Key Insights
The INTS3 Antibody market is projected to experience robust growth, reaching an estimated $285,626.6 million in 2025 and expanding at a Compound Annual Growth Rate (CAGR) of 7% through 2033. This significant expansion is underpinned by the increasing demand for advanced diagnostic and research tools in life sciences, particularly in areas like immunochemistry, immunofluorescence, and western blotting. The rising prevalence of chronic diseases and the growing emphasis on personalized medicine are major drivers, fueling the need for specific and sensitive antibody reagents for accurate disease identification and therapeutic target validation. Furthermore, substantial investments in R&D by leading biotechnology and pharmaceutical companies are contributing to the development of novel antibody-based assays and therapies, thereby bolstering market demand. The market is characterized by a dynamic competitive landscape with numerous players, including Thermo Fisher Scientific, Abcam, and GeneTex, focusing on product innovation and strategic collaborations to expand their market reach.
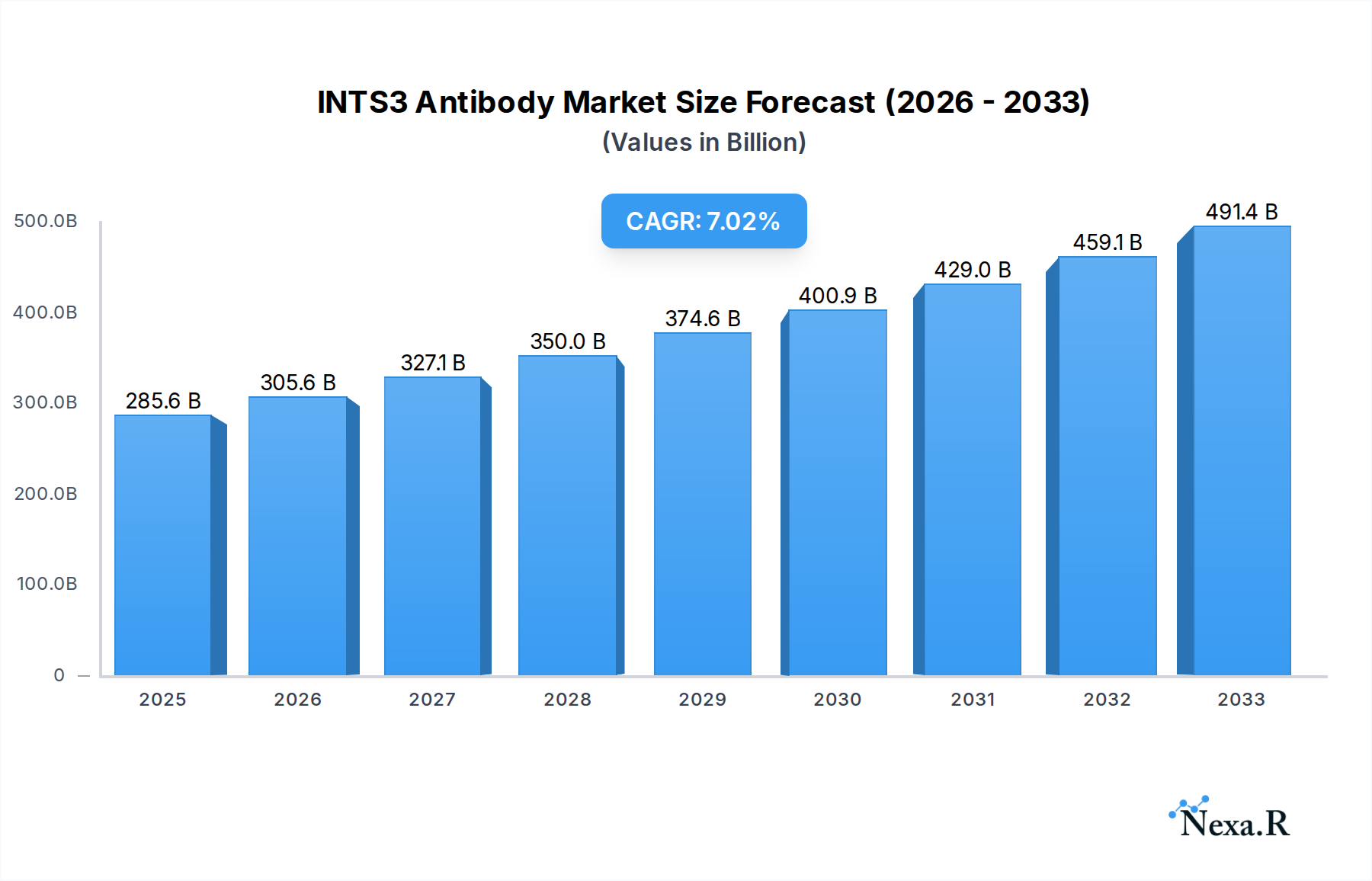
INTS3 Antibody Market Size (In Billion)

The market's growth trajectory is further supported by advancements in antibody engineering and production technologies, leading to higher specificity, sensitivity, and reliability of INTS3 antibodies. These antibodies play a crucial role in understanding the function of integrase interactor 3 (INTS3), a protein implicated in various cellular processes, including gene regulation and DNA repair. The growing applications in areas such as cancer research, neurodegenerative disease studies, and infectious disease diagnostics are expected to drive market expansion. While the market presents substantial opportunities, potential restraints include the high cost of antibody production and validation, as well as stringent regulatory requirements. However, ongoing technological advancements and increasing research funding are expected to mitigate these challenges, ensuring sustained market growth and a broadening array of applications for INTS3 antibodies in the coming years.
INTS3 Antibody Company Market Share

INTS3 Antibody Market Dynamics & Structure
The INTS3 Antibody market is characterized by a moderately concentrated structure, with leading players like Thermo Fisher Scientific and Abcam holding significant market share. Technological innovation is primarily driven by advancements in antibody production techniques, including recombinant DNA technology and single-B cell cloning, leading to higher specificity and reduced batch-to-batch variability. Key regulatory frameworks governing the development, manufacturing, and distribution of biological reagents are crucial, with a strong emphasis on quality control and validation. Competitive product substitutes include other antibodies targeting INTS3 pathway components, as well as alternative detection methods. End-user demographics primarily comprise academic research institutions, pharmaceutical and biotechnology companies, and contract research organizations (CROs) engaged in cancer research, developmental biology, and neuroscience. Mergers and acquisitions (M&A) trends are observed, often aimed at expanding product portfolios, gaining access to new technologies, and consolidating market presence. For instance, strategic acquisitions of smaller antibody developers by larger life science conglomerates can significantly alter market concentration. Barriers to innovation include the high cost of research and development, the complex validation process for diagnostic applications, and the need for specialized expertise.
- Market Concentration: Moderate, with key players like Thermo Fisher Scientific, Abcam, GeneTex, and Proteintech Group dominating.
- Technological Innovation Drivers: Recombinant antibody production, CRISPR-based antibody engineering, improved antibody conjugation technologies.
- Regulatory Frameworks: FDA, EMA, and other regional health authority guidelines for biological reagents and research tools.
- Competitive Product Substitutes: Other INTS3 pathway antibodies, RNA interference (RNAi) based knockdown technologies, small molecule inhibitors.
- End-User Demographics: Academic researchers (oncology, developmental biology, neuroscience), pharmaceutical R&D, biotechnology companies, CROs.
- M&A Trends: Acquisitions aimed at portfolio expansion and technology integration, contributing to market consolidation.
INTS3 Antibody Growth Trends & Insights
The global INTS3 antibody market has witnessed substantial growth, projected to expand at a robust Compound Annual Growth Rate (CAGR) of approximately 8.5% from 2019 to 2033, reaching an estimated market size of $380 million in 2025 and projected to touch $650 million by 2033. This upward trajectory is fueled by an increasing demand for highly specific and sensitive antibodies in fundamental biological research and drug discovery. The base year, 2025, sees a market valuation of $380 million, with the forecast period (2025-2033) anticipating continued expansion driven by evolving scientific understanding of the INTS3 protein’s multifaceted roles in cellular processes, including gene transcription regulation and developmental pathways. Adoption rates for INTS3 antibodies are steadily increasing across various applications, particularly in understanding developmental disorders, cancer progression, and neurological conditions. Technological disruptions, such as the development of highly validated monoclonal antibodies and improved immunoassay techniques, have significantly enhanced the reliability and utility of INTS3 antibodies. Consumer behavior shifts are also playing a crucial role; researchers are increasingly prioritizing antibodies with comprehensive validation data, proven performance across multiple applications, and reliable supply chains. The market penetration of INTS3 antibodies is deepening as its relevance in disease research becomes more apparent, leading to a growing user base in both academic and commercial sectors. The historical period (2019-2024) laid the groundwork for this growth, characterized by foundational research and the initial commercialization of key antibody products. The estimated year, 2025, serves as a benchmark for current market standing, with projections for sustained, accelerated growth. This evolving landscape necessitates continuous innovation from manufacturers to meet the sophisticated demands of the research community. The market’s expansion is intrinsically linked to breakthroughs in understanding complex biological mechanisms where INTS3 plays a pivotal role, thereby driving demand for specific and high-quality antibodies. The overall market size is directly proportional to the research output and drug development pipelines that rely on precise molecular targets.
Dominant Regions, Countries, or Segments in INTS3 Antibody
The North America region currently dominates the INTS3 antibody market, driven by its robust research infrastructure, substantial government funding for scientific research, and a high concentration of leading pharmaceutical and biotechnology companies. The United States, in particular, is a powerhouse, accounting for a significant portion of the global market share, estimated at 35% of the total market value in 2025, projected to reach approximately $133 million. This dominance is underpinned by significant investments in life sciences research, particularly in oncology and developmental biology, where INTS3 plays a critical role. The presence of major academic institutions, research hospitals, and a thriving biotech sector fosters high demand for research reagents like INTS3 antibodies. Favorable regulatory environments and strong intellectual property protection further incentivize innovation and commercialization within the region. Economically, the strong purchasing power of research institutions and a culture of early adoption of advanced technologies contribute to North America's leading position.
Within the segmentation of applications, Immunochemistry (IHC) stands out as the most dominant segment, capturing an estimated 25% of the INTS3 antibody market share in 2025, valued at approximately $95 million. IHC is crucial for visualizing protein expression within its native tissue context, making it indispensable for studying INTS3’s role in cellular localization, disease pathogenesis, and drug efficacy in preclinical and clinical research. The segment's growth is propelled by advancements in multiplex IHC techniques and the increasing demand for precise spatial biology insights.
In terms of antibody types, Monoclonal antibodies are projected to hold a larger market share, estimated at 60% in 2025, valued at approximately $228 million, compared to Polyclonal antibodies, which are estimated at 40%, valued at approximately $152 million. Monoclonal antibodies offer higher specificity and batch-to-batch consistency, which are critical for reproducible research and the development of potential diagnostic or therapeutic applications.
- Dominant Region: North America (United States leading).
- Key Drivers: Strong R&D funding, presence of major pharmaceutical/biotech companies, advanced research infrastructure, favorable regulatory policies.
- Market Share (2025): ~35% of global market value.
- Dominant Application Segment: Immunochemistry (IHC).
- Key Drivers: Essential for spatial and cellular protein localization studies, advancements in multiplexing, role in cancer and developmental biology research.
- Market Share (2025): ~25% of global market value.
- Dominant Antibody Type: Monoclonal Antibodies.
- Key Drivers: High specificity, consistency, suitability for downstream applications and potential diagnostics.
- Market Share (2025): ~60% of antibody type segment.
INTS3 Antibody Product Landscape
The INTS3 antibody product landscape is characterized by continuous innovation aimed at enhancing specificity, sensitivity, and performance across diverse research applications. Manufacturers are focusing on developing highly validated monoclonal antibodies that exhibit minimal cross-reactivity, crucial for accurate downstream analysis. Products are increasingly offered with comprehensive validation data, including Western Blot (WB), Immunohistochemistry (IHC), Immunofluorescence (IF), and Immunoprecipitation (IP) protocols, demonstrating robust performance across multiple platforms. Unique selling propositions include antibodies validated for specific model organisms, conjugated antibodies for direct detection, and recombinant antibodies produced through advanced biotechnological processes for superior consistency and scalability. Technological advancements are leading to improved antibody clones with higher affinity and avidity, enabling detection of low-abundance INTS3 protein levels.
Key Drivers, Barriers & Challenges in INTS3 Antibody
Key Drivers:
- Increasing Research in Cancer and Developmental Biology: INTS3's critical role in gene regulation and cellular development makes it a key target for understanding cancer progression and birth defects, driving demand for INTS3 antibodies.
- Advancements in Proteomics and Genomics: The growing application of proteomic and genomic techniques necessitates highly specific antibodies for validating gene and protein expression, including INTS3.
- Growth of Biopharmaceutical Industry: Expansion in drug discovery and development, particularly in areas related to transcription regulation, fuels the need for reliable research tools like INTS3 antibodies.
- Technological Innovations in Antibody Production: Development of recombinant antibodies and advanced validation techniques enhance product quality and broaden applications.
Barriers & Challenges:
- High Cost of Validation and Production: Rigorous validation processes and specialized manufacturing techniques contribute to the high cost of producing high-quality INTS3 antibodies, impacting affordability.
- Availability of Alternative Research Tools: While INTS3 antibodies are crucial, alternative methods like RNAi or gene editing can sometimes serve as complementary or competitive approaches, potentially limiting market share.
- Stringent Regulatory Requirements for Clinical Applications: For any potential diagnostic or therapeutic use, INTS3 antibodies face lengthy and costly regulatory approval processes.
- Supply Chain Disruptions: Global events or logistical challenges can impact the availability and timely delivery of specialized biological reagents, affecting research continuity.
Emerging Opportunities in INTS3 Antibody
Emerging opportunities in the INTS3 antibody market lie in the development of highly multiplexed immunoassay kits for simultaneous detection of INTS3 and other pathway proteins, enabling a more comprehensive understanding of cellular signaling. The untapped potential of INTS3 antibodies in neurodegenerative disease research, particularly in Alzheimer's and Parkinson's disease, presents a significant growth avenue. Furthermore, the development of INTS3 antibody fragments or nanobodies for targeted drug delivery or imaging applications, and their application in personalized medicine approaches, are nascent but promising areas.
Growth Accelerators in the INTS3 Antibody Industry
Long-term growth in the INTS3 antibody industry will be accelerated by strategic partnerships between antibody manufacturers and academic research institutions focused on translational research. The increasing adoption of automated high-throughput screening platforms in drug discovery will necessitate a greater demand for standardized and reliable INTS3 antibodies. Moreover, the expansion of personalized medicine initiatives, which require precise molecular profiling, will further drive the need for highly specific reagents like INTS3 antibodies, especially in oncology.
Key Players Shaping the INTS3 Antibody Market
- Thermo Fisher Scientific
- LifeSpan BioSciences
- United States Biological
- NSJ Bioreagents
- RayBiotech
- Abcam
- GeneTex
- Bioss
- Affinity Biosciences
- Abbexa
- Proteintech Group
- Novus Biologicals
- Bethyl Laboratories
- St John's Laboratory
- AntibodySystem
- Creative Biolabs
- CUSABIO Technology LLC
- Biobyt
- Jingjie PTM BioLab
Notable Milestones in INTS3 Antibody Sector
- 2019: Increased research publications highlighting INTS3's role in chromatin remodeling and transcription, boosting antibody demand.
- 2020: Development and commercialization of highly validated monoclonal INTS3 antibodies by key players, improving research reproducibility.
- 2021: Advancements in recombinant antibody technology leading to more consistent and scalable INTS3 antibody production.
- 2022: Expansion of INTS3 antibody applications into new research areas like epigenetics and developmental disorders.
- 2023: Introduction of INTS3 antibodies validated for multiplexing applications in advanced imaging techniques.
- 2024: Growing interest in INTS3's role in specific cancer types, leading to increased demand for targeted antibodies from pharmaceutical R&D.
In-Depth INTS3 Antibody Market Outlook
The INTS3 antibody market outlook remains exceptionally strong, driven by its fundamental importance in unraveling complex biological mechanisms. Future growth will be significantly propelled by intensified research efforts in oncology, developmental biology, and neurobiology, where INTS3's intricate roles are continually being elucidated. The market will benefit from the ongoing integration of advanced antibody technologies, such as CRISPR-validated antibodies and custom conjugation services, offering researchers greater precision and flexibility. Strategic collaborations between academic centers and commercial entities will further accelerate the translation of INTS3 research into potential therapeutic insights, solidifying the INTS3 antibody as an indispensable tool in the life sciences ecosystem.
INTS3 Antibody Segmentation
-
1. Application
- 1.1. Immunochemistry (IHC)
- 1.2. Immunofluorescence (IF)
- 1.3. Immunoprecipitation (IP)
- 1.4. Western Blot (WB)
- 1.5. ELISA
- 1.6. Others
-
2. Types
- 2.1. Monoclonal
- 2.2. Polyclonal
INTS3 Antibody Segmentation By Geography
-
1. North America
- 1.1. United States
- 1.2. Canada
- 1.3. Mexico
-
2. South America
- 2.1. Brazil
- 2.2. Argentina
- 2.3. Rest of South America
-
3. Europe
- 3.1. United Kingdom
- 3.2. Germany
- 3.3. France
- 3.4. Italy
- 3.5. Spain
- 3.6. Russia
- 3.7. Benelux
- 3.8. Nordics
- 3.9. Rest of Europe
-
4. Middle East & Africa
- 4.1. Turkey
- 4.2. Israel
- 4.3. GCC
- 4.4. North Africa
- 4.5. South Africa
- 4.6. Rest of Middle East & Africa
-
5. Asia Pacific
- 5.1. China
- 5.2. India
- 5.3. Japan
- 5.4. South Korea
- 5.5. ASEAN
- 5.6. Oceania
- 5.7. Rest of Asia Pacific
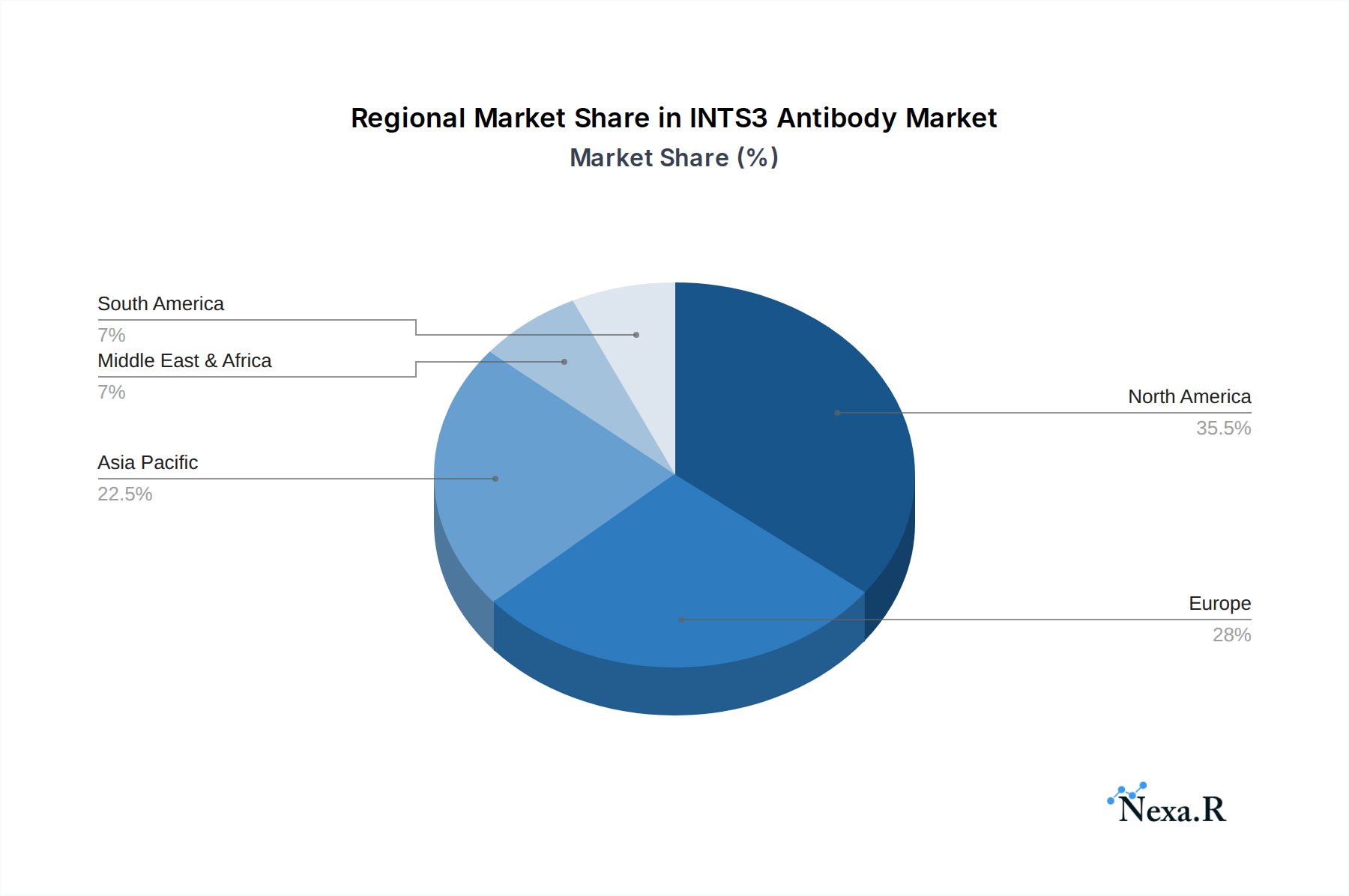
INTS3 Antibody Regional Market Share

Geographic Coverage of INTS3 Antibody
INTS3 Antibody REPORT HIGHLIGHTS
| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 7% from 2020-2034 |
| Segmentation |
|
Table of Contents
- 1. Introduction
- 1.1. Research Scope
- 1.2. Market Segmentation
- 1.3. Research Methodology
- 1.4. Definitions and Assumptions
- 2. Executive Summary
- 2.1. Introduction
- 3. Market Dynamics
- 3.1. Introduction
- 3.2. Market Drivers
- 3.3. Market Restrains
- 3.4. Market Trends
- 4. Market Factor Analysis
- 4.1. Porters Five Forces
- 4.2. Supply/Value Chain
- 4.3. PESTEL analysis
- 4.4. Market Entropy
- 4.5. Patent/Trademark Analysis
- 5. Global INTS3 Antibody Analysis, Insights and Forecast, 2020-2032
- 5.1. Market Analysis, Insights and Forecast - by Application
- 5.1.1. Immunochemistry (IHC)
- 5.1.2. Immunofluorescence (IF)
- 5.1.3. Immunoprecipitation (IP)
- 5.1.4. Western Blot (WB)
- 5.1.5. ELISA
- 5.1.6. Others
- 5.2. Market Analysis, Insights and Forecast - by Types
- 5.2.1. Monoclonal
- 5.2.2. Polyclonal
- 5.3. Market Analysis, Insights and Forecast - by Region
- 5.3.1. North America
- 5.3.2. South America
- 5.3.3. Europe
- 5.3.4. Middle East & Africa
- 5.3.5. Asia Pacific
- 5.1. Market Analysis, Insights and Forecast - by Application
- 6. North America INTS3 Antibody Analysis, Insights and Forecast, 2020-2032
- 6.1. Market Analysis, Insights and Forecast - by Application
- 6.1.1. Immunochemistry (IHC)
- 6.1.2. Immunofluorescence (IF)
- 6.1.3. Immunoprecipitation (IP)
- 6.1.4. Western Blot (WB)
- 6.1.5. ELISA
- 6.1.6. Others
- 6.2. Market Analysis, Insights and Forecast - by Types
- 6.2.1. Monoclonal
- 6.2.2. Polyclonal
- 6.1. Market Analysis, Insights and Forecast - by Application
- 7. South America INTS3 Antibody Analysis, Insights and Forecast, 2020-2032
- 7.1. Market Analysis, Insights and Forecast - by Application
- 7.1.1. Immunochemistry (IHC)
- 7.1.2. Immunofluorescence (IF)
- 7.1.3. Immunoprecipitation (IP)
- 7.1.4. Western Blot (WB)
- 7.1.5. ELISA
- 7.1.6. Others
- 7.2. Market Analysis, Insights and Forecast - by Types
- 7.2.1. Monoclonal
- 7.2.2. Polyclonal
- 7.1. Market Analysis, Insights and Forecast - by Application
- 8. Europe INTS3 Antibody Analysis, Insights and Forecast, 2020-2032
- 8.1. Market Analysis, Insights and Forecast - by Application
- 8.1.1. Immunochemistry (IHC)
- 8.1.2. Immunofluorescence (IF)
- 8.1.3. Immunoprecipitation (IP)
- 8.1.4. Western Blot (WB)
- 8.1.5. ELISA
- 8.1.6. Others
- 8.2. Market Analysis, Insights and Forecast - by Types
- 8.2.1. Monoclonal
- 8.2.2. Polyclonal
- 8.1. Market Analysis, Insights and Forecast - by Application
- 9. Middle East & Africa INTS3 Antibody Analysis, Insights and Forecast, 2020-2032
- 9.1. Market Analysis, Insights and Forecast - by Application
- 9.1.1. Immunochemistry (IHC)
- 9.1.2. Immunofluorescence (IF)
- 9.1.3. Immunoprecipitation (IP)
- 9.1.4. Western Blot (WB)
- 9.1.5. ELISA
- 9.1.6. Others
- 9.2. Market Analysis, Insights and Forecast - by Types
- 9.2.1. Monoclonal
- 9.2.2. Polyclonal
- 9.1. Market Analysis, Insights and Forecast - by Application
- 10. Asia Pacific INTS3 Antibody Analysis, Insights and Forecast, 2020-2032
- 10.1. Market Analysis, Insights and Forecast - by Application
- 10.1.1. Immunochemistry (IHC)
- 10.1.2. Immunofluorescence (IF)
- 10.1.3. Immunoprecipitation (IP)
- 10.1.4. Western Blot (WB)
- 10.1.5. ELISA
- 10.1.6. Others
- 10.2. Market Analysis, Insights and Forecast - by Types
- 10.2.1. Monoclonal
- 10.2.2. Polyclonal
- 10.1. Market Analysis, Insights and Forecast - by Application
- 11. Competitive Analysis
- 11.1. Global Market Share Analysis 2025
- 11.2. Company Profiles
- 11.2.1 Thermo Fisher Scientific
- 11.2.1.1. Overview
- 11.2.1.2. Products
- 11.2.1.3. SWOT Analysis
- 11.2.1.4. Recent Developments
- 11.2.1.5. Financials (Based on Availability)
- 11.2.2 LifeSpan BioSciences
- 11.2.2.1. Overview
- 11.2.2.2. Products
- 11.2.2.3. SWOT Analysis
- 11.2.2.4. Recent Developments
- 11.2.2.5. Financials (Based on Availability)
- 11.2.3 United States Biological
- 11.2.3.1. Overview
- 11.2.3.2. Products
- 11.2.3.3. SWOT Analysis
- 11.2.3.4. Recent Developments
- 11.2.3.5. Financials (Based on Availability)
- 11.2.4 NSJ Bioreagents
- 11.2.4.1. Overview
- 11.2.4.2. Products
- 11.2.4.3. SWOT Analysis
- 11.2.4.4. Recent Developments
- 11.2.4.5. Financials (Based on Availability)
- 11.2.5 RayBiotech
- 11.2.5.1. Overview
- 11.2.5.2. Products
- 11.2.5.3. SWOT Analysis
- 11.2.5.4. Recent Developments
- 11.2.5.5. Financials (Based on Availability)
- 11.2.6 Abcam
- 11.2.6.1. Overview
- 11.2.6.2. Products
- 11.2.6.3. SWOT Analysis
- 11.2.6.4. Recent Developments
- 11.2.6.5. Financials (Based on Availability)
- 11.2.7 GeneTex
- 11.2.7.1. Overview
- 11.2.7.2. Products
- 11.2.7.3. SWOT Analysis
- 11.2.7.4. Recent Developments
- 11.2.7.5. Financials (Based on Availability)
- 11.2.8 Bioss
- 11.2.8.1. Overview
- 11.2.8.2. Products
- 11.2.8.3. SWOT Analysis
- 11.2.8.4. Recent Developments
- 11.2.8.5. Financials (Based on Availability)
- 11.2.9 Affinity Biosciences
- 11.2.9.1. Overview
- 11.2.9.2. Products
- 11.2.9.3. SWOT Analysis
- 11.2.9.4. Recent Developments
- 11.2.9.5. Financials (Based on Availability)
- 11.2.10 Abbexa
- 11.2.10.1. Overview
- 11.2.10.2. Products
- 11.2.10.3. SWOT Analysis
- 11.2.10.4. Recent Developments
- 11.2.10.5. Financials (Based on Availability)
- 11.2.11 Proteintech Group
- 11.2.11.1. Overview
- 11.2.11.2. Products
- 11.2.11.3. SWOT Analysis
- 11.2.11.4. Recent Developments
- 11.2.11.5. Financials (Based on Availability)
- 11.2.12 Novus Biologicals
- 11.2.12.1. Overview
- 11.2.12.2. Products
- 11.2.12.3. SWOT Analysis
- 11.2.12.4. Recent Developments
- 11.2.12.5. Financials (Based on Availability)
- 11.2.13 Bethyl Laboratories
- 11.2.13.1. Overview
- 11.2.13.2. Products
- 11.2.13.3. SWOT Analysis
- 11.2.13.4. Recent Developments
- 11.2.13.5. Financials (Based on Availability)
- 11.2.14 St John's Laboratory
- 11.2.14.1. Overview
- 11.2.14.2. Products
- 11.2.14.3. SWOT Analysis
- 11.2.14.4. Recent Developments
- 11.2.14.5. Financials (Based on Availability)
- 11.2.15 AntibodySystem
- 11.2.15.1. Overview
- 11.2.15.2. Products
- 11.2.15.3. SWOT Analysis
- 11.2.15.4. Recent Developments
- 11.2.15.5. Financials (Based on Availability)
- 11.2.16 Creative Biolabs
- 11.2.16.1. Overview
- 11.2.16.2. Products
- 11.2.16.3. SWOT Analysis
- 11.2.16.4. Recent Developments
- 11.2.16.5. Financials (Based on Availability)
- 11.2.17 CUSABIO Technology LLC
- 11.2.17.1. Overview
- 11.2.17.2. Products
- 11.2.17.3. SWOT Analysis
- 11.2.17.4. Recent Developments
- 11.2.17.5. Financials (Based on Availability)
- 11.2.18 Biobyt
- 11.2.18.1. Overview
- 11.2.18.2. Products
- 11.2.18.3. SWOT Analysis
- 11.2.18.4. Recent Developments
- 11.2.18.5. Financials (Based on Availability)
- 11.2.19 Jingjie PTM BioLab
- 11.2.19.1. Overview
- 11.2.19.2. Products
- 11.2.19.3. SWOT Analysis
- 11.2.19.4. Recent Developments
- 11.2.19.5. Financials (Based on Availability)
- 11.2.1 Thermo Fisher Scientific
List of Figures
- Figure 1: Global INTS3 Antibody Revenue Breakdown (undefined, %) by Region 2025 & 2033
- Figure 2: North America INTS3 Antibody Revenue (undefined), by Application 2025 & 2033
- Figure 3: North America INTS3 Antibody Revenue Share (%), by Application 2025 & 2033
- Figure 4: North America INTS3 Antibody Revenue (undefined), by Types 2025 & 2033
- Figure 5: North America INTS3 Antibody Revenue Share (%), by Types 2025 & 2033
- Figure 6: North America INTS3 Antibody Revenue (undefined), by Country 2025 & 2033
- Figure 7: North America INTS3 Antibody Revenue Share (%), by Country 2025 & 2033
- Figure 8: South America INTS3 Antibody Revenue (undefined), by Application 2025 & 2033
- Figure 9: South America INTS3 Antibody Revenue Share (%), by Application 2025 & 2033
- Figure 10: South America INTS3 Antibody Revenue (undefined), by Types 2025 & 2033
- Figure 11: South America INTS3 Antibody Revenue Share (%), by Types 2025 & 2033
- Figure 12: South America INTS3 Antibody Revenue (undefined), by Country 2025 & 2033
- Figure 13: South America INTS3 Antibody Revenue Share (%), by Country 2025 & 2033
- Figure 14: Europe INTS3 Antibody Revenue (undefined), by Application 2025 & 2033
- Figure 15: Europe INTS3 Antibody Revenue Share (%), by Application 2025 & 2033
- Figure 16: Europe INTS3 Antibody Revenue (undefined), by Types 2025 & 2033
- Figure 17: Europe INTS3 Antibody Revenue Share (%), by Types 2025 & 2033
- Figure 18: Europe INTS3 Antibody Revenue (undefined), by Country 2025 & 2033
- Figure 19: Europe INTS3 Antibody Revenue Share (%), by Country 2025 & 2033
- Figure 20: Middle East & Africa INTS3 Antibody Revenue (undefined), by Application 2025 & 2033
- Figure 21: Middle East & Africa INTS3 Antibody Revenue Share (%), by Application 2025 & 2033
- Figure 22: Middle East & Africa INTS3 Antibody Revenue (undefined), by Types 2025 & 2033
- Figure 23: Middle East & Africa INTS3 Antibody Revenue Share (%), by Types 2025 & 2033
- Figure 24: Middle East & Africa INTS3 Antibody Revenue (undefined), by Country 2025 & 2033
- Figure 25: Middle East & Africa INTS3 Antibody Revenue Share (%), by Country 2025 & 2033
- Figure 26: Asia Pacific INTS3 Antibody Revenue (undefined), by Application 2025 & 2033
- Figure 27: Asia Pacific INTS3 Antibody Revenue Share (%), by Application 2025 & 2033
- Figure 28: Asia Pacific INTS3 Antibody Revenue (undefined), by Types 2025 & 2033
- Figure 29: Asia Pacific INTS3 Antibody Revenue Share (%), by Types 2025 & 2033
- Figure 30: Asia Pacific INTS3 Antibody Revenue (undefined), by Country 2025 & 2033
- Figure 31: Asia Pacific INTS3 Antibody Revenue Share (%), by Country 2025 & 2033
List of Tables
- Table 1: Global INTS3 Antibody Revenue undefined Forecast, by Application 2020 & 2033
- Table 2: Global INTS3 Antibody Revenue undefined Forecast, by Types 2020 & 2033
- Table 3: Global INTS3 Antibody Revenue undefined Forecast, by Region 2020 & 2033
- Table 4: Global INTS3 Antibody Revenue undefined Forecast, by Application 2020 & 2033
- Table 5: Global INTS3 Antibody Revenue undefined Forecast, by Types 2020 & 2033
- Table 6: Global INTS3 Antibody Revenue undefined Forecast, by Country 2020 & 2033
- Table 7: United States INTS3 Antibody Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 8: Canada INTS3 Antibody Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 9: Mexico INTS3 Antibody Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 10: Global INTS3 Antibody Revenue undefined Forecast, by Application 2020 & 2033
- Table 11: Global INTS3 Antibody Revenue undefined Forecast, by Types 2020 & 2033
- Table 12: Global INTS3 Antibody Revenue undefined Forecast, by Country 2020 & 2033
- Table 13: Brazil INTS3 Antibody Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 14: Argentina INTS3 Antibody Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 15: Rest of South America INTS3 Antibody Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 16: Global INTS3 Antibody Revenue undefined Forecast, by Application 2020 & 2033
- Table 17: Global INTS3 Antibody Revenue undefined Forecast, by Types 2020 & 2033
- Table 18: Global INTS3 Antibody Revenue undefined Forecast, by Country 2020 & 2033
- Table 19: United Kingdom INTS3 Antibody Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 20: Germany INTS3 Antibody Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 21: France INTS3 Antibody Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 22: Italy INTS3 Antibody Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 23: Spain INTS3 Antibody Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 24: Russia INTS3 Antibody Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 25: Benelux INTS3 Antibody Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 26: Nordics INTS3 Antibody Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 27: Rest of Europe INTS3 Antibody Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 28: Global INTS3 Antibody Revenue undefined Forecast, by Application 2020 & 2033
- Table 29: Global INTS3 Antibody Revenue undefined Forecast, by Types 2020 & 2033
- Table 30: Global INTS3 Antibody Revenue undefined Forecast, by Country 2020 & 2033
- Table 31: Turkey INTS3 Antibody Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 32: Israel INTS3 Antibody Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 33: GCC INTS3 Antibody Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 34: North Africa INTS3 Antibody Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 35: South Africa INTS3 Antibody Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 36: Rest of Middle East & Africa INTS3 Antibody Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 37: Global INTS3 Antibody Revenue undefined Forecast, by Application 2020 & 2033
- Table 38: Global INTS3 Antibody Revenue undefined Forecast, by Types 2020 & 2033
- Table 39: Global INTS3 Antibody Revenue undefined Forecast, by Country 2020 & 2033
- Table 40: China INTS3 Antibody Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 41: India INTS3 Antibody Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 42: Japan INTS3 Antibody Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 43: South Korea INTS3 Antibody Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 44: ASEAN INTS3 Antibody Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 45: Oceania INTS3 Antibody Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 46: Rest of Asia Pacific INTS3 Antibody Revenue (undefined) Forecast, by Application 2020 & 2033
Frequently Asked Questions
1. What is the projected Compound Annual Growth Rate (CAGR) of the INTS3 Antibody?
The projected CAGR is approximately 7%.
2. Which companies are prominent players in the INTS3 Antibody?
Key companies in the market include Thermo Fisher Scientific, LifeSpan BioSciences, United States Biological, NSJ Bioreagents, RayBiotech, Abcam, GeneTex, Bioss, Affinity Biosciences, Abbexa, Proteintech Group, Novus Biologicals, Bethyl Laboratories, St John's Laboratory, AntibodySystem, Creative Biolabs, CUSABIO Technology LLC, Biobyt, Jingjie PTM BioLab.
3. What are the main segments of the INTS3 Antibody?
The market segments include Application, Types.
4. Can you provide details about the market size?
The market size is estimated to be USD XXX N/A as of 2022.
5. What are some drivers contributing to market growth?
N/A
6. What are the notable trends driving market growth?
N/A
7. Are there any restraints impacting market growth?
N/A
8. Can you provide examples of recent developments in the market?
N/A
9. What pricing options are available for accessing the report?
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 3350.00, USD 5025.00, and USD 6700.00 respectively.
10. Is the market size provided in terms of value or volume?
The market size is provided in terms of value, measured in N/A.
11. Are there any specific market keywords associated with the report?
Yes, the market keyword associated with the report is "INTS3 Antibody," which aids in identifying and referencing the specific market segment covered.
12. How do I determine which pricing option suits my needs best?
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
13. Are there any additional resources or data provided in the INTS3 Antibody report?
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
14. How can I stay updated on further developments or reports in the INTS3 Antibody?
To stay informed about further developments, trends, and reports in the INTS3 Antibody, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.
Methodology
Step 1 - Identification of Relevant Samples Size from Population Database
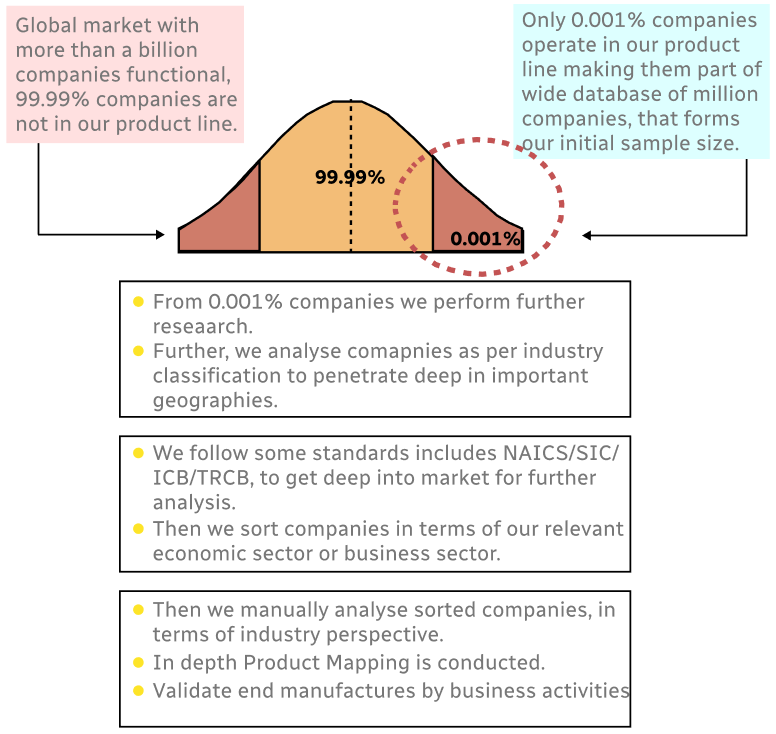
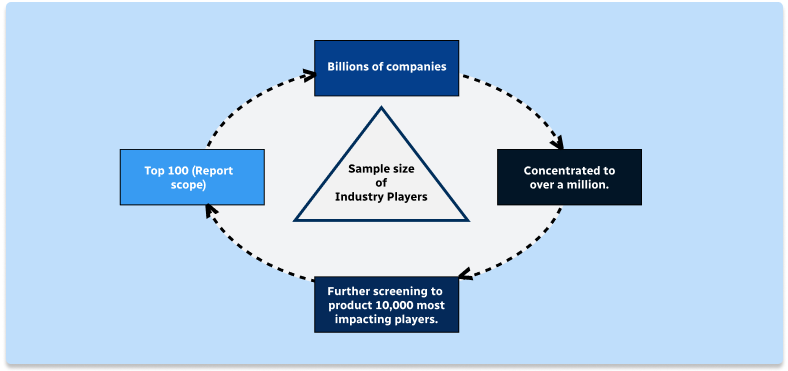
Step 2 - Approaches for Defining Global Market Size (Value, Volume* & Price*)
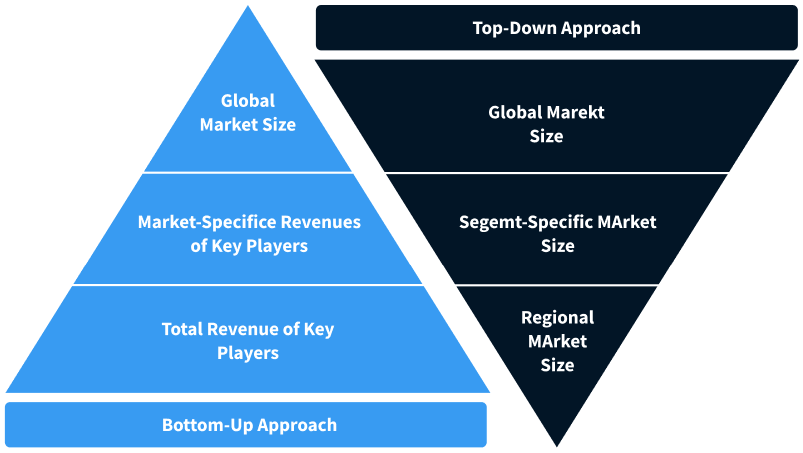
Note*: In applicable scenarios
Step 3 - Data Sources
Primary Research
- Web Analytics
- Survey Reports
- Research Institute
- Latest Research Reports
- Opinion Leaders
Secondary Research
- Annual Reports
- White Paper
- Latest Press Release
- Industry Association
- Paid Database
- Investor Presentations
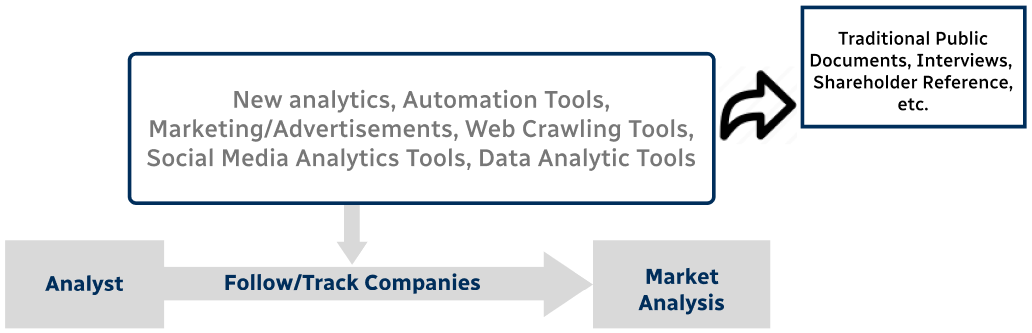
Step 4 - Data Triangulation
Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence


