Key Insights
The global Mycoplasma Testing market is poised for significant expansion, projected to reach $972.3 million in 2025 with a robust Compound Annual Growth Rate (CAGR) of 5.3% through 2033. This growth is propelled by an escalating demand for accurate and reliable mycoplasma detection across various sectors, most notably in biopharmaceutical production and cell line development. The increasing stringency of regulatory guidelines regarding mycoplasma contamination in biologics and cell-based therapies is a primary driver, compelling manufacturers and researchers to adopt advanced testing methodologies. Furthermore, the burgeoning field of cell and gene therapy, which relies heavily on uncontaminated cell lines, is creating a substantial demand for sophisticated mycoplasma testing solutions. The market is characterized by a strong emphasis on technological advancements, with Polymerase Chain Reaction (PCR) and Enzyme-Linked Immunosorbent Assay (ELISA) technologies dominating the landscape due to their sensitivity and speed. The broad application in bioproduction testing, where ensuring the integrity and safety of therapeutic products is paramount, further underscores the market's upward trajectory.
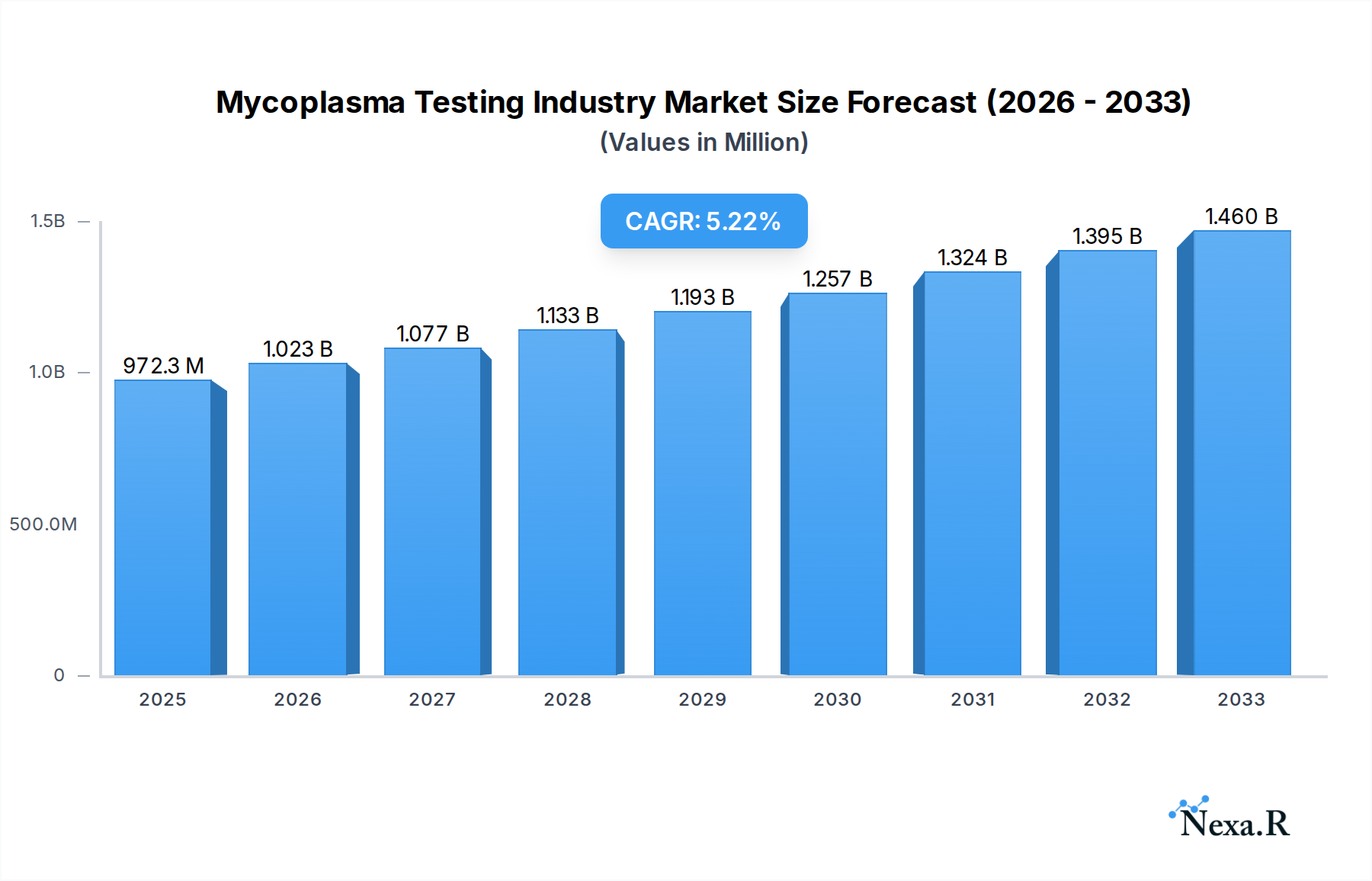
Mycoplasma Testing Industry Market Size (In Million)

The market's expansion is also influenced by a growing awareness of the detrimental effects of mycoplasma contamination on cell culture viability, experimental outcomes, and the overall safety and efficacy of pharmaceutical products. While the market enjoys strong growth drivers, certain restraints, such as the high cost of advanced testing equipment and the need for specialized expertise, may temper its pace in specific segments. However, the continuous innovation in diagnostic tools and the increasing availability of cost-effective solutions are expected to mitigate these challenges. Geographically, North America and Europe are expected to lead the market due to well-established biopharmaceutical industries and stringent regulatory frameworks. The Asia Pacific region, with its rapidly growing biotech sector and increasing investments in life sciences research, presents a significant growth opportunity. Key players like Merck KGaA, Thermo Fisher Scientific, and Eurofins Scientific are actively investing in research and development to introduce novel testing solutions and expand their market presence.
Mycoplasma Testing Industry Company Market Share

Unlocking the Mycoplasma Testing Industry: A Comprehensive Market Analysis (2019-2033)
This in-depth report offers a critical analysis of the global Mycoplasma Testing Industry, providing actionable insights for stakeholders navigating this dynamic and expanding market. We delve into parent and child market segments, offering a granular view of market structure, growth trajectories, and competitive landscapes. With a study period spanning from 2019 to 2033 and a detailed forecast for 2025-2033, this report equips you with the data and strategic foresight necessary to capitalize on emerging opportunities and mitigate potential challenges.
Mycoplasma Testing Industry Market Dynamics & Structure
The Mycoplasma Testing Industry is characterized by a moderately concentrated market structure, driven by significant technological innovation and stringent regulatory frameworks. Key players are investing heavily in research and development to enhance sensitivity, specificity, and speed of mycoplasma detection. The high cost of advanced instrumentation and proprietary kits acts as a barrier to entry for smaller companies, solidifying the dominance of established entities. End-user demographics are increasingly diverse, encompassing pharmaceutical manufacturers, biopharmaceutical companies, academic research institutions, and diagnostic laboratories. Technological innovation, particularly in PCR and Next-Generation Sequencing (NGS) based methods, is a primary growth driver, enabling more accurate and efficient detection. Regulatory compliance, with bodies like the FDA and EMA mandating mycoplasma-free products, further fuels market demand. Competitive product substitutes, while emerging, are yet to fully displace established methods due to validation requirements. Mergers and acquisitions (M&A) are a consistent trend, with larger companies acquiring innovative smaller firms to expand their product portfolios and market reach. In the historical period (2019-2024), we observed an average of 6-8 significant M&A deals annually, often focused on acquiring novel diagnostic technologies. The market is also influenced by the growing complexity of biologics and cell-based therapies, which necessitate highly sensitive detection methods.
- Market Concentration: Moderately concentrated with key players holding substantial market share.
- Technological Innovation Drivers: Advancement in PCR, NGS, and automation technologies.
- Regulatory Frameworks: Strict guidelines from FDA, EMA, and other global health authorities.
- Competitive Product Substitutes: Emerging technologies are gradually gaining traction but require extensive validation.
- End-User Demographics: Pharmaceutical, biopharmaceutical, academic research, and clinical diagnostics.
- M&A Trends: Consistent activity focused on technology acquisition and market expansion.
Mycoplasma Testing Industry Growth Trends & Insights
The Mycoplasma Testing Industry is poised for robust growth, driven by a confluence of factors including the escalating demand for biologics and cell therapies, increasing stringency of regulatory requirements for mycoplasma contamination control, and continuous advancements in diagnostic technologies. The global mycoplasma testing market size is projected to witness a substantial Compound Annual Growth Rate (CAGR) of approximately 9.5% during the forecast period of 2025–2033. In the base year 2025, the market is estimated to be valued at $3,200 million. This expansion is largely attributed to the increasing adoption of advanced testing methodologies such as Polymerase Chain Reaction (PCR) and enzyme-linked immunosorbent assays (ELISA), which offer superior sensitivity and specificity compared to traditional methods. The adoption rate of these advanced technologies is expected to accelerate as their cost-effectiveness and efficiency become more widely recognized. Furthermore, the growing awareness among end-users regarding the detrimental impact of mycoplasma contamination on cell culture viability, product quality, and experimental outcomes is a significant catalyst. Consumer behavior shifts are evident, with a growing preference for rapid, high-throughput, and automation-compatible testing solutions, particularly within the biopharmaceutical sector. The increasing prevalence of cell-based research and the expanding pipeline of gene and cell therapies underscore the critical need for reliable mycoplasma detection. The industry is also witnessing a trend towards multiplex testing, allowing for the simultaneous detection of multiple contaminants, thereby improving workflow efficiency and reducing turnaround times. The market penetration of routine mycoplasma testing in emerging economies is also a key growth driver, as regulatory standards align with global benchmarks. The total market is projected to reach an estimated $6,800 million by 2033.
Dominant Regions, Countries, or Segments in Mycoplasma Testing Industry
The North America region currently dominates the Mycoplasma Testing Industry, driven by a robust biopharmaceutical sector, significant investments in life sciences research, and stringent regulatory oversight from the U.S. Food and Drug Administration (FDA). The presence of leading pharmaceutical and biotechnology companies, coupled with a well-established research infrastructure, creates a consistent and high demand for mycoplasma testing solutions. The United States specifically accounts for a significant share of this regional dominance, boasting a vast market for cell line testing and bioproduction testing. In the Product segment, Kits and Reagents are the leading sub-segment, representing an estimated 55% of the market share in 2025. This is due to their widespread use in routine testing across various applications and their relatively lower initial investment compared to instruments. Within the Technology segment, PCR (Polymerase Chain Reaction) is the dominant technology, estimated to hold 45% of the market share in 2025. Its unparalleled sensitivity, specificity, and speed make it the gold standard for mycoplasma detection in critical applications. The Application segment of Cell Line Testing is a major growth driver, representing approximately 40% of the market share in 2025. This is fueled by the burgeoning cell therapy and biologics market, where ensuring cell line integrity is paramount.
Key drivers for North America's dominance include:
- Economic Policies: Favorable government policies supporting biopharmaceutical research and development.
- Infrastructure: Advanced research laboratories and manufacturing facilities requiring rigorous quality control.
- Market Share: Estimated to hold 38% of the global Mycoplasma Testing Industry market share in 2025.
- Growth Potential: Continued innovation and expansion of cell and gene therapy pipelines ensure sustained demand.
- Regulatory Frameworks: Proactive enforcement of stringent mycoplasma testing regulations by the FDA.
Other regions like Europe also show significant growth, driven by a strong pharmaceutical industry and increasing regulatory emphasis on product safety. Asia-Pacific is an emerging market with high growth potential due to expanding biopharmaceutical manufacturing capabilities and increasing healthcare expenditure.
Mycoplasma Testing Industry Product Landscape
The Mycoplasma Testing Industry product landscape is characterized by continuous innovation aimed at enhancing detection accuracy, speed, and user-friendliness. Kits and reagents remain a core offering, with advancements focusing on lyophilized formulations for improved stability, multiplex panels for simultaneous detection of multiple contaminants, and optimized reagents for specific PCR-based assays. Instruments are increasingly sophisticated, incorporating automation and high-throughput capabilities to meet the demands of large-scale biopharmaceutical production. Innovations in DNA staining methods offer rapid, visual assessment for preliminary screening. The performance metrics of these products are constantly improving, with lower Limits of Detection (LODs) and reduced assay times being key selling points. For instance, next-generation PCR kits can now detect as few as 10 CFU/mL of mycoplasma in under two hours, a significant improvement over traditional culture methods.
Key Drivers, Barriers & Challenges in Mycoplasma Testing Industry
Key Drivers:
- Surge in Biologics and Cell Therapy Production: The exponential growth in biopharmaceutical manufacturing, particularly cell and gene therapies, necessitates stringent mycoplasma control.
- Increasing Regulatory Scrutiny: Global regulatory bodies are tightening guidelines for mycoplasma contamination, mandating more rigorous testing protocols.
- Technological Advancements: Innovations in PCR, NGS, and immunoassay technologies are leading to more sensitive, specific, and faster detection methods.
- Growing Awareness: Increased understanding of the detrimental impact of mycoplasma contamination on research outcomes and product safety.
Barriers & Challenges:
- High Cost of Advanced Technologies: Initial investment in sophisticated instrumentation and validated kits can be substantial, particularly for smaller research institutions.
- Complex Validation Requirements: Revalidation of testing methods after any significant change can be time-consuming and costly.
- Shortage of Skilled Personnel: A need for trained professionals to operate advanced equipment and interpret complex test results.
- Emerging Resistance Patterns: The potential for mycoplasma strains to develop resistance to certain detection methods.
- Supply Chain Disruptions: Vulnerability to disruptions in the supply of critical reagents and components, impacting production timelines. The global supply chain for specialized reagents experienced a 15-20% increase in lead times during 2020-2022 due to pandemic-related issues.
Emerging Opportunities in Mycoplasma Testing Industry
Emerging opportunities in the Mycoplasma Testing Industry lie in the development of point-of-care (POC) diagnostic devices for rapid, on-site mycoplasma detection, particularly for sexually transmitted infections. The expansion of veterinary diagnostics requiring mycoplasma testing for animal health is another burgeoning area. Furthermore, the integration of artificial intelligence (AI) and machine learning (ML) into data analysis of high-throughput screening platforms can enhance predictive capabilities and workflow efficiency. The growing demand for environmental monitoring for mycoplasma in cleanroom environments also presents a significant untapped market. The development of novel biomarkers for early mycoplasma infection detection, beyond direct culture or nucleic acid amplification, could revolutionize diagnostics.
Growth Accelerators in the Mycoplasma Testing Industry Industry
Several factors are accelerating the growth of the Mycoplasma Testing Industry. The expanding pipeline of novel therapeutics, including CAR-T cell therapies and other advanced biologics, directly translates to increased demand for robust mycoplasma testing. Strategic partnerships between diagnostic companies and biopharmaceutical manufacturers are streamlining the integration of testing solutions into production workflows. Furthermore, the increasing global focus on antimicrobial resistance (AMR) is indirectly boosting the importance of accurate diagnostics for all bacterial agents, including mycoplasma. Market expansion into emerging economies with developing biopharmaceutical industries and increasing healthcare investments is a significant growth accelerator. The continuous refinement of existing technologies, leading to more affordable and accessible testing options, also plays a crucial role in market penetration.
Key Players Shaping the Mycoplasma Testing Industry Market
- Merck KGaA
- Eurofins Scientific
- Charles River Laboratories International Inc
- Thermo Fisher Scientific Inc
- Bionique Testing Laboratories Inc
- PromoCell GmbH
- Lonza Group Ltd
- Sartorius AG
- ATCC
- AdvaCare
- Agilent Technologies
Notable Milestones in Mycoplasma Testing Industry Sector
- May 2022: Abbott received United States Food and Drug Administration clearance for its Alinity m STI Assay. The test simultaneously detects and differentiates four common sexually transmitted infections (STIs), including Mycoplasma genitalium (MG), broadening the application of mycoplasma detection in clinical diagnostics.
- April 2022: Performer Availability Screening Service, Inc. (PASS) entered into a partnership with Spankchain to launch a screening and treatment initiative for Mycoplasma genitalium infection in the Las Vegas performer community at no cost for up to 1000 performers, highlighting a targeted public health initiative.
In-Depth Mycoplasma Testing Industry Market Outlook
The Mycoplasma Testing Industry market outlook is exceptionally positive, driven by persistent demand from the biopharmaceutical sector and advancements in diagnostic technologies. The forecasted growth trajectory indicates a substantial increase in market valuation, fueled by an expanding array of cell and gene therapies requiring rigorous mycoplasma-free assurance. Strategic opportunities abound in the development of integrated testing solutions that combine rapid detection with downstream workflow automation. The increasing focus on personalized medicine and the growing prevalence of chronic diseases are expected to further amplify the need for accurate diagnostic tools, including those for mycoplasma detection in various clinical contexts. Continued investment in research and development for novel detection methodologies, coupled with the expanding regulatory landscape, will undoubtedly shape the future of this vital industry, ensuring a sustained and robust growth. The market is projected to continue its upward trend, driven by innovation and critical demand.
Mycoplasma Testing Industry Segmentation
-
1. Product
- 1.1. Instruments
- 1.2. Kits and Reagents
-
2. Technology
- 2.1. PCR
- 2.2. ELISA
- 2.3. Enzymatic Methods
- 2.4. DNA Staining
- 2.5. Other Technologies
-
3. Application
- 3.1. Cell Line Testing
- 3.2. Bioproduction Testing
- 3.3. Other Applications
Mycoplasma Testing Industry Segmentation By Geography
-
1. North America
- 1.1. United States
- 1.2. Canada
- 1.3. Mexico
-
2. Europe
- 2.1. Germany
- 2.2. United Kingdom
- 2.3. France
- 2.4. Italy
- 2.5. Spain
- 2.6. Rest of Europe
-
3. Asia Pacific
- 3.1. China
- 3.2. Japan
- 3.3. India
- 3.4. Australia
- 3.5. South Korea
- 3.6. Rest of Asia Pacific
-
4. Middle East and Africa
- 4.1. GCC
- 4.2. South Africa
- 4.3. Rest of Middle East and Africa
-
5. South America
- 5.1. Brazil
- 5.2. Argentina
- 5.3. Rest of South America
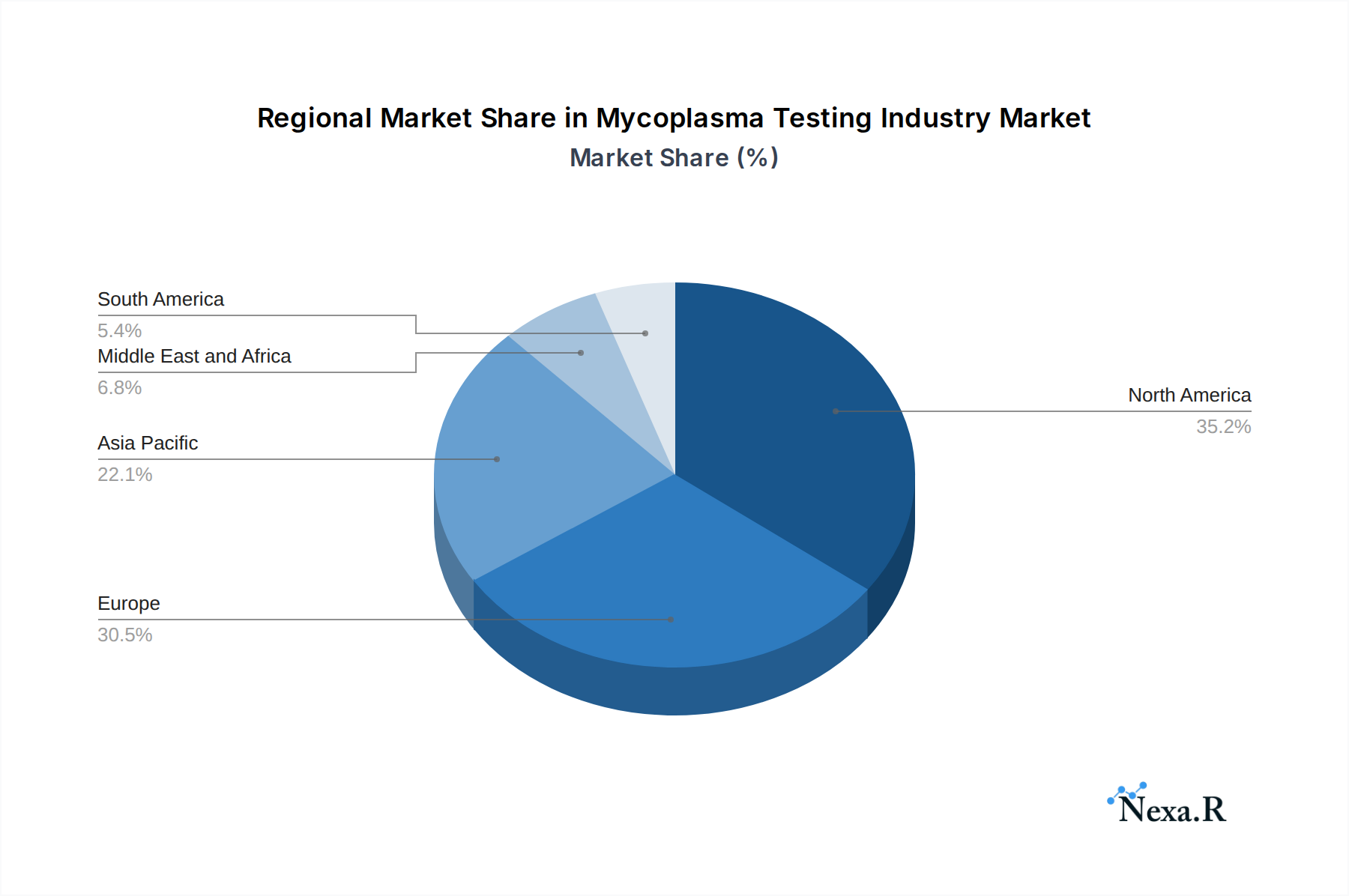
Mycoplasma Testing Industry Regional Market Share

Geographic Coverage of Mycoplasma Testing Industry
Mycoplasma Testing Industry REPORT HIGHLIGHTS
| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 5.3% from 2020-2034 |
| Segmentation |
|
Table of Contents
- 1. Introduction
- 1.1. Research Scope
- 1.2. Market Segmentation
- 1.3. Research Objective
- 1.4. Definitions and Assumptions
- 2. Executive Summary
- 2.1. Market Snapshot
- 3. Market Dynamics
- 3.1. Market Drivers
- 3.2. Market Restrains
- 3.3. Market Trends
- 3.4. Market Opportunities
- 4. Market Factor Analysis
- 4.1. Porters Five Forces
- 4.1.1. Bargaining Power of Suppliers
- 4.1.2. Bargaining Power of Buyers
- 4.1.3. Threat of New Entrants
- 4.1.4. Threat of Substitutes
- 4.1.5. Competitive Rivalry
- 4.2. PESTEL analysis
- 4.3. BCG Analysis
- 4.3.1. Stars (High Growth, High Market Share)
- 4.3.2. Cash Cows (Low Growth, High Market Share)
- 4.3.3. Question Mark (High Growth, Low Market Share)
- 4.3.4. Dogs (Low Growth, Low Market Share)
- 4.4. Ansoff Matrix Analysis
- 4.5. Supply Chain Analysis
- 4.6. Regulatory Landscape
- 4.7. Current Market Potential and Opportunity Assessment (TAM–SAM–SOM Framework)
- 4.8. NRP Analyst Note
- 4.1. Porters Five Forces
- 5. Market Analysis, Insights and Forecast 2021-2033
- 5.1. Market Analysis, Insights and Forecast - by Product
- 5.1.1. Instruments
- 5.1.2. Kits and Reagents
- 5.2. Market Analysis, Insights and Forecast - by Technology
- 5.2.1. PCR
- 5.2.2. ELISA
- 5.2.3. Enzymatic Methods
- 5.2.4. DNA Staining
- 5.2.5. Other Technologies
- 5.3. Market Analysis, Insights and Forecast - by Application
- 5.3.1. Cell Line Testing
- 5.3.2. Bioproduction Testing
- 5.3.3. Other Applications
- 5.4. Market Analysis, Insights and Forecast - by Region
- 5.4.1. North America
- 5.4.2. Europe
- 5.4.3. Asia Pacific
- 5.4.4. Middle East and Africa
- 5.4.5. South America
- 5.1. Market Analysis, Insights and Forecast - by Product
- 6. Global Mycoplasma Testing Industry Analysis, Insights and Forecast, 2021-2033
- 6.1. Market Analysis, Insights and Forecast - by Product
- 6.1.1. Instruments
- 6.1.2. Kits and Reagents
- 6.2. Market Analysis, Insights and Forecast - by Technology
- 6.2.1. PCR
- 6.2.2. ELISA
- 6.2.3. Enzymatic Methods
- 6.2.4. DNA Staining
- 6.2.5. Other Technologies
- 6.3. Market Analysis, Insights and Forecast - by Application
- 6.3.1. Cell Line Testing
- 6.3.2. Bioproduction Testing
- 6.3.3. Other Applications
- 6.1. Market Analysis, Insights and Forecast - by Product
- 7. North America Mycoplasma Testing Industry Analysis, Insights and Forecast, 2020-2032
- 7.1. Market Analysis, Insights and Forecast - by Product
- 7.1.1. Instruments
- 7.1.2. Kits and Reagents
- 7.2. Market Analysis, Insights and Forecast - by Technology
- 7.2.1. PCR
- 7.2.2. ELISA
- 7.2.3. Enzymatic Methods
- 7.2.4. DNA Staining
- 7.2.5. Other Technologies
- 7.3. Market Analysis, Insights and Forecast - by Application
- 7.3.1. Cell Line Testing
- 7.3.2. Bioproduction Testing
- 7.3.3. Other Applications
- 7.1. Market Analysis, Insights and Forecast - by Product
- 8. Europe Mycoplasma Testing Industry Analysis, Insights and Forecast, 2020-2032
- 8.1. Market Analysis, Insights and Forecast - by Product
- 8.1.1. Instruments
- 8.1.2. Kits and Reagents
- 8.2. Market Analysis, Insights and Forecast - by Technology
- 8.2.1. PCR
- 8.2.2. ELISA
- 8.2.3. Enzymatic Methods
- 8.2.4. DNA Staining
- 8.2.5. Other Technologies
- 8.3. Market Analysis, Insights and Forecast - by Application
- 8.3.1. Cell Line Testing
- 8.3.2. Bioproduction Testing
- 8.3.3. Other Applications
- 8.1. Market Analysis, Insights and Forecast - by Product
- 9. Asia Pacific Mycoplasma Testing Industry Analysis, Insights and Forecast, 2020-2032
- 9.1. Market Analysis, Insights and Forecast - by Product
- 9.1.1. Instruments
- 9.1.2. Kits and Reagents
- 9.2. Market Analysis, Insights and Forecast - by Technology
- 9.2.1. PCR
- 9.2.2. ELISA
- 9.2.3. Enzymatic Methods
- 9.2.4. DNA Staining
- 9.2.5. Other Technologies
- 9.3. Market Analysis, Insights and Forecast - by Application
- 9.3.1. Cell Line Testing
- 9.3.2. Bioproduction Testing
- 9.3.3. Other Applications
- 9.1. Market Analysis, Insights and Forecast - by Product
- 10. Middle East and Africa Mycoplasma Testing Industry Analysis, Insights and Forecast, 2020-2032
- 10.1. Market Analysis, Insights and Forecast - by Product
- 10.1.1. Instruments
- 10.1.2. Kits and Reagents
- 10.2. Market Analysis, Insights and Forecast - by Technology
- 10.2.1. PCR
- 10.2.2. ELISA
- 10.2.3. Enzymatic Methods
- 10.2.4. DNA Staining
- 10.2.5. Other Technologies
- 10.3. Market Analysis, Insights and Forecast - by Application
- 10.3.1. Cell Line Testing
- 10.3.2. Bioproduction Testing
- 10.3.3. Other Applications
- 10.1. Market Analysis, Insights and Forecast - by Product
- 11. South America Mycoplasma Testing Industry Analysis, Insights and Forecast, 2020-2032
- 11.1. Market Analysis, Insights and Forecast - by Product
- 11.1.1. Instruments
- 11.1.2. Kits and Reagents
- 11.2. Market Analysis, Insights and Forecast - by Technology
- 11.2.1. PCR
- 11.2.2. ELISA
- 11.2.3. Enzymatic Methods
- 11.2.4. DNA Staining
- 11.2.5. Other Technologies
- 11.3. Market Analysis, Insights and Forecast - by Application
- 11.3.1. Cell Line Testing
- 11.3.2. Bioproduction Testing
- 11.3.3. Other Applications
- 11.1. Market Analysis, Insights and Forecast - by Product
- 12. Competitive Analysis
- 12.1. Company Profiles
- 12.1.1 Merck KGaA
- 12.1.1.1. Company Overview
- 12.1.1.2. Products
- 12.1.1.3. Company Financials
- 12.1.1.4. SWOT Analysis
- 12.1.2 Eurofins Scientific
- 12.1.2.1. Company Overview
- 12.1.2.2. Products
- 12.1.2.3. Company Financials
- 12.1.2.4. SWOT Analysis
- 12.1.3 Charles River Laboratories International Inc
- 12.1.3.1. Company Overview
- 12.1.3.2. Products
- 12.1.3.3. Company Financials
- 12.1.3.4. SWOT Analysis
- 12.1.4 Thermo Fisher Scientific Inc
- 12.1.4.1. Company Overview
- 12.1.4.2. Products
- 12.1.4.3. Company Financials
- 12.1.4.4. SWOT Analysis
- 12.1.5 Bionique Testing Laboratories Inc
- 12.1.5.1. Company Overview
- 12.1.5.2. Products
- 12.1.5.3. Company Financials
- 12.1.5.4. SWOT Analysis
- 12.1.6 PromoCell GmbH
- 12.1.6.1. Company Overview
- 12.1.6.2. Products
- 12.1.6.3. Company Financials
- 12.1.6.4. SWOT Analysis
- 12.1.7 Lonza Group Ltd
- 12.1.7.1. Company Overview
- 12.1.7.2. Products
- 12.1.7.3. Company Financials
- 12.1.7.4. SWOT Analysis
- 12.1.8 Sartorius AG
- 12.1.8.1. Company Overview
- 12.1.8.2. Products
- 12.1.8.3. Company Financials
- 12.1.8.4. SWOT Analysis
- 12.1.9 ATCC
- 12.1.9.1. Company Overview
- 12.1.9.2. Products
- 12.1.9.3. Company Financials
- 12.1.9.4. SWOT Analysis
- 12.1.10 AdvaCare
- 12.1.10.1. Company Overview
- 12.1.10.2. Products
- 12.1.10.3. Company Financials
- 12.1.10.4. SWOT Analysis
- 12.1.11 Agilent Technologies
- 12.1.11.1. Company Overview
- 12.1.11.2. Products
- 12.1.11.3. Company Financials
- 12.1.11.4. SWOT Analysis
- 12.1.1 Merck KGaA
- 12.2. Market Entropy
- 12.2.1 Company's Key Areas Served
- 12.2.2 Recent Developments
- 12.3. Company Market Share Analysis 2025
- 12.3.1 Top 5 Companies Market Share Analysis
- 12.3.2 Top 3 Companies Market Share Analysis
- 12.4. List of Potential Customers
- 13. Research Methodology
List of Figures
- Figure 1: Global Mycoplasma Testing Industry Revenue Breakdown (million, %) by Region 2025 & 2033
- Figure 2: Global Mycoplasma Testing Industry Volume Breakdown (K Unit, %) by Region 2025 & 2033
- Figure 3: North America Mycoplasma Testing Industry Revenue (million), by Product 2025 & 2033
- Figure 4: North America Mycoplasma Testing Industry Volume (K Unit), by Product 2025 & 2033
- Figure 5: North America Mycoplasma Testing Industry Revenue Share (%), by Product 2025 & 2033
- Figure 6: North America Mycoplasma Testing Industry Volume Share (%), by Product 2025 & 2033
- Figure 7: North America Mycoplasma Testing Industry Revenue (million), by Technology 2025 & 2033
- Figure 8: North America Mycoplasma Testing Industry Volume (K Unit), by Technology 2025 & 2033
- Figure 9: North America Mycoplasma Testing Industry Revenue Share (%), by Technology 2025 & 2033
- Figure 10: North America Mycoplasma Testing Industry Volume Share (%), by Technology 2025 & 2033
- Figure 11: North America Mycoplasma Testing Industry Revenue (million), by Application 2025 & 2033
- Figure 12: North America Mycoplasma Testing Industry Volume (K Unit), by Application 2025 & 2033
- Figure 13: North America Mycoplasma Testing Industry Revenue Share (%), by Application 2025 & 2033
- Figure 14: North America Mycoplasma Testing Industry Volume Share (%), by Application 2025 & 2033
- Figure 15: North America Mycoplasma Testing Industry Revenue (million), by Country 2025 & 2033
- Figure 16: North America Mycoplasma Testing Industry Volume (K Unit), by Country 2025 & 2033
- Figure 17: North America Mycoplasma Testing Industry Revenue Share (%), by Country 2025 & 2033
- Figure 18: North America Mycoplasma Testing Industry Volume Share (%), by Country 2025 & 2033
- Figure 19: Europe Mycoplasma Testing Industry Revenue (million), by Product 2025 & 2033
- Figure 20: Europe Mycoplasma Testing Industry Volume (K Unit), by Product 2025 & 2033
- Figure 21: Europe Mycoplasma Testing Industry Revenue Share (%), by Product 2025 & 2033
- Figure 22: Europe Mycoplasma Testing Industry Volume Share (%), by Product 2025 & 2033
- Figure 23: Europe Mycoplasma Testing Industry Revenue (million), by Technology 2025 & 2033
- Figure 24: Europe Mycoplasma Testing Industry Volume (K Unit), by Technology 2025 & 2033
- Figure 25: Europe Mycoplasma Testing Industry Revenue Share (%), by Technology 2025 & 2033
- Figure 26: Europe Mycoplasma Testing Industry Volume Share (%), by Technology 2025 & 2033
- Figure 27: Europe Mycoplasma Testing Industry Revenue (million), by Application 2025 & 2033
- Figure 28: Europe Mycoplasma Testing Industry Volume (K Unit), by Application 2025 & 2033
- Figure 29: Europe Mycoplasma Testing Industry Revenue Share (%), by Application 2025 & 2033
- Figure 30: Europe Mycoplasma Testing Industry Volume Share (%), by Application 2025 & 2033
- Figure 31: Europe Mycoplasma Testing Industry Revenue (million), by Country 2025 & 2033
- Figure 32: Europe Mycoplasma Testing Industry Volume (K Unit), by Country 2025 & 2033
- Figure 33: Europe Mycoplasma Testing Industry Revenue Share (%), by Country 2025 & 2033
- Figure 34: Europe Mycoplasma Testing Industry Volume Share (%), by Country 2025 & 2033
- Figure 35: Asia Pacific Mycoplasma Testing Industry Revenue (million), by Product 2025 & 2033
- Figure 36: Asia Pacific Mycoplasma Testing Industry Volume (K Unit), by Product 2025 & 2033
- Figure 37: Asia Pacific Mycoplasma Testing Industry Revenue Share (%), by Product 2025 & 2033
- Figure 38: Asia Pacific Mycoplasma Testing Industry Volume Share (%), by Product 2025 & 2033
- Figure 39: Asia Pacific Mycoplasma Testing Industry Revenue (million), by Technology 2025 & 2033
- Figure 40: Asia Pacific Mycoplasma Testing Industry Volume (K Unit), by Technology 2025 & 2033
- Figure 41: Asia Pacific Mycoplasma Testing Industry Revenue Share (%), by Technology 2025 & 2033
- Figure 42: Asia Pacific Mycoplasma Testing Industry Volume Share (%), by Technology 2025 & 2033
- Figure 43: Asia Pacific Mycoplasma Testing Industry Revenue (million), by Application 2025 & 2033
- Figure 44: Asia Pacific Mycoplasma Testing Industry Volume (K Unit), by Application 2025 & 2033
- Figure 45: Asia Pacific Mycoplasma Testing Industry Revenue Share (%), by Application 2025 & 2033
- Figure 46: Asia Pacific Mycoplasma Testing Industry Volume Share (%), by Application 2025 & 2033
- Figure 47: Asia Pacific Mycoplasma Testing Industry Revenue (million), by Country 2025 & 2033
- Figure 48: Asia Pacific Mycoplasma Testing Industry Volume (K Unit), by Country 2025 & 2033
- Figure 49: Asia Pacific Mycoplasma Testing Industry Revenue Share (%), by Country 2025 & 2033
- Figure 50: Asia Pacific Mycoplasma Testing Industry Volume Share (%), by Country 2025 & 2033
- Figure 51: Middle East and Africa Mycoplasma Testing Industry Revenue (million), by Product 2025 & 2033
- Figure 52: Middle East and Africa Mycoplasma Testing Industry Volume (K Unit), by Product 2025 & 2033
- Figure 53: Middle East and Africa Mycoplasma Testing Industry Revenue Share (%), by Product 2025 & 2033
- Figure 54: Middle East and Africa Mycoplasma Testing Industry Volume Share (%), by Product 2025 & 2033
- Figure 55: Middle East and Africa Mycoplasma Testing Industry Revenue (million), by Technology 2025 & 2033
- Figure 56: Middle East and Africa Mycoplasma Testing Industry Volume (K Unit), by Technology 2025 & 2033
- Figure 57: Middle East and Africa Mycoplasma Testing Industry Revenue Share (%), by Technology 2025 & 2033
- Figure 58: Middle East and Africa Mycoplasma Testing Industry Volume Share (%), by Technology 2025 & 2033
- Figure 59: Middle East and Africa Mycoplasma Testing Industry Revenue (million), by Application 2025 & 2033
- Figure 60: Middle East and Africa Mycoplasma Testing Industry Volume (K Unit), by Application 2025 & 2033
- Figure 61: Middle East and Africa Mycoplasma Testing Industry Revenue Share (%), by Application 2025 & 2033
- Figure 62: Middle East and Africa Mycoplasma Testing Industry Volume Share (%), by Application 2025 & 2033
- Figure 63: Middle East and Africa Mycoplasma Testing Industry Revenue (million), by Country 2025 & 2033
- Figure 64: Middle East and Africa Mycoplasma Testing Industry Volume (K Unit), by Country 2025 & 2033
- Figure 65: Middle East and Africa Mycoplasma Testing Industry Revenue Share (%), by Country 2025 & 2033
- Figure 66: Middle East and Africa Mycoplasma Testing Industry Volume Share (%), by Country 2025 & 2033
- Figure 67: South America Mycoplasma Testing Industry Revenue (million), by Product 2025 & 2033
- Figure 68: South America Mycoplasma Testing Industry Volume (K Unit), by Product 2025 & 2033
- Figure 69: South America Mycoplasma Testing Industry Revenue Share (%), by Product 2025 & 2033
- Figure 70: South America Mycoplasma Testing Industry Volume Share (%), by Product 2025 & 2033
- Figure 71: South America Mycoplasma Testing Industry Revenue (million), by Technology 2025 & 2033
- Figure 72: South America Mycoplasma Testing Industry Volume (K Unit), by Technology 2025 & 2033
- Figure 73: South America Mycoplasma Testing Industry Revenue Share (%), by Technology 2025 & 2033
- Figure 74: South America Mycoplasma Testing Industry Volume Share (%), by Technology 2025 & 2033
- Figure 75: South America Mycoplasma Testing Industry Revenue (million), by Application 2025 & 2033
- Figure 76: South America Mycoplasma Testing Industry Volume (K Unit), by Application 2025 & 2033
- Figure 77: South America Mycoplasma Testing Industry Revenue Share (%), by Application 2025 & 2033
- Figure 78: South America Mycoplasma Testing Industry Volume Share (%), by Application 2025 & 2033
- Figure 79: South America Mycoplasma Testing Industry Revenue (million), by Country 2025 & 2033
- Figure 80: South America Mycoplasma Testing Industry Volume (K Unit), by Country 2025 & 2033
- Figure 81: South America Mycoplasma Testing Industry Revenue Share (%), by Country 2025 & 2033
- Figure 82: South America Mycoplasma Testing Industry Volume Share (%), by Country 2025 & 2033
List of Tables
- Table 1: Global Mycoplasma Testing Industry Revenue million Forecast, by Product 2020 & 2033
- Table 2: Global Mycoplasma Testing Industry Volume K Unit Forecast, by Product 2020 & 2033
- Table 3: Global Mycoplasma Testing Industry Revenue million Forecast, by Technology 2020 & 2033
- Table 4: Global Mycoplasma Testing Industry Volume K Unit Forecast, by Technology 2020 & 2033
- Table 5: Global Mycoplasma Testing Industry Revenue million Forecast, by Application 2020 & 2033
- Table 6: Global Mycoplasma Testing Industry Volume K Unit Forecast, by Application 2020 & 2033
- Table 7: Global Mycoplasma Testing Industry Revenue million Forecast, by Region 2020 & 2033
- Table 8: Global Mycoplasma Testing Industry Volume K Unit Forecast, by Region 2020 & 2033
- Table 9: Global Mycoplasma Testing Industry Revenue million Forecast, by Product 2020 & 2033
- Table 10: Global Mycoplasma Testing Industry Volume K Unit Forecast, by Product 2020 & 2033
- Table 11: Global Mycoplasma Testing Industry Revenue million Forecast, by Technology 2020 & 2033
- Table 12: Global Mycoplasma Testing Industry Volume K Unit Forecast, by Technology 2020 & 2033
- Table 13: Global Mycoplasma Testing Industry Revenue million Forecast, by Application 2020 & 2033
- Table 14: Global Mycoplasma Testing Industry Volume K Unit Forecast, by Application 2020 & 2033
- Table 15: Global Mycoplasma Testing Industry Revenue million Forecast, by Country 2020 & 2033
- Table 16: Global Mycoplasma Testing Industry Volume K Unit Forecast, by Country 2020 & 2033
- Table 17: United States Mycoplasma Testing Industry Revenue (million) Forecast, by Application 2020 & 2033
- Table 18: United States Mycoplasma Testing Industry Volume (K Unit) Forecast, by Application 2020 & 2033
- Table 19: Canada Mycoplasma Testing Industry Revenue (million) Forecast, by Application 2020 & 2033
- Table 20: Canada Mycoplasma Testing Industry Volume (K Unit) Forecast, by Application 2020 & 2033
- Table 21: Mexico Mycoplasma Testing Industry Revenue (million) Forecast, by Application 2020 & 2033
- Table 22: Mexico Mycoplasma Testing Industry Volume (K Unit) Forecast, by Application 2020 & 2033
- Table 23: Global Mycoplasma Testing Industry Revenue million Forecast, by Product 2020 & 2033
- Table 24: Global Mycoplasma Testing Industry Volume K Unit Forecast, by Product 2020 & 2033
- Table 25: Global Mycoplasma Testing Industry Revenue million Forecast, by Technology 2020 & 2033
- Table 26: Global Mycoplasma Testing Industry Volume K Unit Forecast, by Technology 2020 & 2033
- Table 27: Global Mycoplasma Testing Industry Revenue million Forecast, by Application 2020 & 2033
- Table 28: Global Mycoplasma Testing Industry Volume K Unit Forecast, by Application 2020 & 2033
- Table 29: Global Mycoplasma Testing Industry Revenue million Forecast, by Country 2020 & 2033
- Table 30: Global Mycoplasma Testing Industry Volume K Unit Forecast, by Country 2020 & 2033
- Table 31: Germany Mycoplasma Testing Industry Revenue (million) Forecast, by Application 2020 & 2033
- Table 32: Germany Mycoplasma Testing Industry Volume (K Unit) Forecast, by Application 2020 & 2033
- Table 33: United Kingdom Mycoplasma Testing Industry Revenue (million) Forecast, by Application 2020 & 2033
- Table 34: United Kingdom Mycoplasma Testing Industry Volume (K Unit) Forecast, by Application 2020 & 2033
- Table 35: France Mycoplasma Testing Industry Revenue (million) Forecast, by Application 2020 & 2033
- Table 36: France Mycoplasma Testing Industry Volume (K Unit) Forecast, by Application 2020 & 2033
- Table 37: Italy Mycoplasma Testing Industry Revenue (million) Forecast, by Application 2020 & 2033
- Table 38: Italy Mycoplasma Testing Industry Volume (K Unit) Forecast, by Application 2020 & 2033
- Table 39: Spain Mycoplasma Testing Industry Revenue (million) Forecast, by Application 2020 & 2033
- Table 40: Spain Mycoplasma Testing Industry Volume (K Unit) Forecast, by Application 2020 & 2033
- Table 41: Rest of Europe Mycoplasma Testing Industry Revenue (million) Forecast, by Application 2020 & 2033
- Table 42: Rest of Europe Mycoplasma Testing Industry Volume (K Unit) Forecast, by Application 2020 & 2033
- Table 43: Global Mycoplasma Testing Industry Revenue million Forecast, by Product 2020 & 2033
- Table 44: Global Mycoplasma Testing Industry Volume K Unit Forecast, by Product 2020 & 2033
- Table 45: Global Mycoplasma Testing Industry Revenue million Forecast, by Technology 2020 & 2033
- Table 46: Global Mycoplasma Testing Industry Volume K Unit Forecast, by Technology 2020 & 2033
- Table 47: Global Mycoplasma Testing Industry Revenue million Forecast, by Application 2020 & 2033
- Table 48: Global Mycoplasma Testing Industry Volume K Unit Forecast, by Application 2020 & 2033
- Table 49: Global Mycoplasma Testing Industry Revenue million Forecast, by Country 2020 & 2033
- Table 50: Global Mycoplasma Testing Industry Volume K Unit Forecast, by Country 2020 & 2033
- Table 51: China Mycoplasma Testing Industry Revenue (million) Forecast, by Application 2020 & 2033
- Table 52: China Mycoplasma Testing Industry Volume (K Unit) Forecast, by Application 2020 & 2033
- Table 53: Japan Mycoplasma Testing Industry Revenue (million) Forecast, by Application 2020 & 2033
- Table 54: Japan Mycoplasma Testing Industry Volume (K Unit) Forecast, by Application 2020 & 2033
- Table 55: India Mycoplasma Testing Industry Revenue (million) Forecast, by Application 2020 & 2033
- Table 56: India Mycoplasma Testing Industry Volume (K Unit) Forecast, by Application 2020 & 2033
- Table 57: Australia Mycoplasma Testing Industry Revenue (million) Forecast, by Application 2020 & 2033
- Table 58: Australia Mycoplasma Testing Industry Volume (K Unit) Forecast, by Application 2020 & 2033
- Table 59: South Korea Mycoplasma Testing Industry Revenue (million) Forecast, by Application 2020 & 2033
- Table 60: South Korea Mycoplasma Testing Industry Volume (K Unit) Forecast, by Application 2020 & 2033
- Table 61: Rest of Asia Pacific Mycoplasma Testing Industry Revenue (million) Forecast, by Application 2020 & 2033
- Table 62: Rest of Asia Pacific Mycoplasma Testing Industry Volume (K Unit) Forecast, by Application 2020 & 2033
- Table 63: Global Mycoplasma Testing Industry Revenue million Forecast, by Product 2020 & 2033
- Table 64: Global Mycoplasma Testing Industry Volume K Unit Forecast, by Product 2020 & 2033
- Table 65: Global Mycoplasma Testing Industry Revenue million Forecast, by Technology 2020 & 2033
- Table 66: Global Mycoplasma Testing Industry Volume K Unit Forecast, by Technology 2020 & 2033
- Table 67: Global Mycoplasma Testing Industry Revenue million Forecast, by Application 2020 & 2033
- Table 68: Global Mycoplasma Testing Industry Volume K Unit Forecast, by Application 2020 & 2033
- Table 69: Global Mycoplasma Testing Industry Revenue million Forecast, by Country 2020 & 2033
- Table 70: Global Mycoplasma Testing Industry Volume K Unit Forecast, by Country 2020 & 2033
- Table 71: GCC Mycoplasma Testing Industry Revenue (million) Forecast, by Application 2020 & 2033
- Table 72: GCC Mycoplasma Testing Industry Volume (K Unit) Forecast, by Application 2020 & 2033
- Table 73: South Africa Mycoplasma Testing Industry Revenue (million) Forecast, by Application 2020 & 2033
- Table 74: South Africa Mycoplasma Testing Industry Volume (K Unit) Forecast, by Application 2020 & 2033
- Table 75: Rest of Middle East and Africa Mycoplasma Testing Industry Revenue (million) Forecast, by Application 2020 & 2033
- Table 76: Rest of Middle East and Africa Mycoplasma Testing Industry Volume (K Unit) Forecast, by Application 2020 & 2033
- Table 77: Global Mycoplasma Testing Industry Revenue million Forecast, by Product 2020 & 2033
- Table 78: Global Mycoplasma Testing Industry Volume K Unit Forecast, by Product 2020 & 2033
- Table 79: Global Mycoplasma Testing Industry Revenue million Forecast, by Technology 2020 & 2033
- Table 80: Global Mycoplasma Testing Industry Volume K Unit Forecast, by Technology 2020 & 2033
- Table 81: Global Mycoplasma Testing Industry Revenue million Forecast, by Application 2020 & 2033
- Table 82: Global Mycoplasma Testing Industry Volume K Unit Forecast, by Application 2020 & 2033
- Table 83: Global Mycoplasma Testing Industry Revenue million Forecast, by Country 2020 & 2033
- Table 84: Global Mycoplasma Testing Industry Volume K Unit Forecast, by Country 2020 & 2033
- Table 85: Brazil Mycoplasma Testing Industry Revenue (million) Forecast, by Application 2020 & 2033
- Table 86: Brazil Mycoplasma Testing Industry Volume (K Unit) Forecast, by Application 2020 & 2033
- Table 87: Argentina Mycoplasma Testing Industry Revenue (million) Forecast, by Application 2020 & 2033
- Table 88: Argentina Mycoplasma Testing Industry Volume (K Unit) Forecast, by Application 2020 & 2033
- Table 89: Rest of South America Mycoplasma Testing Industry Revenue (million) Forecast, by Application 2020 & 2033
- Table 90: Rest of South America Mycoplasma Testing Industry Volume (K Unit) Forecast, by Application 2020 & 2033
Frequently Asked Questions
1. What is the projected Compound Annual Growth Rate (CAGR) of the Mycoplasma Testing Industry?
The projected CAGR is approximately 5.3%.
2. Which companies are prominent players in the Mycoplasma Testing Industry?
Key companies in the market include Merck KGaA, Eurofins Scientific, Charles River Laboratories International Inc, Thermo Fisher Scientific Inc, Bionique Testing Laboratories Inc, PromoCell GmbH, Lonza Group Ltd, Sartorius AG, ATCC, AdvaCare, Agilent Technologies.
3. What are the main segments of the Mycoplasma Testing Industry?
The market segments include Product, Technology, Application.
4. Can you provide details about the market size?
The market size is estimated to be USD 972.3 million as of 2022.
5. What are some drivers contributing to market growth?
Increased Government Initiatives and Funding in Research Activities; Increasing Demand for Fast. Accurate. and Affordable Testing; Increasing Cell Culture Contamination.
6. What are the notable trends driving market growth?
Polymerase Chain Reaction (PCR) is Expected to Hold a Significant Market Share Over the Forecast Period.
7. Are there any restraints impacting market growth?
Long and Laborious Detection Process.
8. Can you provide examples of recent developments in the market?
In May 2022, Abbott received United States Food and Drug Administration clearance for its Alinity m STI Assay. The test simultaneously detects and differentiates four common sexually transmitted infections (STIs), including Mycoplasma genitalium (MG).
9. What pricing options are available for accessing the report?
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 4750, USD 5250, and USD 8750 respectively.
10. Is the market size provided in terms of value or volume?
The market size is provided in terms of value, measured in million and volume, measured in K Unit.
11. Are there any specific market keywords associated with the report?
Yes, the market keyword associated with the report is "Mycoplasma Testing Industry," which aids in identifying and referencing the specific market segment covered.
12. How do I determine which pricing option suits my needs best?
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
13. Are there any additional resources or data provided in the Mycoplasma Testing Industry report?
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
14. How can I stay updated on further developments or reports in the Mycoplasma Testing Industry?
To stay informed about further developments, trends, and reports in the Mycoplasma Testing Industry, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.
Methodology
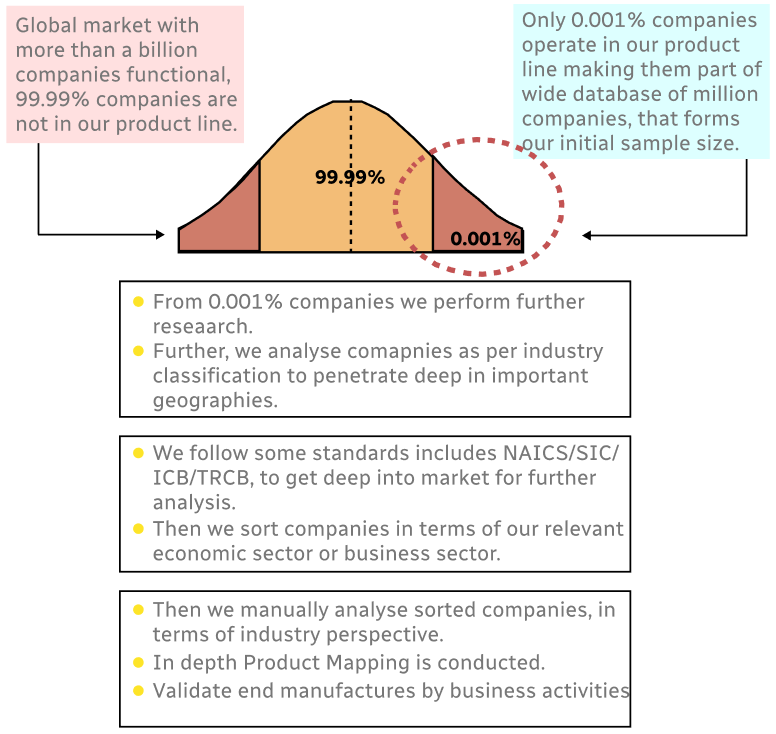
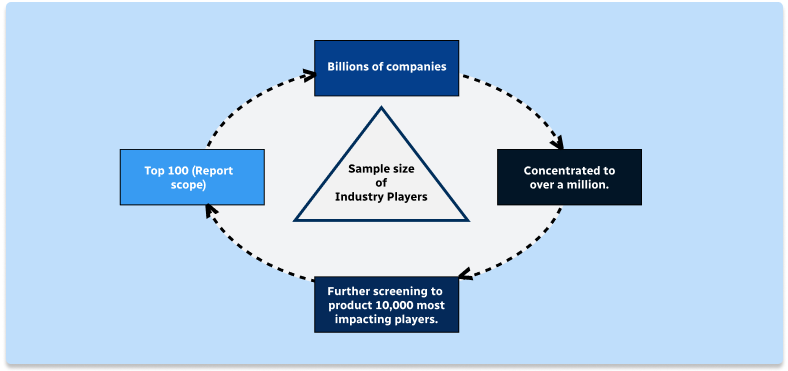
Step 1 - Identification of Relevant Samples Size from Population Database
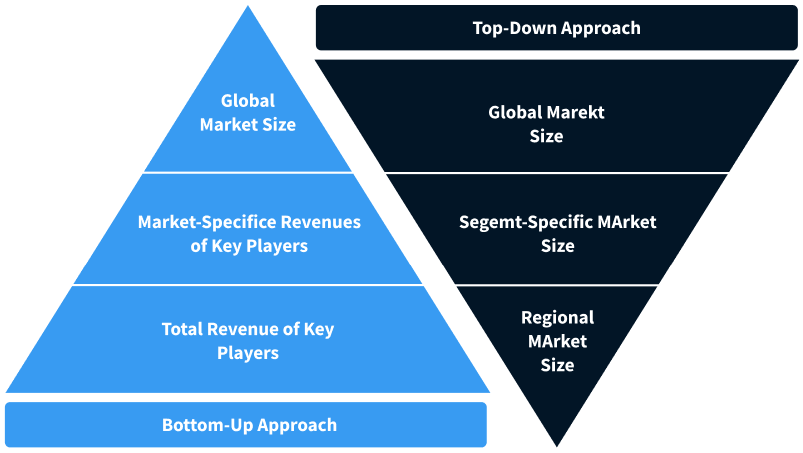
Step 2 - Approaches for Defining Global Market Size (Value, Volume* & Price*)
Note*: In applicable scenarios
Step 3 - Data Sources
Primary Research
- Web Analytics
- Survey Reports
- Research Institute
- Latest Research Reports
- Opinion Leaders
Secondary Research
- Annual Reports
- White Paper
- Latest Press Release
- Industry Association
- Paid Database
- Investor Presentations
Step 4 - Data Triangulation
Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence


