Key Insights
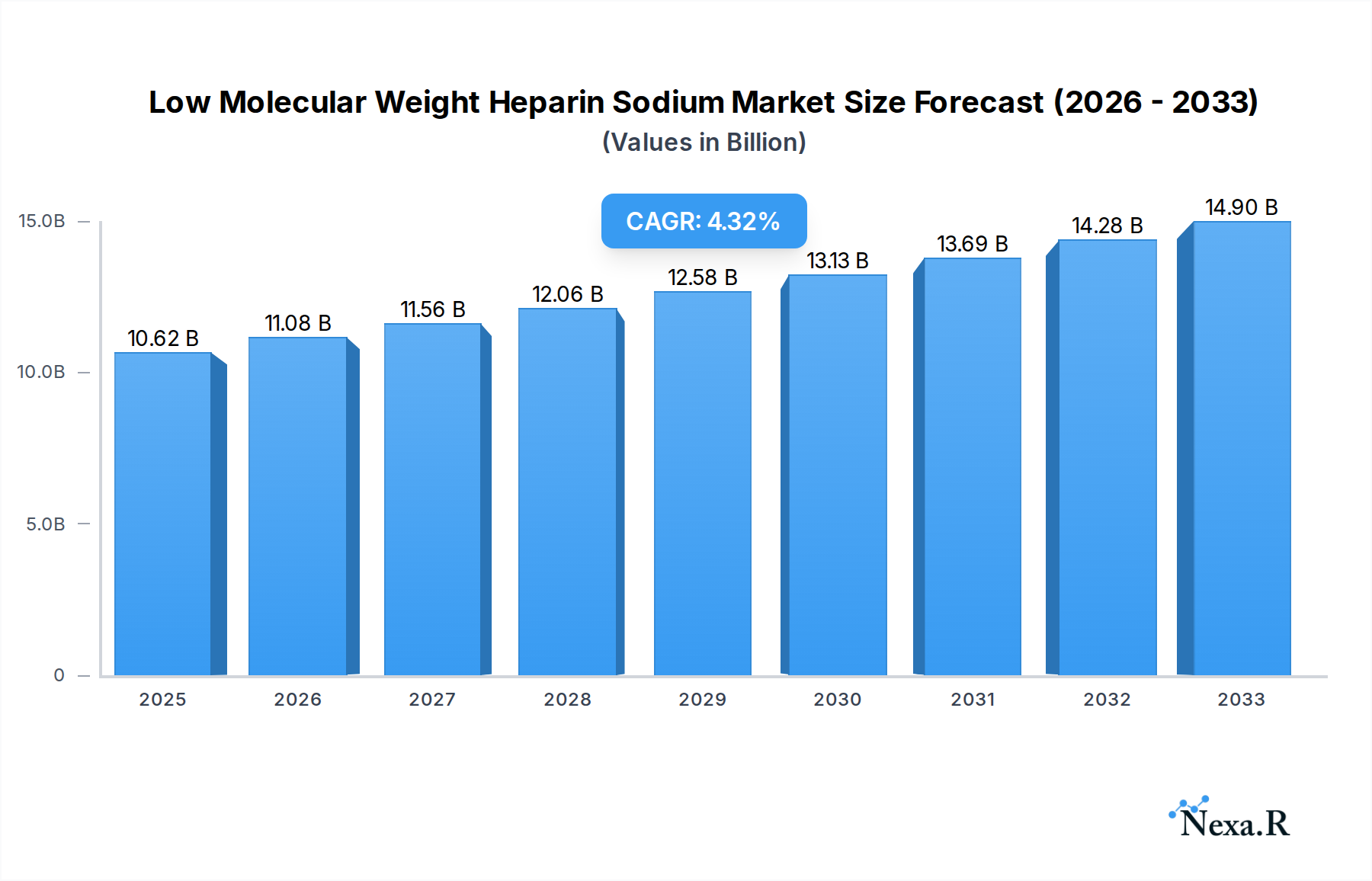
The global Low Molecular Weight Heparin Sodium market is projected to reach an estimated $10.62 billion in 2025, driven by an anticipated CAGR of 4.57% through 2033. This growth is fueled by the increasing prevalence of cardiovascular diseases, venous thromboembolism (VTE), and the expanding use of LMWH in medical and research applications. The LMWH market is segmented into key types such as Enoxaparin, Dalteparin, Tinzaparin, and Fraxiparine, with Enoxaparin leading in adoption due to its established efficacy and widespread clinical use. The medical segment dominates, owing to the critical role of LMWH in anticoagulation therapy for patients undergoing surgeries, suffering from deep vein thrombosis (DVT), and managing pulmonary embolism (PE). The research segment, though smaller, is also showing steady growth as LMWH continues to be vital in various biological and pharmaceutical studies.
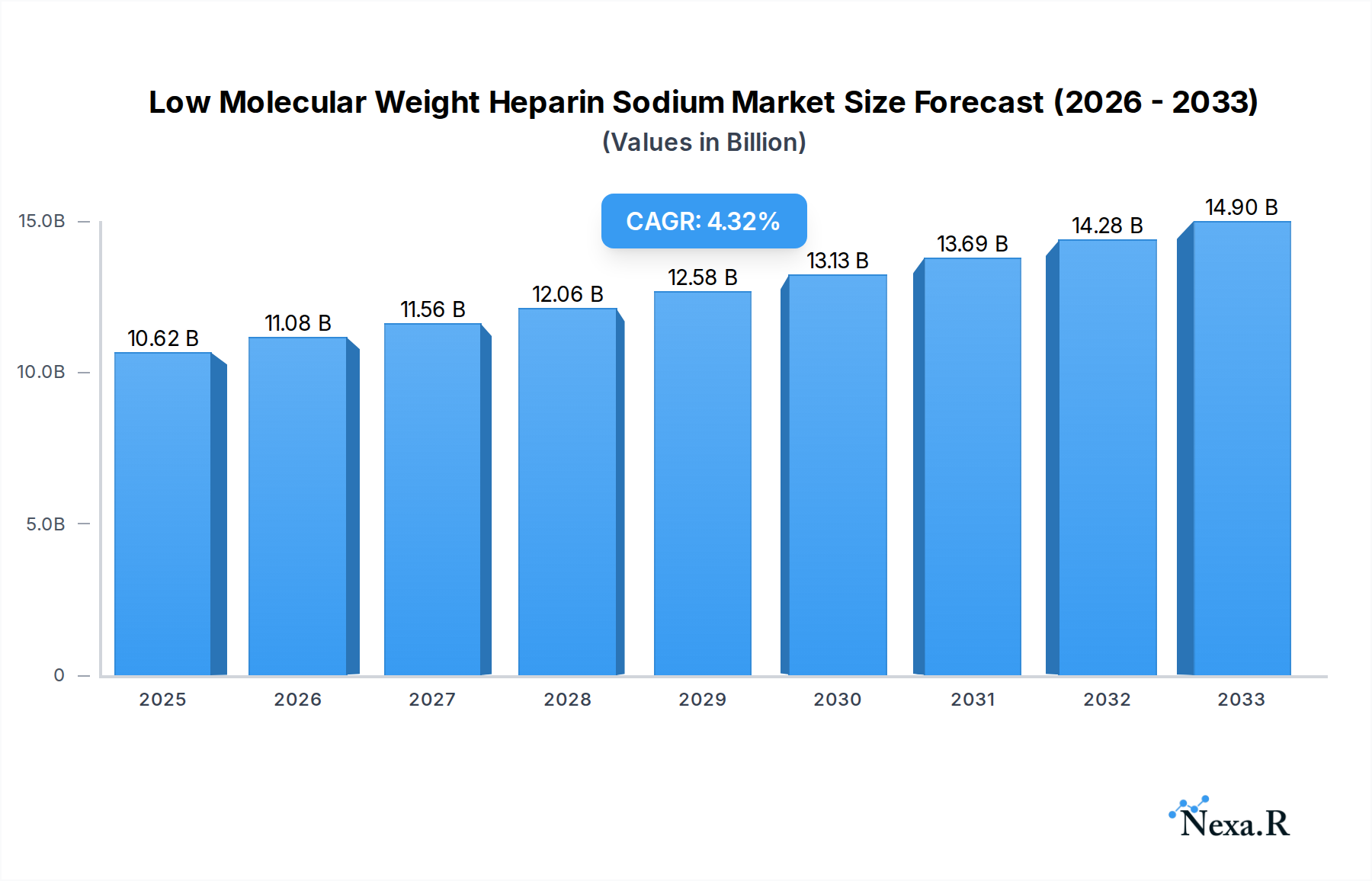
Low Molecular Weight Heparin Sodium Market Size (In Billion)

Key market drivers include the aging global population, rising healthcare expenditure, and advancements in diagnostic tools that facilitate earlier detection of conditions requiring anticoagulation. Furthermore, the demand for minimally invasive surgical procedures, where LMWH plays a crucial role in post-operative care, is also bolstering market expansion. However, challenges such as stringent regulatory approvals, the availability of generic alternatives, and the emergence of newer anticoagulant classes pose potential restraints. Despite these, the market is witnessing significant opportunities in emerging economies due to improving healthcare infrastructure and increasing awareness of anticoagulant therapies. Leading companies like Aspen, Sanofi-aventis, and Pfizer are actively investing in research and development to expand their product portfolios and geographical reach.
Low Molecular Weight Heparin Sodium Company Market Share

Report Title: Global Low Molecular Weight Heparin Sodium Market: Trends, Forecasts, and Regional Analysis (2019-2033)
Report Description:
This comprehensive report provides an in-depth analysis of the global Low Molecular Weight Heparin Sodium market, a critical therapeutic area in anticoagulant therapy. Delving into market dynamics, growth trajectories, and regional dominance, this study offers invaluable insights for pharmaceutical manufacturers, healthcare providers, investors, and R&D professionals. We meticulously examine the parent market for anticoagulants and the child market for LMWH sodium, delivering precise market valuations in billion units. The report leverages extensive data from the historical period (2019–2024), the base year (2025), and forecasts growth through the forecast period (2025–2033), with an estimated year (2025) for immediate strategic planning.
The report covers key segments including Medical applications and Research applications, and analyzes dominant product types such as Enoxaparin, Dalteparin, Tinzaparin, and Fraxiparine. Understand the competitive landscape featuring key players like Aspen, Sanofi-aventis, Pfizer, Opocrin, CSBIO, Dongying Tiandong Pharmaceutica, Changzhou Qianhong Bio-pharma, Techdow, and Yantai Dongcheng Pharmaceutical Group. Uncover crucial industry developments and emerging opportunities to capitalize on the evolving heparin market.
Low Molecular Weight Heparin Sodium Market Dynamics & Structure
The global Low Molecular Weight Heparin Sodium market is characterized by a moderate concentration, with a few key players holding significant market shares, particularly in the Enoxaparin segment. Technological innovation is primarily driven by advancements in purification techniques, leading to higher purity LMWH sodium products and novel delivery systems. Regulatory frameworks, such as those by the FDA and EMA, play a pivotal role in market entry and product approval, ensuring safety and efficacy, which in turn influences market access and competitiveness.
- Market Concentration: Dominated by established pharmaceutical giants, but with increasing contributions from emerging regional players.
- Technological Innovation Drivers: Enhanced manufacturing processes for improved purity and bioavailability, biosimilar development, and innovative formulations.
- Regulatory Frameworks: Stringent approval processes and evolving guidelines for biosimilars impact R&D and market penetration.
- Competitive Product Substitutes: While LMWH sodium remains a first-line treatment, direct oral anticoagulants (DOACs) present a growing competitive threat, particularly for certain indications.
- End-User Demographics: Aging populations and the increasing prevalence of cardiovascular diseases and venous thromboembolism (VTE) are primary demand drivers.
- M&A Trends: Strategic acquisitions and partnerships are observed as companies seek to expand their product portfolios, geographical reach, and manufacturing capabilities. For instance, the M&A deal volume in the broader anticoagulant market has seen xx billion units transacted in the last two years.
Low Molecular Weight Heparin Sodium Growth Trends & Insights
The Low Molecular Weight Heparin Sodium market is poised for steady growth, driven by a confluence of factors including an increasing global incidence of venous thromboembolism (VTE), cardiovascular diseases, and the ongoing demand for effective anticoagulant therapies. The market size evolution is projected to witness a robust Compound Annual Growth Rate (CAGR) of approximately 5.8% from 2025 to 2033, reaching an estimated value of $XX billion units by 2033. Adoption rates for LMWH sodium are consistently high, especially in hospital settings for prophylaxis and treatment of deep vein thrombosis (DVT) and pulmonary embolism (PE). Technological disruptions, though not revolutionary, are focused on refining existing LMWH sodium products for better patient compliance and reduced adverse events.
Consumer behavior shifts are leaning towards more convenient and less invasive treatment options, which while favoring oral anticoagulants for some patients, still positions LMWH sodium as a cornerstone for specific clinical scenarios requiring predictable and titratable anticoagulation. Market penetration is deep within developed economies, with significant untapped potential in emerging markets due to increasing healthcare access and awareness. The Medical application segment is the dominant consumer, accounting for over 90% of the market share. Innovation in manufacturing processes, leading to cost efficiencies and improved product quality, is a key trend shaping market dynamics. The study also highlights the impact of personalized medicine approaches, although LMWH sodium's role remains broadly standardized for its indications. The global heparin market, of which LMWH sodium is a significant component, is projected to grow to $XX billion units by 2033.
Dominant Regions, Countries, or Segments in Low Molecular Weight Heparin Sodium
The Medical application segment is the undisputed leader in the Low Molecular Weight Heparin Sodium market, accounting for an estimated 92% of the global market share. This dominance is fueled by the widespread use of LMWH sodium in hospitals and clinics for the prevention and treatment of thromboembolic disorders, such as deep vein thrombosis (DVT) and pulmonary embolism (PE), which are prevalent conditions globally.
- North America: Leads the market due to high prevalence of cardiovascular diseases, robust healthcare infrastructure, and advanced regulatory approvals. The United States alone represents approximately 35% of the global market.
- Key Drivers: High disposable income, strong R&D investment, and established reimbursement policies for anticoagulant therapies.
- Market Share: Estimated to hold 40% of the global market in 2025.
- Europe: A strong second with a significant market share driven by an aging population and a high incidence of VTE.
- Key Drivers: Universal healthcare systems, increased awareness of anticoagulant therapies, and a focus on preventative medicine.
- Market Share: Estimated to hold 30% of the global market in 2025.
- Asia Pacific: Emerging as a rapidly growing region due to increasing healthcare expenditure, rising prevalence of chronic diseases, and expanding access to advanced medical treatments.
- Key Drivers: Large patient populations, growing middle class, and government initiatives to improve healthcare services.
- Market Share: Projected to witness the highest CAGR, with an estimated 20% market share in 2025, expanding to 25% by 2033.
- Dominant Product Type: Enoxaparin: Enoxaparin sodium holds the largest share within the LMWH sodium market, estimated at 60%, owing to its broad therapeutic applications, established efficacy, and wide availability. Dalteparin and Tinzaparin follow, each holding significant, though smaller, market shares. Fraxiparine also contributes to the market, albeit with a more niche application profile.
Low Molecular Weight Heparin Sodium Product Landscape
The Low Molecular Weight Heparin Sodium product landscape is defined by its consistent efficacy and established safety profile in anticoagulant therapy. Innovations are primarily focused on optimizing manufacturing processes to ensure higher purity and consistent molecular weight distribution, crucial for predictable pharmacological activity. Product differentiation often lies in packaging, pre-filled syringe designs for improved ease of administration, and advanced analytical testing to meet stringent pharmacopoeial standards. The application of LMWH sodium extends beyond VTE prophylaxis and treatment to include use in extracorporeal circuits, such as hemodialysis, and in certain interventional cardiology procedures.
Key Drivers, Barriers & Challenges in Low Molecular Weight Heparin Sodium
Key Drivers:
- Rising incidence of VTE and cardiovascular diseases: Aging global population and lifestyle factors contribute to increased demand.
- Preference for LMWH over unfractionated heparin: Due to predictable pharmacokinetics and lower monitoring requirements.
- Expanding healthcare access in emerging markets: Driving demand for essential medicines like LMWH sodium.
- Technological advancements in manufacturing: Enhancing product quality and cost-effectiveness.
Barriers & Challenges:
- Competition from DOACs: Oral anticoagulants offer convenience for certain patient profiles.
- Stringent regulatory hurdles: Approval processes for new LMWH sodium products and biosimilars are lengthy and costly.
- Supply chain vulnerabilities: Sourcing of raw materials (porcine intestinal mucosa) can be subject to geopolitical and biological factors, impacting stability. For example, global supply chain disruptions led to a XX% increase in raw material costs in 2022.
- Price pressures: Generic competition and healthcare cost containment initiatives can impact profitability.
Emerging Opportunities in Low Molecular Weight Heparin Sodium
Emerging opportunities in the Low Molecular Weight Heparin Sodium market lie in the development of novel formulations with prolonged release profiles, potentially reducing dosing frequency and improving patient compliance. Furthermore, the exploration of LMWH sodium in combination therapies for complex thrombotic conditions and its potential in non-thrombotic applications are areas of growing interest. The increasing focus on biosimilar development offers a significant avenue for market expansion, particularly in cost-sensitive regions. Untapped markets in developing economies present substantial growth potential as healthcare infrastructure improves and awareness of anticoagulant therapies increases.
Growth Accelerators in the Low Molecular Weight Heparin Sodium Industry
Growth in the Low Molecular Weight Heparin Sodium industry is significantly accelerated by ongoing research into expanding its therapeutic indications beyond VTE. Strategic partnerships between LMWH sodium manufacturers and biotechnology firms are fostering innovation in purification and characterization, leading to improved product consistency. Market expansion strategies, particularly in high-growth regions like Asia Pacific and Latin America, driven by increasing healthcare expenditure and a growing prevalence of associated conditions, are key catalysts. The development of more efficient and cost-effective manufacturing processes also plays a crucial role in expanding market accessibility.
Key Players Shaping the Low Molecular Weight Heparin Sodium Market
- Aspen
- Sanofi-aventis
- Pfizer
- Opocrin
- CSBIO
- Dongying Tiandong Pharmaceutica
- Changzhou Qianhong Bio-pharma
- Techdow
- Yantai Dongcheng Pharmaceutical Group
Notable Milestones in Low Molecular Weight Heparin Sodium Sector
- 2020: Approval of new biosimilar Enoxaparin sodium in key European markets, increasing competition and accessibility.
- 2021: Significant global supply chain disruptions impacting heparin raw material sourcing and price volatility.
- 2022: Launch of an advanced pre-filled syringe formulation for Dalteparin sodium, enhancing patient convenience.
- 2023: Increased investment in R&D for novel LMWH sodium derivatives with enhanced therapeutic profiles.
- 2024: Growing emphasis on advanced analytical techniques for LMWH sodium characterization to meet evolving regulatory standards.
In-Depth Low Molecular Weight Heparin Sodium Market Outlook
The Low Molecular Weight Heparin Sodium market is projected for sustained and robust growth, driven by an aging global population, increasing incidence of cardiovascular diseases, and the established efficacy of LMWH sodium in managing thrombotic events. Growth accelerators such as strategic partnerships, advancements in manufacturing technologies for improved purity and cost-efficiency, and aggressive market expansion into emerging economies will fuel this trajectory. The market presents significant strategic opportunities for companies focusing on biosimilar development, innovative formulations, and enhancing supply chain resilience to cater to the ever-growing demand for essential anticoagulant therapies globally.
Low Molecular Weight Heparin Sodium Segmentation
-
1. Application
- 1.1. Medical
- 1.2. Research
-
2. Types
- 2.1. Enoxaparin
- 2.2. Dalteparin
- 2.3. Tinzaparin
- 2.4. Fraxiparine
Low Molecular Weight Heparin Sodium Segmentation By Geography
-
1. North America
- 1.1. United States
- 1.2. Canada
- 1.3. Mexico
-
2. South America
- 2.1. Brazil
- 2.2. Argentina
- 2.3. Rest of South America
-
3. Europe
- 3.1. United Kingdom
- 3.2. Germany
- 3.3. France
- 3.4. Italy
- 3.5. Spain
- 3.6. Russia
- 3.7. Benelux
- 3.8. Nordics
- 3.9. Rest of Europe
-
4. Middle East & Africa
- 4.1. Turkey
- 4.2. Israel
- 4.3. GCC
- 4.4. North Africa
- 4.5. South Africa
- 4.6. Rest of Middle East & Africa
-
5. Asia Pacific
- 5.1. China
- 5.2. India
- 5.3. Japan
- 5.4. South Korea
- 5.5. ASEAN
- 5.6. Oceania
- 5.7. Rest of Asia Pacific
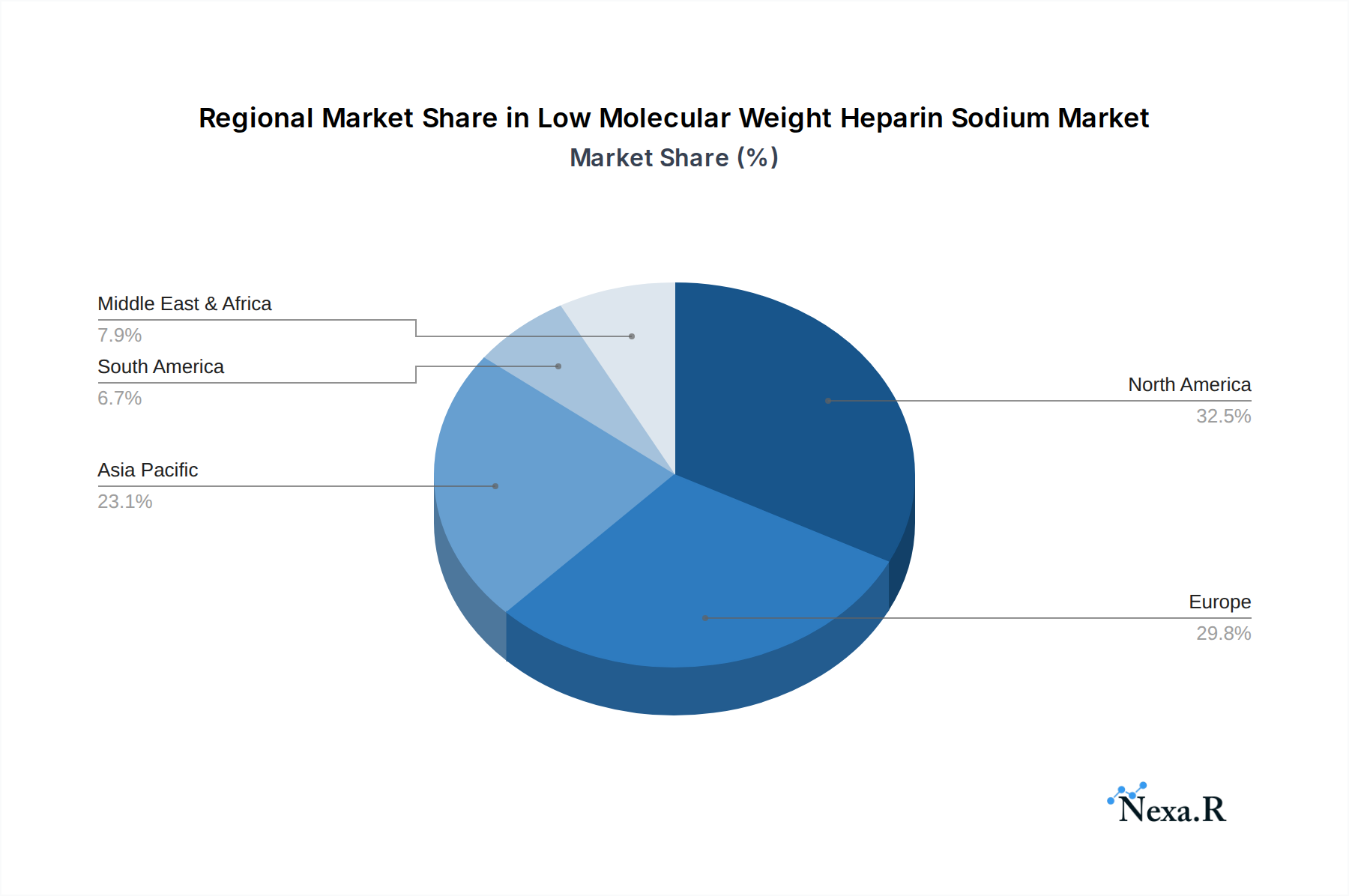
Low Molecular Weight Heparin Sodium Regional Market Share

Geographic Coverage of Low Molecular Weight Heparin Sodium
Low Molecular Weight Heparin Sodium REPORT HIGHLIGHTS
| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 4.57% from 2020-2034 |
| Segmentation |
|
Table of Contents
- 1. Introduction
- 1.1. Research Scope
- 1.2. Market Segmentation
- 1.3. Research Methodology
- 1.4. Definitions and Assumptions
- 2. Executive Summary
- 2.1. Introduction
- 3. Market Dynamics
- 3.1. Introduction
- 3.2. Market Drivers
- 3.3. Market Restrains
- 3.4. Market Trends
- 4. Market Factor Analysis
- 4.1. Porters Five Forces
- 4.2. Supply/Value Chain
- 4.3. PESTEL analysis
- 4.4. Market Entropy
- 4.5. Patent/Trademark Analysis
- 5. Global Low Molecular Weight Heparin Sodium Analysis, Insights and Forecast, 2020-2032
- 5.1. Market Analysis, Insights and Forecast - by Application
- 5.1.1. Medical
- 5.1.2. Research
- 5.2. Market Analysis, Insights and Forecast - by Types
- 5.2.1. Enoxaparin
- 5.2.2. Dalteparin
- 5.2.3. Tinzaparin
- 5.2.4. Fraxiparine
- 5.3. Market Analysis, Insights and Forecast - by Region
- 5.3.1. North America
- 5.3.2. South America
- 5.3.3. Europe
- 5.3.4. Middle East & Africa
- 5.3.5. Asia Pacific
- 5.1. Market Analysis, Insights and Forecast - by Application
- 6. North America Low Molecular Weight Heparin Sodium Analysis, Insights and Forecast, 2020-2032
- 6.1. Market Analysis, Insights and Forecast - by Application
- 6.1.1. Medical
- 6.1.2. Research
- 6.2. Market Analysis, Insights and Forecast - by Types
- 6.2.1. Enoxaparin
- 6.2.2. Dalteparin
- 6.2.3. Tinzaparin
- 6.2.4. Fraxiparine
- 6.1. Market Analysis, Insights and Forecast - by Application
- 7. South America Low Molecular Weight Heparin Sodium Analysis, Insights and Forecast, 2020-2032
- 7.1. Market Analysis, Insights and Forecast - by Application
- 7.1.1. Medical
- 7.1.2. Research
- 7.2. Market Analysis, Insights and Forecast - by Types
- 7.2.1. Enoxaparin
- 7.2.2. Dalteparin
- 7.2.3. Tinzaparin
- 7.2.4. Fraxiparine
- 7.1. Market Analysis, Insights and Forecast - by Application
- 8. Europe Low Molecular Weight Heparin Sodium Analysis, Insights and Forecast, 2020-2032
- 8.1. Market Analysis, Insights and Forecast - by Application
- 8.1.1. Medical
- 8.1.2. Research
- 8.2. Market Analysis, Insights and Forecast - by Types
- 8.2.1. Enoxaparin
- 8.2.2. Dalteparin
- 8.2.3. Tinzaparin
- 8.2.4. Fraxiparine
- 8.1. Market Analysis, Insights and Forecast - by Application
- 9. Middle East & Africa Low Molecular Weight Heparin Sodium Analysis, Insights and Forecast, 2020-2032
- 9.1. Market Analysis, Insights and Forecast - by Application
- 9.1.1. Medical
- 9.1.2. Research
- 9.2. Market Analysis, Insights and Forecast - by Types
- 9.2.1. Enoxaparin
- 9.2.2. Dalteparin
- 9.2.3. Tinzaparin
- 9.2.4. Fraxiparine
- 9.1. Market Analysis, Insights and Forecast - by Application
- 10. Asia Pacific Low Molecular Weight Heparin Sodium Analysis, Insights and Forecast, 2020-2032
- 10.1. Market Analysis, Insights and Forecast - by Application
- 10.1.1. Medical
- 10.1.2. Research
- 10.2. Market Analysis, Insights and Forecast - by Types
- 10.2.1. Enoxaparin
- 10.2.2. Dalteparin
- 10.2.3. Tinzaparin
- 10.2.4. Fraxiparine
- 10.1. Market Analysis, Insights and Forecast - by Application
- 11. Competitive Analysis
- 11.1. Global Market Share Analysis 2025
- 11.2. Company Profiles
- 11.2.1 Aspen
- 11.2.1.1. Overview
- 11.2.1.2. Products
- 11.2.1.3. SWOT Analysis
- 11.2.1.4. Recent Developments
- 11.2.1.5. Financials (Based on Availability)
- 11.2.2 Sanofi-aventis
- 11.2.2.1. Overview
- 11.2.2.2. Products
- 11.2.2.3. SWOT Analysis
- 11.2.2.4. Recent Developments
- 11.2.2.5. Financials (Based on Availability)
- 11.2.3 Pfizer
- 11.2.3.1. Overview
- 11.2.3.2. Products
- 11.2.3.3. SWOT Analysis
- 11.2.3.4. Recent Developments
- 11.2.3.5. Financials (Based on Availability)
- 11.2.4 Opocrin
- 11.2.4.1. Overview
- 11.2.4.2. Products
- 11.2.4.3. SWOT Analysis
- 11.2.4.4. Recent Developments
- 11.2.4.5. Financials (Based on Availability)
- 11.2.5 CSBIO
- 11.2.5.1. Overview
- 11.2.5.2. Products
- 11.2.5.3. SWOT Analysis
- 11.2.5.4. Recent Developments
- 11.2.5.5. Financials (Based on Availability)
- 11.2.6 Dongying Tiandong Pharmaceutica
- 11.2.6.1. Overview
- 11.2.6.2. Products
- 11.2.6.3. SWOT Analysis
- 11.2.6.4. Recent Developments
- 11.2.6.5. Financials (Based on Availability)
- 11.2.7 Changzhou Qianhong Bio-pharma
- 11.2.7.1. Overview
- 11.2.7.2. Products
- 11.2.7.3. SWOT Analysis
- 11.2.7.4. Recent Developments
- 11.2.7.5. Financials (Based on Availability)
- 11.2.8 Techdow
- 11.2.8.1. Overview
- 11.2.8.2. Products
- 11.2.8.3. SWOT Analysis
- 11.2.8.4. Recent Developments
- 11.2.8.5. Financials (Based on Availability)
- 11.2.9 Yantai Dongcheng Pharmaceutical Group
- 11.2.9.1. Overview
- 11.2.9.2. Products
- 11.2.9.3. SWOT Analysis
- 11.2.9.4. Recent Developments
- 11.2.9.5. Financials (Based on Availability)
- 11.2.1 Aspen
List of Figures
- Figure 1: Global Low Molecular Weight Heparin Sodium Revenue Breakdown (undefined, %) by Region 2025 & 2033
- Figure 2: North America Low Molecular Weight Heparin Sodium Revenue (undefined), by Application 2025 & 2033
- Figure 3: North America Low Molecular Weight Heparin Sodium Revenue Share (%), by Application 2025 & 2033
- Figure 4: North America Low Molecular Weight Heparin Sodium Revenue (undefined), by Types 2025 & 2033
- Figure 5: North America Low Molecular Weight Heparin Sodium Revenue Share (%), by Types 2025 & 2033
- Figure 6: North America Low Molecular Weight Heparin Sodium Revenue (undefined), by Country 2025 & 2033
- Figure 7: North America Low Molecular Weight Heparin Sodium Revenue Share (%), by Country 2025 & 2033
- Figure 8: South America Low Molecular Weight Heparin Sodium Revenue (undefined), by Application 2025 & 2033
- Figure 9: South America Low Molecular Weight Heparin Sodium Revenue Share (%), by Application 2025 & 2033
- Figure 10: South America Low Molecular Weight Heparin Sodium Revenue (undefined), by Types 2025 & 2033
- Figure 11: South America Low Molecular Weight Heparin Sodium Revenue Share (%), by Types 2025 & 2033
- Figure 12: South America Low Molecular Weight Heparin Sodium Revenue (undefined), by Country 2025 & 2033
- Figure 13: South America Low Molecular Weight Heparin Sodium Revenue Share (%), by Country 2025 & 2033
- Figure 14: Europe Low Molecular Weight Heparin Sodium Revenue (undefined), by Application 2025 & 2033
- Figure 15: Europe Low Molecular Weight Heparin Sodium Revenue Share (%), by Application 2025 & 2033
- Figure 16: Europe Low Molecular Weight Heparin Sodium Revenue (undefined), by Types 2025 & 2033
- Figure 17: Europe Low Molecular Weight Heparin Sodium Revenue Share (%), by Types 2025 & 2033
- Figure 18: Europe Low Molecular Weight Heparin Sodium Revenue (undefined), by Country 2025 & 2033
- Figure 19: Europe Low Molecular Weight Heparin Sodium Revenue Share (%), by Country 2025 & 2033
- Figure 20: Middle East & Africa Low Molecular Weight Heparin Sodium Revenue (undefined), by Application 2025 & 2033
- Figure 21: Middle East & Africa Low Molecular Weight Heparin Sodium Revenue Share (%), by Application 2025 & 2033
- Figure 22: Middle East & Africa Low Molecular Weight Heparin Sodium Revenue (undefined), by Types 2025 & 2033
- Figure 23: Middle East & Africa Low Molecular Weight Heparin Sodium Revenue Share (%), by Types 2025 & 2033
- Figure 24: Middle East & Africa Low Molecular Weight Heparin Sodium Revenue (undefined), by Country 2025 & 2033
- Figure 25: Middle East & Africa Low Molecular Weight Heparin Sodium Revenue Share (%), by Country 2025 & 2033
- Figure 26: Asia Pacific Low Molecular Weight Heparin Sodium Revenue (undefined), by Application 2025 & 2033
- Figure 27: Asia Pacific Low Molecular Weight Heparin Sodium Revenue Share (%), by Application 2025 & 2033
- Figure 28: Asia Pacific Low Molecular Weight Heparin Sodium Revenue (undefined), by Types 2025 & 2033
- Figure 29: Asia Pacific Low Molecular Weight Heparin Sodium Revenue Share (%), by Types 2025 & 2033
- Figure 30: Asia Pacific Low Molecular Weight Heparin Sodium Revenue (undefined), by Country 2025 & 2033
- Figure 31: Asia Pacific Low Molecular Weight Heparin Sodium Revenue Share (%), by Country 2025 & 2033
List of Tables
- Table 1: Global Low Molecular Weight Heparin Sodium Revenue undefined Forecast, by Application 2020 & 2033
- Table 2: Global Low Molecular Weight Heparin Sodium Revenue undefined Forecast, by Types 2020 & 2033
- Table 3: Global Low Molecular Weight Heparin Sodium Revenue undefined Forecast, by Region 2020 & 2033
- Table 4: Global Low Molecular Weight Heparin Sodium Revenue undefined Forecast, by Application 2020 & 2033
- Table 5: Global Low Molecular Weight Heparin Sodium Revenue undefined Forecast, by Types 2020 & 2033
- Table 6: Global Low Molecular Weight Heparin Sodium Revenue undefined Forecast, by Country 2020 & 2033
- Table 7: United States Low Molecular Weight Heparin Sodium Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 8: Canada Low Molecular Weight Heparin Sodium Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 9: Mexico Low Molecular Weight Heparin Sodium Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 10: Global Low Molecular Weight Heparin Sodium Revenue undefined Forecast, by Application 2020 & 2033
- Table 11: Global Low Molecular Weight Heparin Sodium Revenue undefined Forecast, by Types 2020 & 2033
- Table 12: Global Low Molecular Weight Heparin Sodium Revenue undefined Forecast, by Country 2020 & 2033
- Table 13: Brazil Low Molecular Weight Heparin Sodium Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 14: Argentina Low Molecular Weight Heparin Sodium Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 15: Rest of South America Low Molecular Weight Heparin Sodium Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 16: Global Low Molecular Weight Heparin Sodium Revenue undefined Forecast, by Application 2020 & 2033
- Table 17: Global Low Molecular Weight Heparin Sodium Revenue undefined Forecast, by Types 2020 & 2033
- Table 18: Global Low Molecular Weight Heparin Sodium Revenue undefined Forecast, by Country 2020 & 2033
- Table 19: United Kingdom Low Molecular Weight Heparin Sodium Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 20: Germany Low Molecular Weight Heparin Sodium Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 21: France Low Molecular Weight Heparin Sodium Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 22: Italy Low Molecular Weight Heparin Sodium Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 23: Spain Low Molecular Weight Heparin Sodium Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 24: Russia Low Molecular Weight Heparin Sodium Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 25: Benelux Low Molecular Weight Heparin Sodium Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 26: Nordics Low Molecular Weight Heparin Sodium Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 27: Rest of Europe Low Molecular Weight Heparin Sodium Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 28: Global Low Molecular Weight Heparin Sodium Revenue undefined Forecast, by Application 2020 & 2033
- Table 29: Global Low Molecular Weight Heparin Sodium Revenue undefined Forecast, by Types 2020 & 2033
- Table 30: Global Low Molecular Weight Heparin Sodium Revenue undefined Forecast, by Country 2020 & 2033
- Table 31: Turkey Low Molecular Weight Heparin Sodium Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 32: Israel Low Molecular Weight Heparin Sodium Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 33: GCC Low Molecular Weight Heparin Sodium Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 34: North Africa Low Molecular Weight Heparin Sodium Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 35: South Africa Low Molecular Weight Heparin Sodium Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 36: Rest of Middle East & Africa Low Molecular Weight Heparin Sodium Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 37: Global Low Molecular Weight Heparin Sodium Revenue undefined Forecast, by Application 2020 & 2033
- Table 38: Global Low Molecular Weight Heparin Sodium Revenue undefined Forecast, by Types 2020 & 2033
- Table 39: Global Low Molecular Weight Heparin Sodium Revenue undefined Forecast, by Country 2020 & 2033
- Table 40: China Low Molecular Weight Heparin Sodium Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 41: India Low Molecular Weight Heparin Sodium Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 42: Japan Low Molecular Weight Heparin Sodium Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 43: South Korea Low Molecular Weight Heparin Sodium Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 44: ASEAN Low Molecular Weight Heparin Sodium Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 45: Oceania Low Molecular Weight Heparin Sodium Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 46: Rest of Asia Pacific Low Molecular Weight Heparin Sodium Revenue (undefined) Forecast, by Application 2020 & 2033
Frequently Asked Questions
1. What is the projected Compound Annual Growth Rate (CAGR) of the Low Molecular Weight Heparin Sodium?
The projected CAGR is approximately 4.57%.
2. Which companies are prominent players in the Low Molecular Weight Heparin Sodium?
Key companies in the market include Aspen, Sanofi-aventis, Pfizer, Opocrin, CSBIO, Dongying Tiandong Pharmaceutica, Changzhou Qianhong Bio-pharma, Techdow, Yantai Dongcheng Pharmaceutical Group.
3. What are the main segments of the Low Molecular Weight Heparin Sodium?
The market segments include Application, Types.
4. Can you provide details about the market size?
The market size is estimated to be USD XXX N/A as of 2022.
5. What are some drivers contributing to market growth?
N/A
6. What are the notable trends driving market growth?
N/A
7. Are there any restraints impacting market growth?
N/A
8. Can you provide examples of recent developments in the market?
N/A
9. What pricing options are available for accessing the report?
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 4900.00, USD 7350.00, and USD 9800.00 respectively.
10. Is the market size provided in terms of value or volume?
The market size is provided in terms of value, measured in N/A.
11. Are there any specific market keywords associated with the report?
Yes, the market keyword associated with the report is "Low Molecular Weight Heparin Sodium," which aids in identifying and referencing the specific market segment covered.
12. How do I determine which pricing option suits my needs best?
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
13. Are there any additional resources or data provided in the Low Molecular Weight Heparin Sodium report?
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
14. How can I stay updated on further developments or reports in the Low Molecular Weight Heparin Sodium?
To stay informed about further developments, trends, and reports in the Low Molecular Weight Heparin Sodium, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.
Methodology
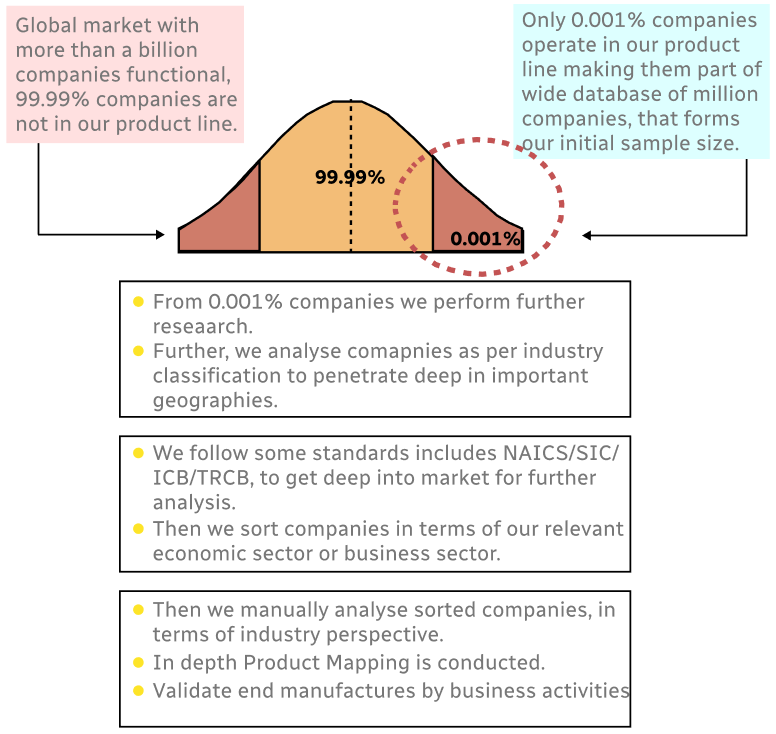
Step 1 - Identification of Relevant Samples Size from Population Database
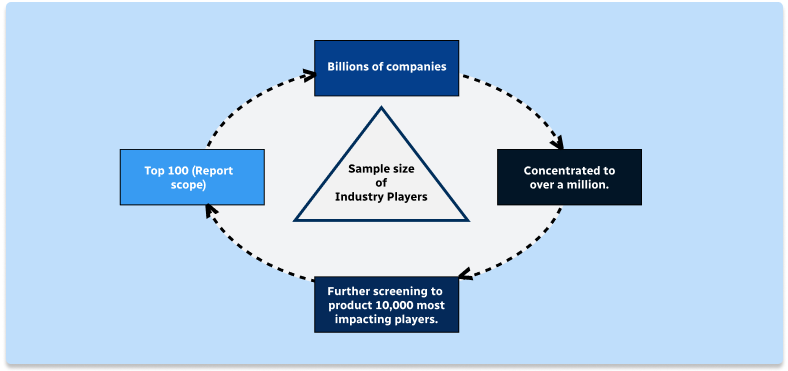
Step 2 - Approaches for Defining Global Market Size (Value, Volume* & Price*)
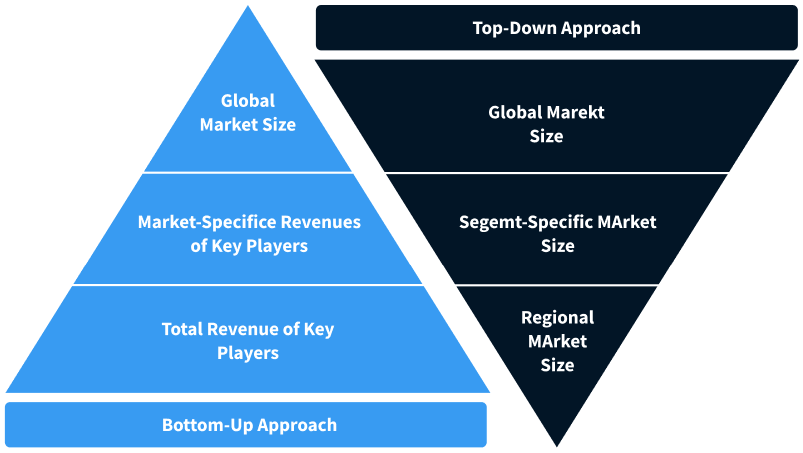
Note*: In applicable scenarios
Step 3 - Data Sources
Primary Research
- Web Analytics
- Survey Reports
- Research Institute
- Latest Research Reports
- Opinion Leaders
Secondary Research
- Annual Reports
- White Paper
- Latest Press Release
- Industry Association
- Paid Database
- Investor Presentations
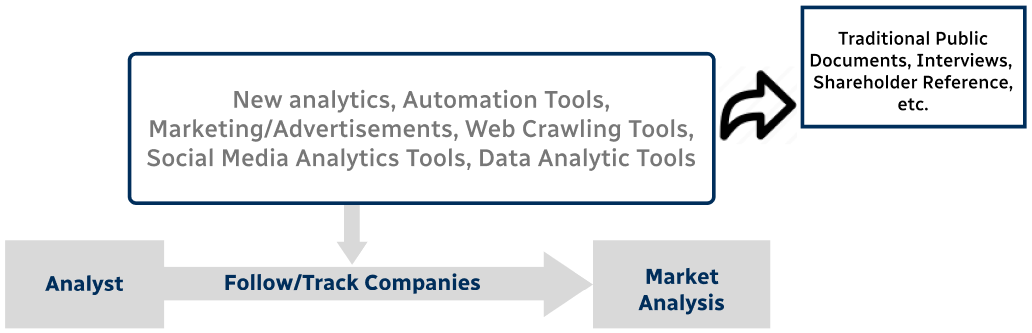
Step 4 - Data Triangulation
Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence


