast, by Test Type 2020 & 2033
- Table 34: Global Automated Sterility Testing Industry Revenue Million Forecast, by Application 2020 & 2033
- Table 35: Global Automated Sterility Testing Industry Revenue Million Forecast, by Country 2020 & 2033
- Table 36: GCC Automated Sterility Testing Industry Revenue (Million) Forecast, by Application 2020 & 2033
- Table 37: South Africa Automated Sterility Testing Industry Revenue (Million) Forecast, by Application 2020 & 2033
- Table 38: Rest of Middle East and Africa Automated Sterility Testing Industry Revenue (Million) Forecast, by Application 2020 & 2033
- Table 39: Global Automated Sterility Testing Industry Revenue Million Forecast, by Product Type 2020 & 2033
- Table 40: Global Automated Sterility Testing Industry Revenue Million Forecast, by Test Type 2020 & 2033
- Table 41: Global Automated Sterility Testing Industry Revenue Million Forecast, by Application 2020 & 2033
- Table 42: Global Automated Sterility Testing Industry Revenue Million Forecast, by Country 2020 & 2033
- Table 43: Brazil Automated Sterility Testing Industry Revenue (Million) Forecast, by Application 2020 & 2033
- Table 44: Argentina Automated Sterility Testing Industry Revenue (Million) Forecast, by Application 2020 & 2033
- Table 45: Rest of South America Automated Sterility Testing Industry Revenue (Million) Forecast, by Application 2020 & 2033
Frequently Asked Questions
1. What is the projected Compound Annual Growth Rate (CAGR) of the Automated Sterility Testing Industry?
The projected CAGR is approximately 11.10%.
2. Which companies are prominent players in the Automated Sterility Testing Industry?
Key companies in the market include Charles River Laboratories, Merck KGaA, Rapid Micro Biosystems Inc, Laboratory Corporation of America Holdings (TOXIKON), Sotera Health (Nelson Laboratories LLC), Sartorius AG, SGS SA, bioMerieux SA, WuXi AppTec, Pace Analytical, Thermo Fisher Scientific, STERIS.
3. What are the main segments of the Automated Sterility Testing Industry?
The market segments include Product Type, Test Type, Application.
4. Can you provide details about the market size?
The market size is estimated to be USD 1.43 Million as of 2022.
5. What are some drivers contributing to market growth?
Increasing Research and Development in Life Sciences; Increasing Number of Drug Launches; Rise in Demand for Sterilized Products.
6. What are the notable trends driving market growth?
Pharmaceutical and Biological Manufacturing Segment Expected to Grow Over the Forecast Period.
7. Are there any restraints impacting market growth?
Stringent Regulatory Framework.
8. Can you provide examples of recent developments in the market?
Jun 2022: STEMart launched comprehensive microbiology and sterility testing for sterile, non-pyrogenic medical devices.
9. What pricing options are available for accessing the report?
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 4750, USD 5250, and USD 8750 respectively.
10. Is the market size provided in terms of value or volume?
The market size is provided in terms of value, measured in Million.
11. Are there any specific market keywords associated with the report?
Yes, the market keyword associated with the report is "Automated Sterility Testing Industry," which aids in identifying and referencing the specific market segment covered.
12. How do I determine which pricing option suits my needs best?
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
13. Are there any additional resources or data provided in the Automated Sterility Testing Industry report?
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
14. How can I stay updated on further developments or reports in the Automated Sterility Testing Industry?
To stay informed about further developments, trends, and reports in the Automated Sterility Testing Industry, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.
Methodology
Step 1 - Identification of Relevant Samples Size from Population Database
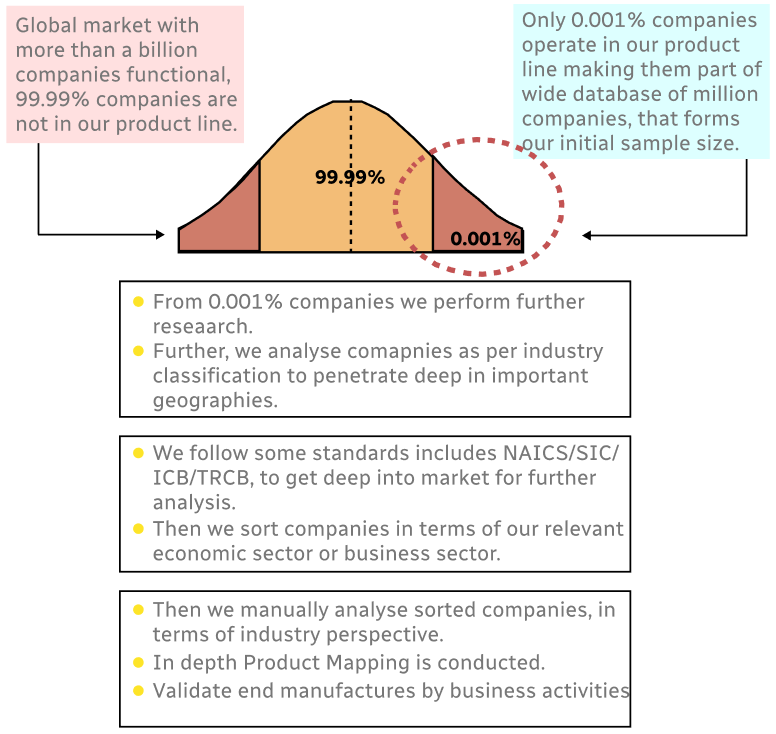
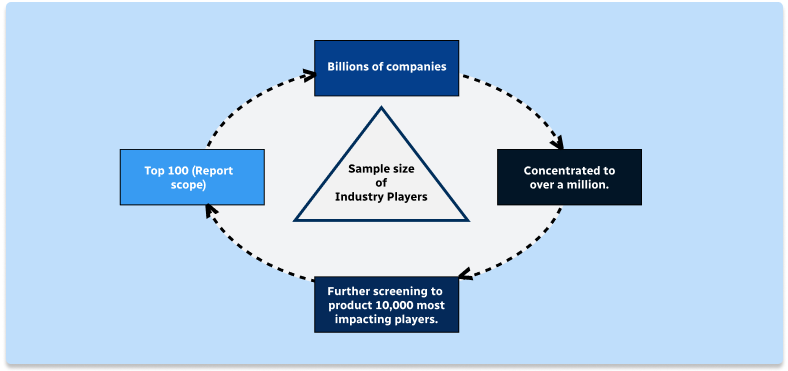
Step 2 - Approaches for Defining Global Market Size (Value, Volume* & Price*)
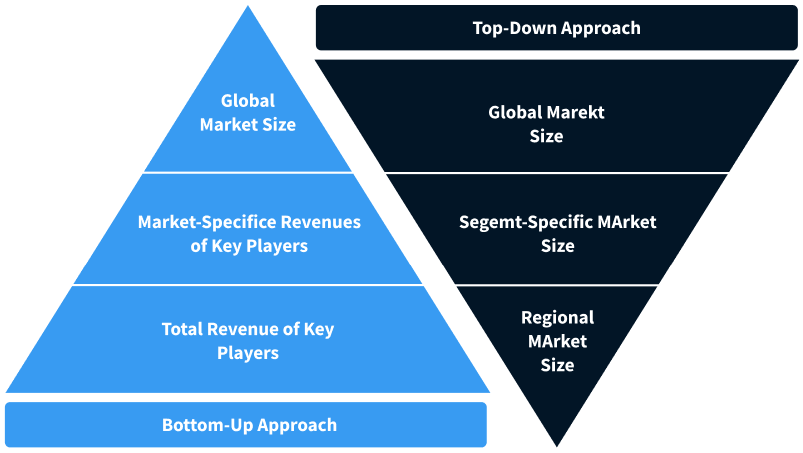
Note*: In applicable scenarios
Step 3 - Data Sources
Primary Research
- Web Analytics
- Survey Reports
- Research Institute
- Latest Research Reports
- Opinion Leaders
Secondary Research
- Annual Reports
- White Paper
- Latest Press Release
- Industry Association
- Paid Database
- Investor Presentations
Step 4 - Data Triangulation
Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence


