Key Insights
The global Pertussis Booster Vaccine market is poised for significant expansion, projected to reach an estimated $6.9 billion in 2025 and expand at a robust Compound Annual Growth Rate (CAGR) of 5% through the forecast period of 2025-2033. This growth is propelled by a confluence of factors including the persistent threat of pertussis outbreaks, increasing awareness among parents and healthcare providers regarding the importance of timely booster vaccinations, and government initiatives aimed at enhancing immunization coverage. The rising incidence of whooping cough, particularly in developed nations where immunity wanes over time, underscores the critical need for these booster doses. Furthermore, advancements in vaccine technology, leading to the development of more effective and potentially safer acellular vaccines, are also contributing to market momentum. Pediatric populations, representing a substantial segment for booster doses, are a primary focus, alongside an emerging demand for adolescent and adult boosters to mitigate community transmission.
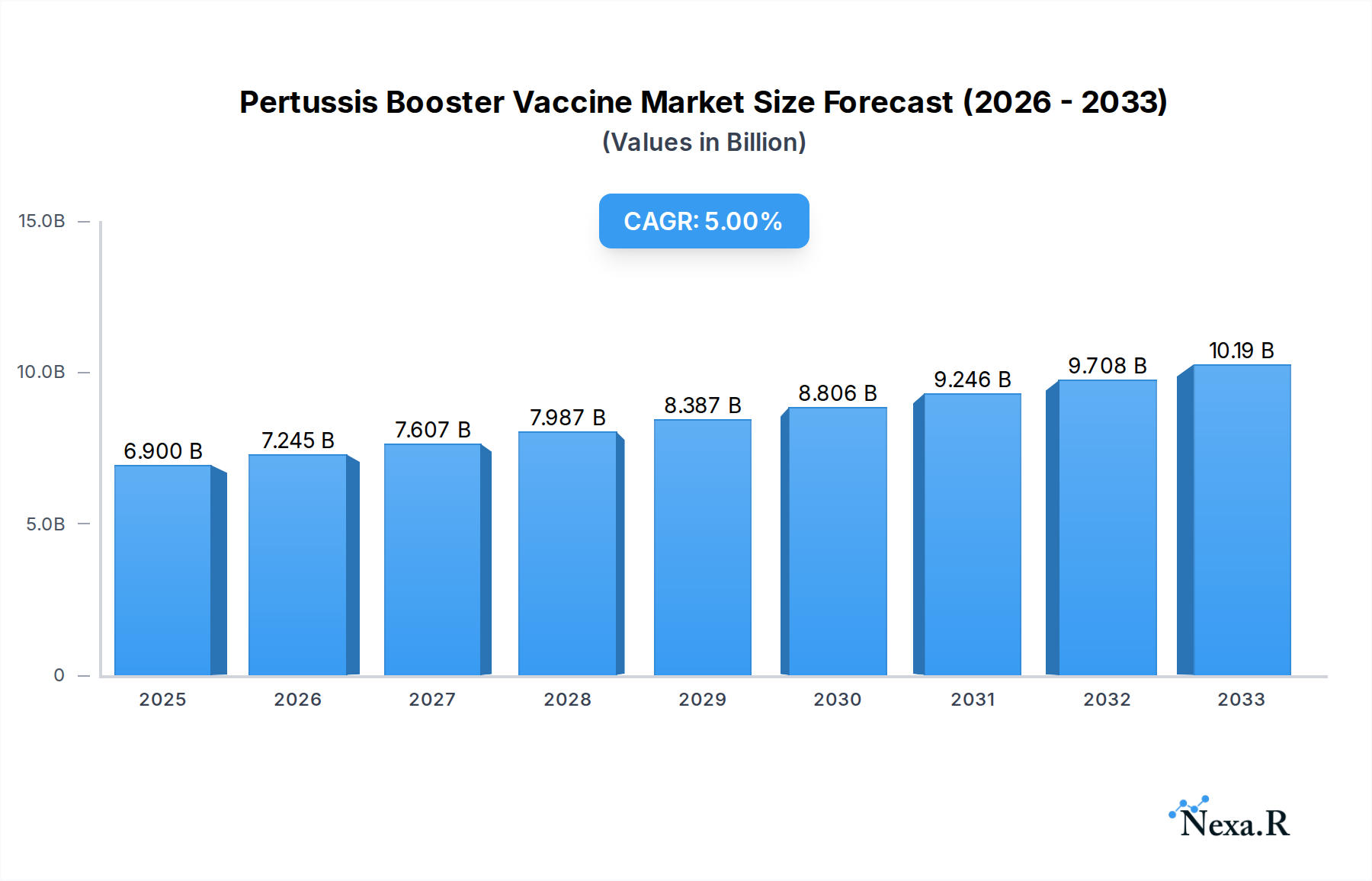
Pertussis Booster Vaccine Market Size (In Billion)

The market's trajectory is further shaped by evolving public health policies and the ongoing efforts of major pharmaceutical companies to develop and distribute innovative pertussis vaccines. Key drivers include the establishment of routine immunization schedules that incorporate booster doses and the strategic expansion of manufacturing capabilities by leading players such as Sanofi Pasteur and GSK. While the market exhibits strong growth potential, certain challenges such as vaccine hesitancy and the complexities of cold chain logistics in certain regions could present minor headwinds. However, the overarching trend points towards a sustained demand for pertussis booster vaccines as a vital public health tool, with market segmentation anticipated to show robust performance across both acellular and whole-cell vaccine types, catering to diverse age groups and healthcare requirements. The Asia Pacific region, with its large populations and increasing focus on public health infrastructure, is expected to be a significant contributor to market growth.
Pertussis Booster Vaccine Company Market Share

Pertussis Booster Vaccine Market: Comprehensive Analysis and Future Outlook (2019-2033)
This in-depth report provides an exhaustive analysis of the global Pertussis Booster Vaccine market, meticulously examining its current landscape, historical trajectory, and future potential. Leveraging high-traffic keywords and segmenting the market by parent and child applications, this report offers critical insights for industry professionals, investors, and stakeholders. We delve into market dynamics, growth trends, regional dominance, product innovation, key drivers, challenges, emerging opportunities, and strategic growth accelerators, all underpinned by robust data and expert analysis. The study period spans from 2019 to 2033, with a base year of 2025, providing a comprehensive view of both past performance and projected future evolution.
Pertussis Booster Vaccine Market Dynamics & Structure
The global Pertussis Booster Vaccine market exhibits a moderate to high concentration, dominated by a few key players. Technological innovation, particularly in developing more effective and safer acellular vaccine formulations, is a significant driver. Stringent regulatory frameworks from health authorities like the FDA and EMA ensure vaccine safety and efficacy, influencing product development and market entry. Competitive product substitutes, while limited within the booster segment for pertussis, may arise from novel combination vaccines or alternative preventative health strategies. End-user demographics are primarily driven by national immunization schedules and public health initiatives, impacting demand for both infant and adolescent/adult boosters. Mergers and acquisitions (M&A) activity, though not extremely high, is strategically focused on acquiring innovative technologies or expanding manufacturing capabilities.
- Market Concentration: Dominated by a few major pharmaceutical companies.
- Technological Innovation: Focus on enhanced immunogenicity and reduced side effects in acellular vaccines.
- Regulatory Frameworks: Strict approval processes and post-market surveillance by global health agencies.
- Competitive Landscape: Limited direct substitutes for pertussis boosters, but combination vaccines offer indirect competition.
- End-User Demographics: Driven by routine childhood immunizations and adolescent/adult catch-up programs.
- M&A Trends: Strategic acquisitions to bolster product portfolios and R&D capabilities.
Pertussis Booster Vaccine Growth Trends & Insights
The global Pertussis Booster Vaccine market has witnessed consistent growth, propelled by rising awareness of pertussis (whooping cough) and the necessity of maintaining vaccine-induced immunity. The market size evolution is characterized by steady increases, reflecting ongoing vaccination programs and the global effort to control outbreaks. Adoption rates for booster doses are largely dictated by public health policies and the efficacy of existing vaccines in preventing disease transmission. Technological disruptions have primarily centered around the refinement of acellular pertussis vaccines, which offer improved safety profiles compared to older whole-cell vaccines, leading to greater acceptance and wider application, especially in developed nations. Consumer behavior shifts, influenced by public health campaigns and parental concerns regarding vaccine-preventable diseases, have also played a crucial role in driving demand. The forecast period anticipates continued expansion, supported by global health initiatives and the ongoing need for herd immunity. Estimated market size for 2025 is projected at xx billion units, with a compound annual growth rate (CAGR) of xx% expected through 2033. Market penetration is already high in developed countries but continues to expand in emerging economies as healthcare infrastructure improves.
Dominant Regions, Countries, or Segments in Pertussis Booster Vaccine
North America, particularly the United States, currently dominates the Pertussis Booster Vaccine market. This regional dominance is fueled by well-established public health infrastructure, robust national immunization schedules that mandate booster doses, and high public awareness regarding the importance of pertussis prevention. The robust economic policies supporting healthcare access and the proactive stance of regulatory bodies in ensuring vaccine availability contribute significantly to this leadership. Within the Application segment, "12 Months Above" is the most dominant, encompassing the crucial adolescent and adult booster doses, which are increasingly recognized as vital for sustained immunity and preventing resurgence of the disease.
Key drivers for North America's dominance include:
- Strong Public Health Programs: Comprehensive childhood and adolescent immunization schedules.
- High Healthcare Spending: Significant investment in preventative healthcare and vaccine procurement.
- Regulatory Support: Proactive approval and recommendation of booster vaccines.
- Public Awareness: High parental and individual understanding of vaccine benefits.
In terms of Type, Acellular Vaccines hold the dominant position. Their superior safety profile, characterized by fewer adverse events compared to whole-cell vaccines, has made them the preferred choice for booster administrations across most developed nations. This preference is driven by a combination of medical recommendations, patient comfort, and the desire to minimize any potential barriers to vaccination. The market share for acellular vaccines is estimated at xx% in 2025, with significant growth potential attributed to ongoing research and development aimed at further enhancing their immunogenicity.
Pertussis Booster Vaccine Product Landscape
The product landscape for Pertussis Booster Vaccines is characterized by a focus on acellular formulations, primarily targeting the adolescent and adult populations. Leading companies are investing in research and development to optimize immunogenicity and duration of protection. These vaccines are typically administered as part of combination shots, such as Tdap (Tetanus, Diphtheria, and Acellular Pertussis), offering convenience and broad protection against multiple diseases. Unique selling propositions often revolve around established safety profiles, ease of administration, and compatibility with existing vaccination schedules. Technological advancements are geared towards improving antigen delivery and adjuvant technologies to elicit stronger and longer-lasting immune responses.
Key Drivers, Barriers & Challenges in Pertussis Booster Vaccine
Key Drivers:
- Rising Pertussis Incidence: Periodic outbreaks and concerns about waning immunity necessitate booster doses.
- Government Immunization Programs: Mandated booster schedules in many countries.
- Technological Advancements: Development of safer and more effective acellular vaccines.
- Increased Public Health Awareness: Greater understanding of vaccine-preventable diseases.
Key Barriers & Challenges:
- Waning Immunity: The need for boosters due to decreasing immunity over time.
- Vaccine Hesitancy: Persistent concerns and misinformation regarding vaccine safety and efficacy.
- Cold Chain Logistics: Maintaining the integrity of vaccines during transport and storage, especially in developing regions.
- Regulatory Hurdles: Stringent and time-consuming approval processes for new vaccine formulations.
- Competitive Pricing: Pressure from generic manufacturers and government tender processes.
- Supply Chain Disruptions: Potential for disruptions due to manufacturing issues or global events.
Emerging Opportunities in Pertussis Booster Vaccine
Emerging opportunities in the Pertussis Booster Vaccine sector lie in expanding market penetration in underserved regions and developing novel combination vaccines that offer broader protection with fewer injections. Untapped markets in lower and middle-income countries present significant growth potential as healthcare infrastructure improves. Innovative applications could involve research into enhanced adjuvant technologies to prolong immunity or address specific demographic needs. Evolving consumer preferences are leaning towards vaccines with minimized side effects and convenient administration schedules, creating a demand for next-generation boosters. The development of vaccines targeting specific age groups with tailored immunogenicity profiles also represents a significant avenue for growth.
Growth Accelerators in the Pertussis Booster Vaccine Industry
Long-term growth in the Pertussis Booster Vaccine industry is being catalyzed by several key factors. Technological breakthroughs in vaccine development, particularly in recombinant DNA technology and novel adjuvant systems, are enabling the creation of more potent and durable vaccines. Strategic partnerships between pharmaceutical companies and research institutions are accelerating the pace of innovation and clinical trials. Furthermore, market expansion strategies, including targeted outreach programs in emerging economies and collaborations with international health organizations, are crucial for increasing global access and uptake. The increasing recognition of pertussis as a threat to all age groups, not just infants, is driving the demand for adult and adolescent boosters, a significant growth accelerator.
Key Players Shaping the Pertussis Booster Vaccine Market
- Sanofi Pasteur
- GSK
- Mitsubishi Tanabe Pharma
- KM Biologics
- Wuhan Institute of Biological Products
- Walvax Biotechnology
- Chengdu Institute of Biological Products
- Minhai Biotechnology
Notable Milestones in Pertussis Booster Vaccine Sector
- 2019: Continued advancements in acellular pertussis vaccine formulations, leading to improved efficacy data.
- 2020: Increased focus on pertussis booster campaigns due to observed rises in whooping cough cases in several regions.
- 2021: Significant investment in R&D for next-generation combination vaccines including acellular pertussis components.
- 2022: Publication of epidemiological data highlighting the importance of adolescent and adult boosters in disease control.
- 2023: Regulatory approvals for new Tdap vaccine formulations with extended shelf lives.
- 2024: Partnerships formed to address vaccine access challenges in low- and middle-income countries.
In-Depth Pertussis Booster Vaccine Market Outlook
The future outlook for the Pertussis Booster Vaccine market is exceptionally positive, driven by sustained demand for effective disease prevention and ongoing innovation. Growth accelerators, including technological breakthroughs in vaccine formulation and the strategic expansion into emerging markets, will continue to propel the industry forward. The increasing emphasis on lifelong immunization and the growing recognition of pertussis as a public health concern across all age demographics present significant opportunities for market expansion. Strategic partnerships and government-backed immunization programs will remain pivotal in ensuring broad vaccine access and sustained demand. The market is poised for steady and robust growth, underscoring its critical role in global public health.
Pertussis Booster Vaccine Segmentation
-
1. Application
- 1.1. 12 Months Below
- 1.2. 12 Months Above
-
2. Types
- 2.1. Acellular Vaccines
- 2.2. Whole-Cell Vaccines
Pertussis Booster Vaccine Segmentation By Geography
-
1. North America
- 1.1. United States
- 1.2. Canada
- 1.3. Mexico
-
2. South America
- 2.1. Brazil
- 2.2. Argentina
- 2.3. Rest of South America
-
3. Europe
- 3.1. United Kingdom
- 3.2. Germany
- 3.3. France
- 3.4. Italy
- 3.5. Spain
- 3.6. Russia
- 3.7. Benelux
- 3.8. Nordics
- 3.9. Rest of Europe
-
4. Middle East & Africa
- 4.1. Turkey
- 4.2. Israel
- 4.3. GCC
- 4.4. North Africa
- 4.5. South Africa
- 4.6. Rest of Middle East & Africa
-
5. Asia Pacific
- 5.1. China
- 5.2. India
- 5.3. Japan
- 5.4. South Korea
- 5.5. ASEAN
- 5.6. Oceania
- 5.7. Rest of Asia Pacific
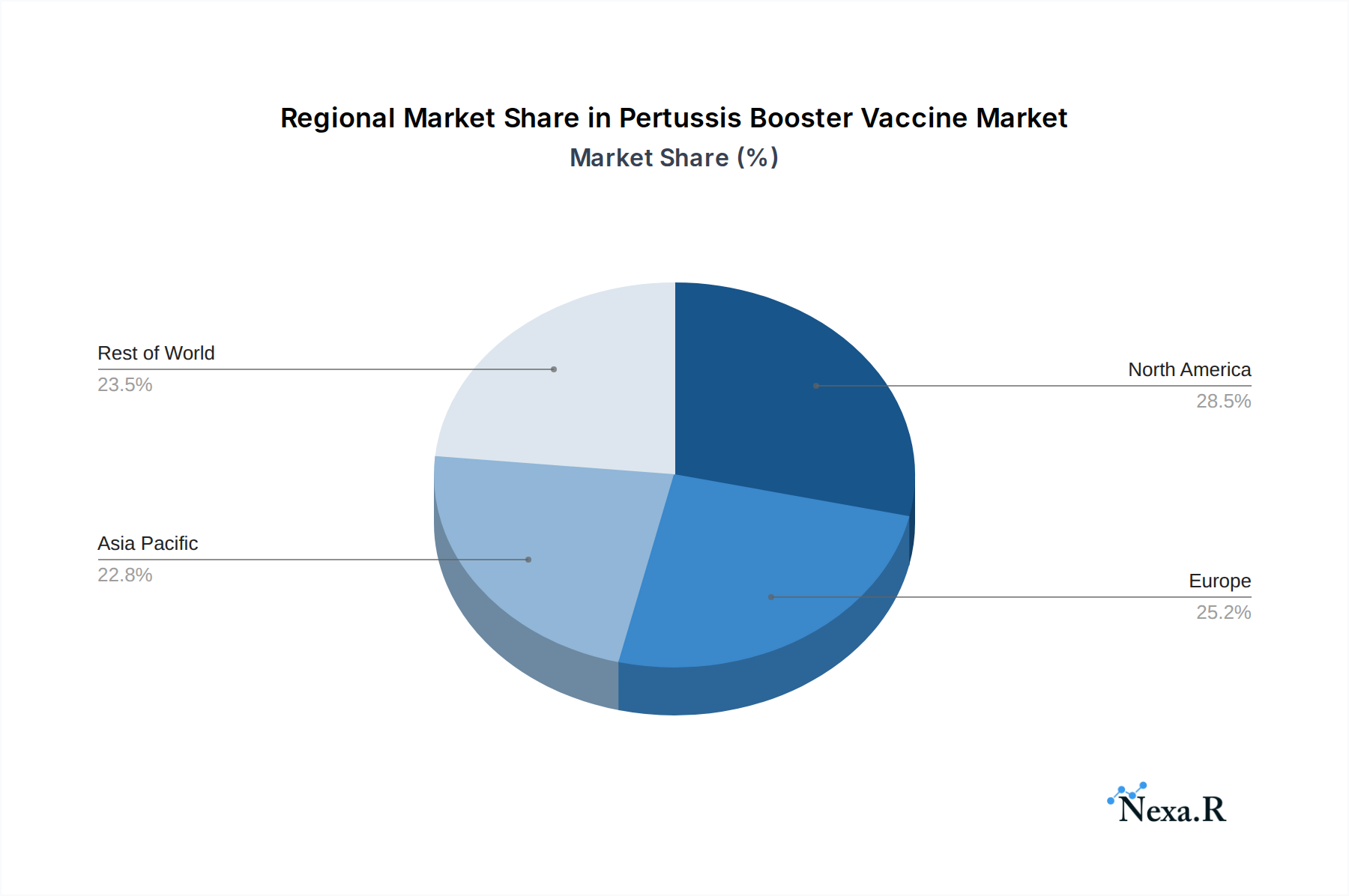
Pertussis Booster Vaccine Regional Market Share

Geographic Coverage of Pertussis Booster Vaccine
Pertussis Booster Vaccine REPORT HIGHLIGHTS
| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 5% from 2020-2034 |
| Segmentation |
|
Table of Contents
- 1. Introduction
- 1.1. Research Scope
- 1.2. Market Segmentation
- 1.3. Research Methodology
- 1.4. Definitions and Assumptions
- 2. Executive Summary
- 2.1. Introduction
- 3. Market Dynamics
- 3.1. Introduction
- 3.2. Market Drivers
- 3.3. Market Restrains
- 3.4. Market Trends
- 4. Market Factor Analysis
- 4.1. Porters Five Forces
- 4.2. Supply/Value Chain
- 4.3. PESTEL analysis
- 4.4. Market Entropy
- 4.5. Patent/Trademark Analysis
- 5. Global Pertussis Booster Vaccine Analysis, Insights and Forecast, 2020-2032
- 5.1. Market Analysis, Insights and Forecast - by Application
- 5.1.1. 12 Months Below
- 5.1.2. 12 Months Above
- 5.2. Market Analysis, Insights and Forecast - by Types
- 5.2.1. Acellular Vaccines
- 5.2.2. Whole-Cell Vaccines
- 5.3. Market Analysis, Insights and Forecast - by Region
- 5.3.1. North America
- 5.3.2. South America
- 5.3.3. Europe
- 5.3.4. Middle East & Africa
- 5.3.5. Asia Pacific
- 5.1. Market Analysis, Insights and Forecast - by Application
- 6. North America Pertussis Booster Vaccine Analysis, Insights and Forecast, 2020-2032
- 6.1. Market Analysis, Insights and Forecast - by Application
- 6.1.1. 12 Months Below
- 6.1.2. 12 Months Above
- 6.2. Market Analysis, Insights and Forecast - by Types
- 6.2.1. Acellular Vaccines
- 6.2.2. Whole-Cell Vaccines
- 6.1. Market Analysis, Insights and Forecast - by Application
- 7. South America Pertussis Booster Vaccine Analysis, Insights and Forecast, 2020-2032
- 7.1. Market Analysis, Insights and Forecast - by Application
- 7.1.1. 12 Months Below
- 7.1.2. 12 Months Above
- 7.2. Market Analysis, Insights and Forecast - by Types
- 7.2.1. Acellular Vaccines
- 7.2.2. Whole-Cell Vaccines
- 7.1. Market Analysis, Insights and Forecast - by Application
- 8. Europe Pertussis Booster Vaccine Analysis, Insights and Forecast, 2020-2032
- 8.1. Market Analysis, Insights and Forecast - by Application
- 8.1.1. 12 Months Below
- 8.1.2. 12 Months Above
- 8.2. Market Analysis, Insights and Forecast - by Types
- 8.2.1. Acellular Vaccines
- 8.2.2. Whole-Cell Vaccines
- 8.1. Market Analysis, Insights and Forecast - by Application
- 9. Middle East & Africa Pertussis Booster Vaccine Analysis, Insights and Forecast, 2020-2032
- 9.1. Market Analysis, Insights and Forecast - by Application
- 9.1.1. 12 Months Below
- 9.1.2. 12 Months Above
- 9.2. Market Analysis, Insights and Forecast - by Types
- 9.2.1. Acellular Vaccines
- 9.2.2. Whole-Cell Vaccines
- 9.1. Market Analysis, Insights and Forecast - by Application
- 10. Asia Pacific Pertussis Booster Vaccine Analysis, Insights and Forecast, 2020-2032
- 10.1. Market Analysis, Insights and Forecast - by Application
- 10.1.1. 12 Months Below
- 10.1.2. 12 Months Above
- 10.2. Market Analysis, Insights and Forecast - by Types
- 10.2.1. Acellular Vaccines
- 10.2.2. Whole-Cell Vaccines
- 10.1. Market Analysis, Insights and Forecast - by Application
- 11. Competitive Analysis
- 11.1. Global Market Share Analysis 2025
- 11.2. Company Profiles
- 11.2.1 Sanofi Pasteur
- 11.2.1.1. Overview
- 11.2.1.2. Products
- 11.2.1.3. SWOT Analysis
- 11.2.1.4. Recent Developments
- 11.2.1.5. Financials (Based on Availability)
- 11.2.2 GSK
- 11.2.2.1. Overview
- 11.2.2.2. Products
- 11.2.2.3. SWOT Analysis
- 11.2.2.4. Recent Developments
- 11.2.2.5. Financials (Based on Availability)
- 11.2.3 Mitsubishi Tanabe Pharma
- 11.2.3.1. Overview
- 11.2.3.2. Products
- 11.2.3.3. SWOT Analysis
- 11.2.3.4. Recent Developments
- 11.2.3.5. Financials (Based on Availability)
- 11.2.4 KM Biologics
- 11.2.4.1. Overview
- 11.2.4.2. Products
- 11.2.4.3. SWOT Analysis
- 11.2.4.4. Recent Developments
- 11.2.4.5. Financials (Based on Availability)
- 11.2.5 Wuhan Institute of Biological Products
- 11.2.5.1. Overview
- 11.2.5.2. Products
- 11.2.5.3. SWOT Analysis
- 11.2.5.4. Recent Developments
- 11.2.5.5. Financials (Based on Availability)
- 11.2.6 Walvax Biotechnology
- 11.2.6.1. Overview
- 11.2.6.2. Products
- 11.2.6.3. SWOT Analysis
- 11.2.6.4. Recent Developments
- 11.2.6.5. Financials (Based on Availability)
- 11.2.7 Chengdu Institute of Biological Products
- 11.2.7.1. Overview
- 11.2.7.2. Products
- 11.2.7.3. SWOT Analysis
- 11.2.7.4. Recent Developments
- 11.2.7.5. Financials (Based on Availability)
- 11.2.8 Minhai Biotechnology
- 11.2.8.1. Overview
- 11.2.8.2. Products
- 11.2.8.3. SWOT Analysis
- 11.2.8.4. Recent Developments
- 11.2.8.5. Financials (Based on Availability)
- 11.2.1 Sanofi Pasteur
List of Figures
- Figure 1: Global Pertussis Booster Vaccine Revenue Breakdown (undefined, %) by Region 2025 & 2033
- Figure 2: Global Pertussis Booster Vaccine Volume Breakdown (K, %) by Region 2025 & 2033
- Figure 3: North America Pertussis Booster Vaccine Revenue (undefined), by Application 2025 & 2033
- Figure 4: North America Pertussis Booster Vaccine Volume (K), by Application 2025 & 2033
- Figure 5: North America Pertussis Booster Vaccine Revenue Share (%), by Application 2025 & 2033
- Figure 6: North America Pertussis Booster Vaccine Volume Share (%), by Application 2025 & 2033
- Figure 7: North America Pertussis Booster Vaccine Revenue (undefined), by Types 2025 & 2033
- Figure 8: North America Pertussis Booster Vaccine Volume (K), by Types 2025 & 2033
- Figure 9: North America Pertussis Booster Vaccine Revenue Share (%), by Types 2025 & 2033
- Figure 10: North America Pertussis Booster Vaccine Volume Share (%), by Types 2025 & 2033
- Figure 11: North America Pertussis Booster Vaccine Revenue (undefined), by Country 2025 & 2033
- Figure 12: North America Pertussis Booster Vaccine Volume (K), by Country 2025 & 2033
- Figure 13: North America Pertussis Booster Vaccine Revenue Share (%), by Country 2025 & 2033
- Figure 14: North America Pertussis Booster Vaccine Volume Share (%), by Country 2025 & 2033
- Figure 15: South America Pertussis Booster Vaccine Revenue (undefined), by Application 2025 & 2033
- Figure 16: South America Pertussis Booster Vaccine Volume (K), by Application 2025 & 2033
- Figure 17: South America Pertussis Booster Vaccine Revenue Share (%), by Application 2025 & 2033
- Figure 18: South America Pertussis Booster Vaccine Volume Share (%), by Application 2025 & 2033
- Figure 19: South America Pertussis Booster Vaccine Revenue (undefined), by Types 2025 & 2033
- Figure 20: South America Pertussis Booster Vaccine Volume (K), by Types 2025 & 2033
- Figure 21: South America Pertussis Booster Vaccine Revenue Share (%), by Types 2025 & 2033
- Figure 22: South America Pertussis Booster Vaccine Volume Share (%), by Types 2025 & 2033
- Figure 23: South America Pertussis Booster Vaccine Revenue (undefined), by Country 2025 & 2033
- Figure 24: South America Pertussis Booster Vaccine Volume (K), by Country 2025 & 2033
- Figure 25: South America Pertussis Booster Vaccine Revenue Share (%), by Country 2025 & 2033
- Figure 26: South America Pertussis Booster Vaccine Volume Share (%), by Country 2025 & 2033
- Figure 27: Europe Pertussis Booster Vaccine Revenue (undefined), by Application 2025 & 2033
- Figure 28: Europe Pertussis Booster Vaccine Volume (K), by Application 2025 & 2033
- Figure 29: Europe Pertussis Booster Vaccine Revenue Share (%), by Application 2025 & 2033
- Figure 30: Europe Pertussis Booster Vaccine Volume Share (%), by Application 2025 & 2033
- Figure 31: Europe Pertussis Booster Vaccine Revenue (undefined), by Types 2025 & 2033
- Figure 32: Europe Pertussis Booster Vaccine Volume (K), by Types 2025 & 2033
- Figure 33: Europe Pertussis Booster Vaccine Revenue Share (%), by Types 2025 & 2033
- Figure 34: Europe Pertussis Booster Vaccine Volume Share (%), by Types 2025 & 2033
- Figure 35: Europe Pertussis Booster Vaccine Revenue (undefined), by Country 2025 & 2033
- Figure 36: Europe Pertussis Booster Vaccine Volume (K), by Country 2025 & 2033
- Figure 37: Europe Pertussis Booster Vaccine Revenue Share (%), by Country 2025 & 2033
- Figure 38: Europe Pertussis Booster Vaccine Volume Share (%), by Country 2025 & 2033
- Figure 39: Middle East & Africa Pertussis Booster Vaccine Revenue (undefined), by Application 2025 & 2033
- Figure 40: Middle East & Africa Pertussis Booster Vaccine Volume (K), by Application 2025 & 2033
- Figure 41: Middle East & Africa Pertussis Booster Vaccine Revenue Share (%), by Application 2025 & 2033
- Figure 42: Middle East & Africa Pertussis Booster Vaccine Volume Share (%), by Application 2025 & 2033
- Figure 43: Middle East & Africa Pertussis Booster Vaccine Revenue (undefined), by Types 2025 & 2033
- Figure 44: Middle East & Africa Pertussis Booster Vaccine Volume (K), by Types 2025 & 2033
- Figure 45: Middle East & Africa Pertussis Booster Vaccine Revenue Share (%), by Types 2025 & 2033
- Figure 46: Middle East & Africa Pertussis Booster Vaccine Volume Share (%), by Types 2025 & 2033
- Figure 47: Middle East & Africa Pertussis Booster Vaccine Revenue (undefined), by Country 2025 & 2033
- Figure 48: Middle East & Africa Pertussis Booster Vaccine Volume (K), by Country 2025 & 2033
- Figure 49: Middle East & Africa Pertussis Booster Vaccine Revenue Share (%), by Country 2025 & 2033
- Figure 50: Middle East & Africa Pertussis Booster Vaccine Volume Share (%), by Country 2025 & 2033
- Figure 51: Asia Pacific Pertussis Booster Vaccine Revenue (undefined), by Application 2025 & 2033
- Figure 52: Asia Pacific Pertussis Booster Vaccine Volume (K), by Application 2025 & 2033
- Figure 53: Asia Pacific Pertussis Booster Vaccine Revenue Share (%), by Application 2025 & 2033
- Figure 54: Asia Pacific Pertussis Booster Vaccine Volume Share (%), by Application 2025 & 2033
- Figure 55: Asia Pacific Pertussis Booster Vaccine Revenue (undefined), by Types 2025 & 2033
- Figure 56: Asia Pacific Pertussis Booster Vaccine Volume (K), by Types 2025 & 2033
- Figure 57: Asia Pacific Pertussis Booster Vaccine Revenue Share (%), by Types 2025 & 2033
- Figure 58: Asia Pacific Pertussis Booster Vaccine Volume Share (%), by Types 2025 & 2033
- Figure 59: Asia Pacific Pertussis Booster Vaccine Revenue (undefined), by Country 2025 & 2033
- Figure 60: Asia Pacific Pertussis Booster Vaccine Volume (K), by Country 2025 & 2033
- Figure 61: Asia Pacific Pertussis Booster Vaccine Revenue Share (%), by Country 2025 & 2033
- Figure 62: Asia Pacific Pertussis Booster Vaccine Volume Share (%), by Country 2025 & 2033
List of Tables
- Table 1: Global Pertussis Booster Vaccine Revenue undefined Forecast, by Application 2020 & 2033
- Table 2: Global Pertussis Booster Vaccine Volume K Forecast, by Application 2020 & 2033
- Table 3: Global Pertussis Booster Vaccine Revenue undefined Forecast, by Types 2020 & 2033
- Table 4: Global Pertussis Booster Vaccine Volume K Forecast, by Types 2020 & 2033
- Table 5: Global Pertussis Booster Vaccine Revenue undefined Forecast, by Region 2020 & 2033
- Table 6: Global Pertussis Booster Vaccine Volume K Forecast, by Region 2020 & 2033
- Table 7: Global Pertussis Booster Vaccine Revenue undefined Forecast, by Application 2020 & 2033
- Table 8: Global Pertussis Booster Vaccine Volume K Forecast, by Application 2020 & 2033
- Table 9: Global Pertussis Booster Vaccine Revenue undefined Forecast, by Types 2020 & 2033
- Table 10: Global Pertussis Booster Vaccine Volume K Forecast, by Types 2020 & 2033
- Table 11: Global Pertussis Booster Vaccine Revenue undefined Forecast, by Country 2020 & 2033
- Table 12: Global Pertussis Booster Vaccine Volume K Forecast, by Country 2020 & 2033
- Table 13: United States Pertussis Booster Vaccine Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 14: United States Pertussis Booster Vaccine Volume (K) Forecast, by Application 2020 & 2033
- Table 15: Canada Pertussis Booster Vaccine Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 16: Canada Pertussis Booster Vaccine Volume (K) Forecast, by Application 2020 & 2033
- Table 17: Mexico Pertussis Booster Vaccine Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 18: Mexico Pertussis Booster Vaccine Volume (K) Forecast, by Application 2020 & 2033
- Table 19: Global Pertussis Booster Vaccine Revenue undefined Forecast, by Application 2020 & 2033
- Table 20: Global Pertussis Booster Vaccine Volume K Forecast, by Application 2020 & 2033
- Table 21: Global Pertussis Booster Vaccine Revenue undefined Forecast, by Types 2020 & 2033
- Table 22: Global Pertussis Booster Vaccine Volume K Forecast, by Types 2020 & 2033
- Table 23: Global Pertussis Booster Vaccine Revenue undefined Forecast, by Country 2020 & 2033
- Table 24: Global Pertussis Booster Vaccine Volume K Forecast, by Country 2020 & 2033
- Table 25: Brazil Pertussis Booster Vaccine Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 26: Brazil Pertussis Booster Vaccine Volume (K) Forecast, by Application 2020 & 2033
- Table 27: Argentina Pertussis Booster Vaccine Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 28: Argentina Pertussis Booster Vaccine Volume (K) Forecast, by Application 2020 & 2033
- Table 29: Rest of South America Pertussis Booster Vaccine Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 30: Rest of South America Pertussis Booster Vaccine Volume (K) Forecast, by Application 2020 & 2033
- Table 31: Global Pertussis Booster Vaccine Revenue undefined Forecast, by Application 2020 & 2033
- Table 32: Global Pertussis Booster Vaccine Volume K Forecast, by Application 2020 & 2033
- Table 33: Global Pertussis Booster Vaccine Revenue undefined Forecast, by Types 2020 & 2033
- Table 34: Global Pertussis Booster Vaccine Volume K Forecast, by Types 2020 & 2033
- Table 35: Global Pertussis Booster Vaccine Revenue undefined Forecast, by Country 2020 & 2033
- Table 36: Global Pertussis Booster Vaccine Volume K Forecast, by Country 2020 & 2033
- Table 37: United Kingdom Pertussis Booster Vaccine Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 38: United Kingdom Pertussis Booster Vaccine Volume (K) Forecast, by Application 2020 & 2033
- Table 39: Germany Pertussis Booster Vaccine Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 40: Germany Pertussis Booster Vaccine Volume (K) Forecast, by Application 2020 & 2033
- Table 41: France Pertussis Booster Vaccine Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 42: France Pertussis Booster Vaccine Volume (K) Forecast, by Application 2020 & 2033
- Table 43: Italy Pertussis Booster Vaccine Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 44: Italy Pertussis Booster Vaccine Volume (K) Forecast, by Application 2020 & 2033
- Table 45: Spain Pertussis Booster Vaccine Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 46: Spain Pertussis Booster Vaccine Volume (K) Forecast, by Application 2020 & 2033
- Table 47: Russia Pertussis Booster Vaccine Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 48: Russia Pertussis Booster Vaccine Volume (K) Forecast, by Application 2020 & 2033
- Table 49: Benelux Pertussis Booster Vaccine Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 50: Benelux Pertussis Booster Vaccine Volume (K) Forecast, by Application 2020 & 2033
- Table 51: Nordics Pertussis Booster Vaccine Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 52: Nordics Pertussis Booster Vaccine Volume (K) Forecast, by Application 2020 & 2033
- Table 53: Rest of Europe Pertussis Booster Vaccine Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 54: Rest of Europe Pertussis Booster Vaccine Volume (K) Forecast, by Application 2020 & 2033
- Table 55: Global Pertussis Booster Vaccine Revenue undefined Forecast, by Application 2020 & 2033
- Table 56: Global Pertussis Booster Vaccine Volume K Forecast, by Application 2020 & 2033
- Table 57: Global Pertussis Booster Vaccine Revenue undefined Forecast, by Types 2020 & 2033
- Table 58: Global Pertussis Booster Vaccine Volume K Forecast, by Types 2020 & 2033
- Table 59: Global Pertussis Booster Vaccine Revenue undefined Forecast, by Country 2020 & 2033
- Table 60: Global Pertussis Booster Vaccine Volume K Forecast, by Country 2020 & 2033
- Table 61: Turkey Pertussis Booster Vaccine Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 62: Turkey Pertussis Booster Vaccine Volume (K) Forecast, by Application 2020 & 2033
- Table 63: Israel Pertussis Booster Vaccine Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 64: Israel Pertussis Booster Vaccine Volume (K) Forecast, by Application 2020 & 2033
- Table 65: GCC Pertussis Booster Vaccine Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 66: GCC Pertussis Booster Vaccine Volume (K) Forecast, by Application 2020 & 2033
- Table 67: North Africa Pertussis Booster Vaccine Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 68: North Africa Pertussis Booster Vaccine Volume (K) Forecast, by Application 2020 & 2033
- Table 69: South Africa Pertussis Booster Vaccine Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 70: South Africa Pertussis Booster Vaccine Volume (K) Forecast, by Application 2020 & 2033
- Table 71: Rest of Middle East & Africa Pertussis Booster Vaccine Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 72: Rest of Middle East & Africa Pertussis Booster Vaccine Volume (K) Forecast, by Application 2020 & 2033
- Table 73: Global Pertussis Booster Vaccine Revenue undefined Forecast, by Application 2020 & 2033
- Table 74: Global Pertussis Booster Vaccine Volume K Forecast, by Application 2020 & 2033
- Table 75: Global Pertussis Booster Vaccine Revenue undefined Forecast, by Types 2020 & 2033
- Table 76: Global Pertussis Booster Vaccine Volume K Forecast, by Types 2020 & 2033
- Table 77: Global Pertussis Booster Vaccine Revenue undefined Forecast, by Country 2020 & 2033
- Table 78: Global Pertussis Booster Vaccine Volume K Forecast, by Country 2020 & 2033
- Table 79: China Pertussis Booster Vaccine Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 80: China Pertussis Booster Vaccine Volume (K) Forecast, by Application 2020 & 2033
- Table 81: India Pertussis Booster Vaccine Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 82: India Pertussis Booster Vaccine Volume (K) Forecast, by Application 2020 & 2033
- Table 83: Japan Pertussis Booster Vaccine Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 84: Japan Pertussis Booster Vaccine Volume (K) Forecast, by Application 2020 & 2033
- Table 85: South Korea Pertussis Booster Vaccine Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 86: South Korea Pertussis Booster Vaccine Volume (K) Forecast, by Application 2020 & 2033
- Table 87: ASEAN Pertussis Booster Vaccine Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 88: ASEAN Pertussis Booster Vaccine Volume (K) Forecast, by Application 2020 & 2033
- Table 89: Oceania Pertussis Booster Vaccine Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 90: Oceania Pertussis Booster Vaccine Volume (K) Forecast, by Application 2020 & 2033
- Table 91: Rest of Asia Pacific Pertussis Booster Vaccine Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 92: Rest of Asia Pacific Pertussis Booster Vaccine Volume (K) Forecast, by Application 2020 & 2033
Frequently Asked Questions
1. What is the projected Compound Annual Growth Rate (CAGR) of the Pertussis Booster Vaccine?
The projected CAGR is approximately 5%.
2. Which companies are prominent players in the Pertussis Booster Vaccine?
Key companies in the market include Sanofi Pasteur, GSK, Mitsubishi Tanabe Pharma, KM Biologics, Wuhan Institute of Biological Products, Walvax Biotechnology, Chengdu Institute of Biological Products, Minhai Biotechnology.
3. What are the main segments of the Pertussis Booster Vaccine?
The market segments include Application, Types.
4. Can you provide details about the market size?
The market size is estimated to be USD XXX N/A as of 2022.
5. What are some drivers contributing to market growth?
N/A
6. What are the notable trends driving market growth?
N/A
7. Are there any restraints impacting market growth?
N/A
8. Can you provide examples of recent developments in the market?
N/A
9. What pricing options are available for accessing the report?
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 4350.00, USD 6525.00, and USD 8700.00 respectively.
10. Is the market size provided in terms of value or volume?
The market size is provided in terms of value, measured in N/A and volume, measured in K.
11. Are there any specific market keywords associated with the report?
Yes, the market keyword associated with the report is "Pertussis Booster Vaccine," which aids in identifying and referencing the specific market segment covered.
12. How do I determine which pricing option suits my needs best?
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
13. Are there any additional resources or data provided in the Pertussis Booster Vaccine report?
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
14. How can I stay updated on further developments or reports in the Pertussis Booster Vaccine?
To stay informed about further developments, trends, and reports in the Pertussis Booster Vaccine, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.
Methodology
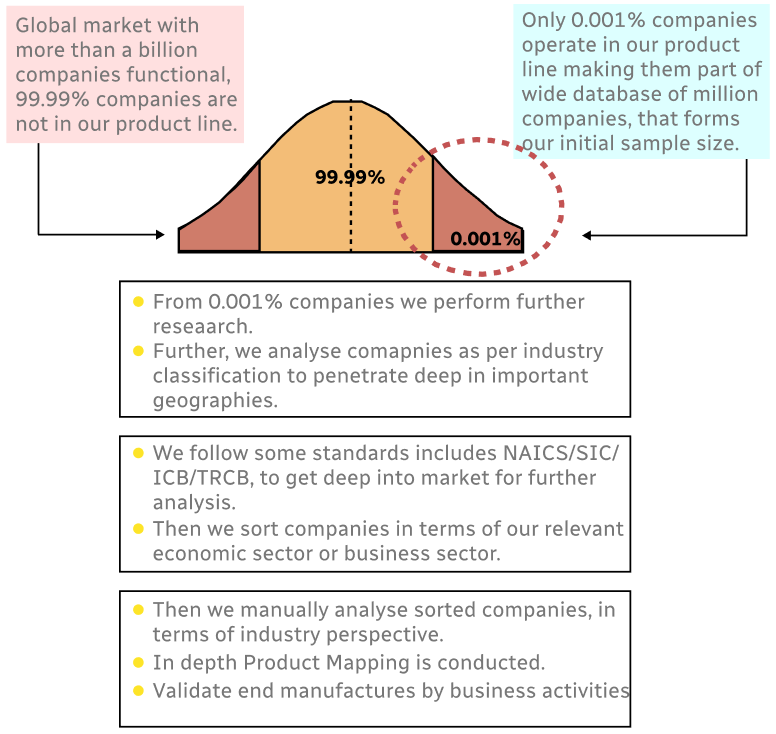
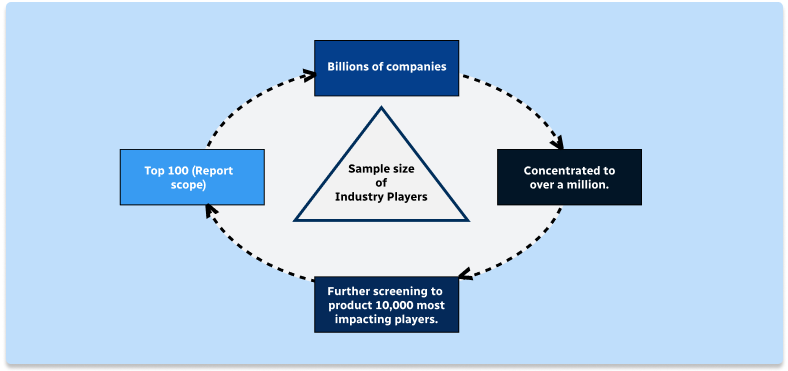
Step 1 - Identification of Relevant Samples Size from Population Database
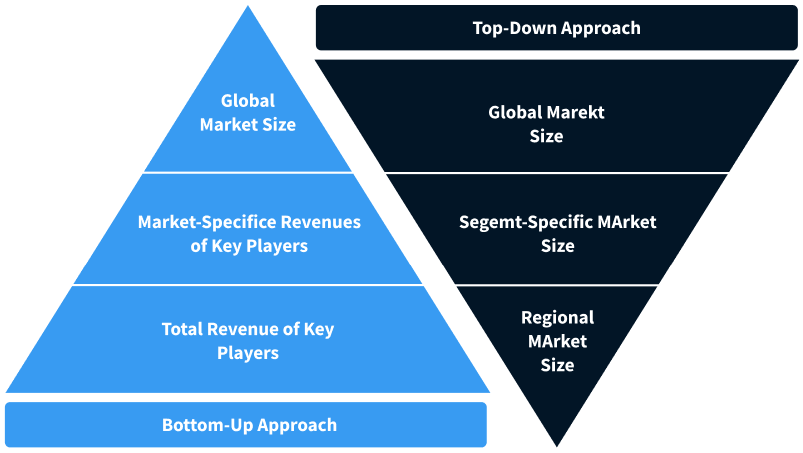
Step 2 - Approaches for Defining Global Market Size (Value, Volume* & Price*)
Note*: In applicable scenarios
Step 3 - Data Sources
Primary Research
- Web Analytics
- Survey Reports
- Research Institute
- Latest Research Reports
- Opinion Leaders
Secondary Research
- Annual Reports
- White Paper
- Latest Press Release
- Industry Association
- Paid Database
- Investor Presentations
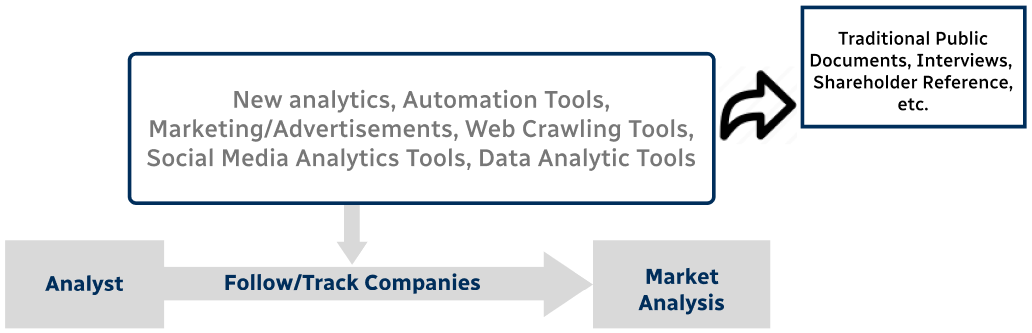
Step 4 - Data Triangulation
Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence


