Key Insights
The global handheld vital signs monitor market is experiencing robust growth, driven by the increasing prevalence of chronic diseases, rising demand for point-of-care diagnostics, and technological advancements leading to more compact and user-friendly devices. The market's expansion is further fueled by the growing adoption of telehealth and remote patient monitoring, enabling healthcare professionals to efficiently track vital signs in diverse settings – from homes and ambulances to clinics and hospitals. This demand is particularly pronounced in developing economies experiencing rapid healthcare infrastructure development and an expanding elderly population. Key players like Philips, General Electric, and Mindray are strategically investing in R&D to incorporate advanced features such as wireless connectivity, data analytics capabilities, and improved accuracy, enhancing the overall market value proposition. The integration of these monitors into existing Electronic Health Record (EHR) systems is also contributing to market expansion, streamlining data management and improving patient care.
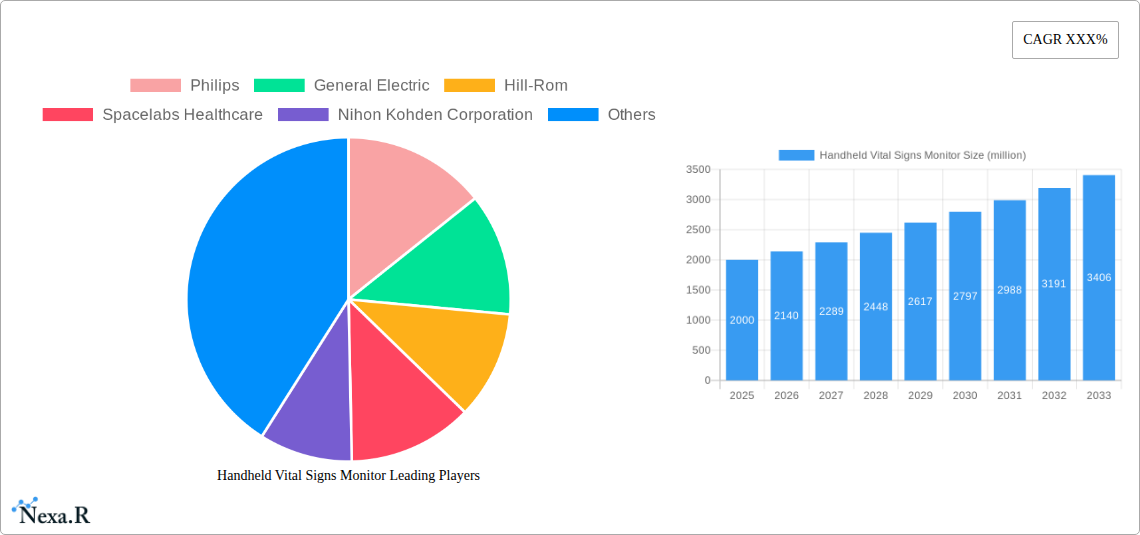
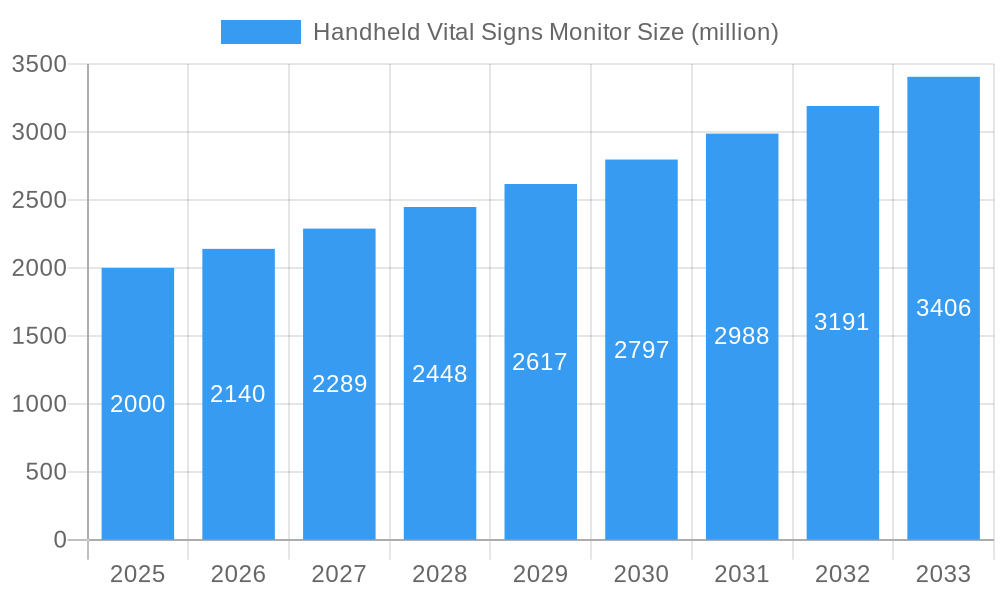
Handheld Vital Signs Monitor Market Size (In Billion)

Despite the positive growth trajectory, challenges remain. The market faces constraints related to regulatory approvals in various regions and the high cost associated with advanced features. Competition among established players and emerging market entrants is intensifying, necessitating continuous innovation and strategic partnerships to maintain a competitive edge. Segmentation within the market is primarily driven by device type (e.g., blood pressure monitors, pulse oximeters, thermometers), connectivity options (wireless vs. wired), and end-user (hospitals, clinics, home healthcare). While precise market sizing data is absent, projecting a conservative CAGR of 7% from a 2025 market size of $2 billion (an estimated figure based on industry reports on related medical device markets) suggests a significant market expansion over the next decade, with projections exceeding $3.5 billion by 2033.
Handheld Vital Signs Monitor Company Market Share

Handheld Vital Signs Monitor Market Report: 2019-2033
This comprehensive report provides a detailed analysis of the global handheld vital signs monitor market, offering invaluable insights for industry professionals, investors, and strategic decision-makers. The report covers the period 2019-2033, with a base year of 2025 and a forecast period of 2025-2033. The study leverages extensive primary and secondary research to deliver a precise and actionable understanding of market dynamics, growth trends, and competitive landscape. The market is segmented by various factors including technology, application and end-user, further broken down by geography. The total market value in 2025 is estimated at XX million units, projected to reach XX million units by 2033.
Handheld Vital Signs Monitor Market Dynamics & Structure
The handheld vital signs monitor market is characterized by a moderately consolidated structure, with key players such as Philips, General Electric, and Hill-Rom holding significant market share. Technological innovation, particularly in wireless connectivity and data analytics, is a major driver of growth. Stringent regulatory frameworks concerning medical device approvals influence market entry and product development. The market faces competition from alternative monitoring technologies, but the portability and ease of use of handheld devices maintain their strong position. M&A activity in the sector has been moderate, with several smaller companies acquired by larger players to expand their product portfolios and geographic reach. The market's end-user demographic is diverse, ranging from hospitals and clinics to home healthcare settings and emergency medical services.
- Market Concentration: Moderately consolidated, with top 5 players holding approximately xx% market share in 2025.
- Technological Innovation: Focus on wireless capabilities, improved accuracy, and integrated data analysis.
- Regulatory Framework: Stringent FDA/CE approvals impacting product development and market entry.
- Competitive Substitutes: Increasing competition from alternative monitoring technologies (e.g., wearable sensors).
- End-User Demographics: Hospitals, clinics, home healthcare, emergency medical services.
- M&A Trends: Moderate activity, with larger players acquiring smaller companies to expand market reach.
Handheld Vital Signs Monitor Growth Trends & Insights
The handheld vital signs monitor market experienced significant growth during the historical period (2019-2024), driven by increasing demand for point-of-care diagnostics, rising prevalence of chronic diseases, and advancements in medical technology. The market is projected to maintain a healthy CAGR of xx% during the forecast period (2025-2033), fueled by factors such as increasing adoption in remote patient monitoring programs, the growing geriatric population, and technological advancements enabling enhanced data analysis and integration. The market penetration is currently at xx% in developed markets and is expected to reach xx% by 2033. Technological disruptions, such as the integration of AI and machine learning for improved diagnostic accuracy, are reshaping consumer behavior and driving demand for more sophisticated devices.
Dominant Regions, Countries, or Segments in Handheld Vital Signs Monitor
North America currently holds the largest market share in the handheld vital signs monitor market, driven by factors such as high healthcare expenditure, advanced healthcare infrastructure, and early adoption of new technologies. However, regions like Asia-Pacific are experiencing rapid growth due to rising healthcare awareness, increasing disposable incomes, and government initiatives to improve healthcare access. The hospital segment is the leading end-user, owing to its high volume of patient monitoring needs.
- Key Drivers in North America: High healthcare spending, advanced medical infrastructure, strong regulatory support.
- Key Drivers in Asia-Pacific: Growing healthcare awareness, rising disposable incomes, government initiatives.
- Leading Segment: Hospital segment due to high volume of patient monitoring.
Handheld Vital Signs Monitor Product Landscape
Handheld vital signs monitors have evolved significantly, offering improved accuracy, portability, and integrated functionalities. Modern devices often incorporate wireless connectivity for seamless data transfer, advanced algorithms for improved diagnostic capabilities, and user-friendly interfaces for ease of operation. Key features include the ability to measure multiple vital signs (blood pressure, pulse oximetry, temperature, ECG), data storage and analysis capabilities, and compatibility with EMR systems. Unique selling propositions often center around advanced features such as early warning systems for critical changes in vital signs, improved battery life, and compact design for better portability.
Key Drivers, Barriers & Challenges in Handheld Vital Signs Monitor
Key Drivers:
- Increasing prevalence of chronic diseases.
- Growing demand for point-of-care diagnostics.
- Technological advancements, including wireless connectivity and AI integration.
- Rising healthcare expenditure in developing economies.
Challenges:
- High initial cost of devices limiting adoption in resource-constrained settings.
- Stringent regulatory requirements impacting product development and market entry.
- Competition from alternative monitoring technologies.
- Supply chain disruptions affecting device availability and pricing.
Emerging Opportunities in Handheld Vital Signs Monitor
- Expansion into untapped markets in developing countries.
- Development of specialized devices for niche applications (e.g., pediatric care, veterinary medicine).
- Integration of telehealth platforms for remote patient monitoring.
- Incorporation of advanced analytics and predictive capabilities.
Growth Accelerators in the Handheld Vital Signs Monitor Industry
Strategic partnerships between device manufacturers and healthcare providers are driving market growth, particularly in telehealth and remote patient monitoring initiatives. The integration of AI and machine learning is expected to significantly enhance diagnostic accuracy and improve treatment outcomes. Continuous technological advancements in device design and functionality will lead to greater adoption across various healthcare settings. Expansion into new markets, particularly in developing economies, presents substantial growth opportunities.
Key Players Shaping the Handheld Vital Signs Monitor Market
- Philips
- General Electric
- Hill-Rom
- Spacelabs Healthcare
- Nihon Kohden Corporation
- CAS Medical Systems
- Smiths Group plc
- Infinium Medical
- Mindray Medical International Limited
- Biolight
- Creative Medical
- Contec Medical Systems
- aXcent Medical
- Comen
Notable Milestones in Handheld Vital Signs Monitor Sector
- 2021: Philips launched a new handheld vital signs monitor with integrated telehealth capabilities.
- 2022: Mindray Medical received FDA approval for its next-generation handheld device with improved accuracy.
- 2023: A major merger between two smaller companies expanded the market's product portfolio.
In-Depth Handheld Vital Signs Monitor Market Outlook
The handheld vital signs monitor market is poised for continued growth, driven by technological advancements, increasing healthcare expenditure, and rising demand for point-of-care diagnostics. Strategic partnerships, expansion into emerging markets, and the integration of AI and machine learning present significant opportunities for market players. The focus on improving device portability, accuracy, and connectivity will shape the future of this dynamic market. The long-term market outlook remains positive, with substantial growth potential across various segments and geographical regions.
Handheld Vital Signs Monitor Segmentation
-
1. Application
- 1.1. Hospital
- 1.2. Clinic
- 1.3. Others
-
2. Type
- 2.1. Traditional Monitor
- 2.2. Microwave Monitor
Handheld Vital Signs Monitor Segmentation By Geography
-
1. North America
- 1.1. United States
- 1.2. Canada
- 1.3. Mexico
-
2. South America
- 2.1. Brazil
- 2.2. Argentina
- 2.3. Rest of South America
-
3. Europe
- 3.1. United Kingdom
- 3.2. Germany
- 3.3. France
- 3.4. Italy
- 3.5. Spain
- 3.6. Russia
- 3.7. Benelux
- 3.8. Nordics
- 3.9. Rest of Europe
-
4. Middle East & Africa
- 4.1. Turkey
- 4.2. Israel
- 4.3. GCC
- 4.4. North Africa
- 4.5. South Africa
- 4.6. Rest of Middle East & Africa
-
5. Asia Pacific
- 5.1. China
- 5.2. India
- 5.3. Japan
- 5.4. South Korea
- 5.5. ASEAN
- 5.6. Oceania
- 5.7. Rest of Asia Pacific
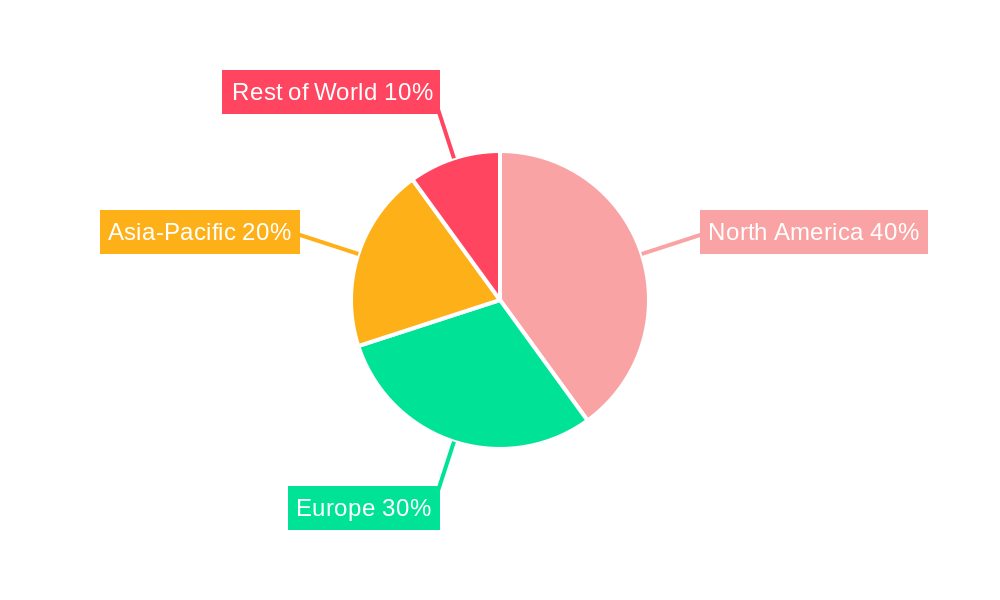
Handheld Vital Signs Monitor Regional Market Share

Geographic Coverage of Handheld Vital Signs Monitor
Handheld Vital Signs Monitor REPORT HIGHLIGHTS
| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 8.5% from 2020-2034 |
| Segmentation |
|
Table of Contents
- 1. Introduction
- 1.1. Research Scope
- 1.2. Market Segmentation
- 1.3. Research Methodology
- 1.4. Definitions and Assumptions
- 2. Executive Summary
- 2.1. Introduction
- 3. Market Dynamics
- 3.1. Introduction
- 3.2. Market Drivers
- 3.3. Market Restrains
- 3.4. Market Trends
- 4. Market Factor Analysis
- 4.1. Porters Five Forces
- 4.2. Supply/Value Chain
- 4.3. PESTEL analysis
- 4.4. Market Entropy
- 4.5. Patent/Trademark Analysis
- 5. Global Handheld Vital Signs Monitor Analysis, Insights and Forecast, 2020-2032
- 5.1. Market Analysis, Insights and Forecast - by Application
- 5.1.1. Hospital
- 5.1.2. Clinic
- 5.1.3. Others
- 5.2. Market Analysis, Insights and Forecast - by Type
- 5.2.1. Traditional Monitor
- 5.2.2. Microwave Monitor
- 5.3. Market Analysis, Insights and Forecast - by Region
- 5.3.1. North America
- 5.3.2. South America
- 5.3.3. Europe
- 5.3.4. Middle East & Africa
- 5.3.5. Asia Pacific
- 5.1. Market Analysis, Insights and Forecast - by Application
- 6. North America Handheld Vital Signs Monitor Analysis, Insights and Forecast, 2020-2032
- 6.1. Market Analysis, Insights and Forecast - by Application
- 6.1.1. Hospital
- 6.1.2. Clinic
- 6.1.3. Others
- 6.2. Market Analysis, Insights and Forecast - by Type
- 6.2.1. Traditional Monitor
- 6.2.2. Microwave Monitor
- 6.1. Market Analysis, Insights and Forecast - by Application
- 7. South America Handheld Vital Signs Monitor Analysis, Insights and Forecast, 2020-2032
- 7.1. Market Analysis, Insights and Forecast - by Application
- 7.1.1. Hospital
- 7.1.2. Clinic
- 7.1.3. Others
- 7.2. Market Analysis, Insights and Forecast - by Type
- 7.2.1. Traditional Monitor
- 7.2.2. Microwave Monitor
- 7.1. Market Analysis, Insights and Forecast - by Application
- 8. Europe Handheld Vital Signs Monitor Analysis, Insights and Forecast, 2020-2032
- 8.1. Market Analysis, Insights and Forecast - by Application
- 8.1.1. Hospital
- 8.1.2. Clinic
- 8.1.3. Others
- 8.2. Market Analysis, Insights and Forecast - by Type
- 8.2.1. Traditional Monitor
- 8.2.2. Microwave Monitor
- 8.1. Market Analysis, Insights and Forecast - by Application
- 9. Middle East & Africa Handheld Vital Signs Monitor Analysis, Insights and Forecast, 2020-2032
- 9.1. Market Analysis, Insights and Forecast - by Application
- 9.1.1. Hospital
- 9.1.2. Clinic
- 9.1.3. Others
- 9.2. Market Analysis, Insights and Forecast - by Type
- 9.2.1. Traditional Monitor
- 9.2.2. Microwave Monitor
- 9.1. Market Analysis, Insights and Forecast - by Application
- 10. Asia Pacific Handheld Vital Signs Monitor Analysis, Insights and Forecast, 2020-2032
- 10.1. Market Analysis, Insights and Forecast - by Application
- 10.1.1. Hospital
- 10.1.2. Clinic
- 10.1.3. Others
- 10.2. Market Analysis, Insights and Forecast - by Type
- 10.2.1. Traditional Monitor
- 10.2.2. Microwave Monitor
- 10.1. Market Analysis, Insights and Forecast - by Application
- 11. Competitive Analysis
- 11.1. Global Market Share Analysis 2025
- 11.2. Company Profiles
- 11.2.1 Philips
- 11.2.1.1. Overview
- 11.2.1.2. Products
- 11.2.1.3. SWOT Analysis
- 11.2.1.4. Recent Developments
- 11.2.1.5. Financials (Based on Availability)
- 11.2.2 General Electric
- 11.2.2.1. Overview
- 11.2.2.2. Products
- 11.2.2.3. SWOT Analysis
- 11.2.2.4. Recent Developments
- 11.2.2.5. Financials (Based on Availability)
- 11.2.3 Hill-Rom
- 11.2.3.1. Overview
- 11.2.3.2. Products
- 11.2.3.3. SWOT Analysis
- 11.2.3.4. Recent Developments
- 11.2.3.5. Financials (Based on Availability)
- 11.2.4 Spacelabs Healthcare
- 11.2.4.1. Overview
- 11.2.4.2. Products
- 11.2.4.3. SWOT Analysis
- 11.2.4.4. Recent Developments
- 11.2.4.5. Financials (Based on Availability)
- 11.2.5 Nihon Kohden Corporation
- 11.2.5.1. Overview
- 11.2.5.2. Products
- 11.2.5.3. SWOT Analysis
- 11.2.5.4. Recent Developments
- 11.2.5.5. Financials (Based on Availability)
- 11.2.6 CAS Medical Systems
- 11.2.6.1. Overview
- 11.2.6.2. Products
- 11.2.6.3. SWOT Analysis
- 11.2.6.4. Recent Developments
- 11.2.6.5. Financials (Based on Availability)
- 11.2.7 Smiths Group plc
- 11.2.7.1. Overview
- 11.2.7.2. Products
- 11.2.7.3. SWOT Analysis
- 11.2.7.4. Recent Developments
- 11.2.7.5. Financials (Based on Availability)
- 11.2.8 Infinium Medical
- 11.2.8.1. Overview
- 11.2.8.2. Products
- 11.2.8.3. SWOT Analysis
- 11.2.8.4. Recent Developments
- 11.2.8.5. Financials (Based on Availability)
- 11.2.9 Mindray Medical International Limited
- 11.2.9.1. Overview
- 11.2.9.2. Products
- 11.2.9.3. SWOT Analysis
- 11.2.9.4. Recent Developments
- 11.2.9.5. Financials (Based on Availability)
- 11.2.10 Biolight
- 11.2.10.1. Overview
- 11.2.10.2. Products
- 11.2.10.3. SWOT Analysis
- 11.2.10.4. Recent Developments
- 11.2.10.5. Financials (Based on Availability)
- 11.2.11 Creative Medical
- 11.2.11.1. Overview
- 11.2.11.2. Products
- 11.2.11.3. SWOT Analysis
- 11.2.11.4. Recent Developments
- 11.2.11.5. Financials (Based on Availability)
- 11.2.12 ContecMedical Systems
- 11.2.12.1. Overview
- 11.2.12.2. Products
- 11.2.12.3. SWOT Analysis
- 11.2.12.4. Recent Developments
- 11.2.12.5. Financials (Based on Availability)
- 11.2.13 aXcent Medical
- 11.2.13.1. Overview
- 11.2.13.2. Products
- 11.2.13.3. SWOT Analysis
- 11.2.13.4. Recent Developments
- 11.2.13.5. Financials (Based on Availability)
- 11.2.14 Comen
- 11.2.14.1. Overview
- 11.2.14.2. Products
- 11.2.14.3. SWOT Analysis
- 11.2.14.4. Recent Developments
- 11.2.14.5. Financials (Based on Availability)
- 11.2.1 Philips
List of Figures
- Figure 1: Global Handheld Vital Signs Monitor Revenue Breakdown (undefined, %) by Region 2025 & 2033
- Figure 2: North America Handheld Vital Signs Monitor Revenue (undefined), by Application 2025 & 2033
- Figure 3: North America Handheld Vital Signs Monitor Revenue Share (%), by Application 2025 & 2033
- Figure 4: North America Handheld Vital Signs Monitor Revenue (undefined), by Type 2025 & 2033
- Figure 5: North America Handheld Vital Signs Monitor Revenue Share (%), by Type 2025 & 2033
- Figure 6: North America Handheld Vital Signs Monitor Revenue (undefined), by Country 2025 & 2033
- Figure 7: North America Handheld Vital Signs Monitor Revenue Share (%), by Country 2025 & 2033
- Figure 8: South America Handheld Vital Signs Monitor Revenue (undefined), by Application 2025 & 2033
- Figure 9: South America Handheld Vital Signs Monitor Revenue Share (%), by Application 2025 & 2033
- Figure 10: South America Handheld Vital Signs Monitor Revenue (undefined), by Type 2025 & 2033
- Figure 11: South America Handheld Vital Signs Monitor Revenue Share (%), by Type 2025 & 2033
- Figure 12: South America Handheld Vital Signs Monitor Revenue (undefined), by Country 2025 & 2033
- Figure 13: South America Handheld Vital Signs Monitor Revenue Share (%), by Country 2025 & 2033
- Figure 14: Europe Handheld Vital Signs Monitor Revenue (undefined), by Application 2025 & 2033
- Figure 15: Europe Handheld Vital Signs Monitor Revenue Share (%), by Application 2025 & 2033
- Figure 16: Europe Handheld Vital Signs Monitor Revenue (undefined), by Type 2025 & 2033
- Figure 17: Europe Handheld Vital Signs Monitor Revenue Share (%), by Type 2025 & 2033
- Figure 18: Europe Handheld Vital Signs Monitor Revenue (undefined), by Country 2025 & 2033
- Figure 19: Europe Handheld Vital Signs Monitor Revenue Share (%), by Country 2025 & 2033
- Figure 20: Middle East & Africa Handheld Vital Signs Monitor Revenue (undefined), by Application 2025 & 2033
- Figure 21: Middle East & Africa Handheld Vital Signs Monitor Revenue Share (%), by Application 2025 & 2033
- Figure 22: Middle East & Africa Handheld Vital Signs Monitor Revenue (undefined), by Type 2025 & 2033
- Figure 23: Middle East & Africa Handheld Vital Signs Monitor Revenue Share (%), by Type 2025 & 2033
- Figure 24: Middle East & Africa Handheld Vital Signs Monitor Revenue (undefined), by Country 2025 & 2033
- Figure 25: Middle East & Africa Handheld Vital Signs Monitor Revenue Share (%), by Country 2025 & 2033
- Figure 26: Asia Pacific Handheld Vital Signs Monitor Revenue (undefined), by Application 2025 & 2033
- Figure 27: Asia Pacific Handheld Vital Signs Monitor Revenue Share (%), by Application 2025 & 2033
- Figure 28: Asia Pacific Handheld Vital Signs Monitor Revenue (undefined), by Type 2025 & 2033
- Figure 29: Asia Pacific Handheld Vital Signs Monitor Revenue Share (%), by Type 2025 & 2033
- Figure 30: Asia Pacific Handheld Vital Signs Monitor Revenue (undefined), by Country 2025 & 2033
- Figure 31: Asia Pacific Handheld Vital Signs Monitor Revenue Share (%), by Country 2025 & 2033
List of Tables
- Table 1: Global Handheld Vital Signs Monitor Revenue undefined Forecast, by Application 2020 & 2033
- Table 2: Global Handheld Vital Signs Monitor Revenue undefined Forecast, by Type 2020 & 2033
- Table 3: Global Handheld Vital Signs Monitor Revenue undefined Forecast, by Region 2020 & 2033
- Table 4: Global Handheld Vital Signs Monitor Revenue undefined Forecast, by Application 2020 & 2033
- Table 5: Global Handheld Vital Signs Monitor Revenue undefined Forecast, by Type 2020 & 2033
- Table 6: Global Handheld Vital Signs Monitor Revenue undefined Forecast, by Country 2020 & 2033
- Table 7: United States Handheld Vital Signs Monitor Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 8: Canada Handheld Vital Signs Monitor Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 9: Mexico Handheld Vital Signs Monitor Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 10: Global Handheld Vital Signs Monitor Revenue undefined Forecast, by Application 2020 & 2033
- Table 11: Global Handheld Vital Signs Monitor Revenue undefined Forecast, by Type 2020 & 2033
- Table 12: Global Handheld Vital Signs Monitor Revenue undefined Forecast, by Country 2020 & 2033
- Table 13: Brazil Handheld Vital Signs Monitor Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 14: Argentina Handheld Vital Signs Monitor Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 15: Rest of South America Handheld Vital Signs Monitor Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 16: Global Handheld Vital Signs Monitor Revenue undefined Forecast, by Application 2020 & 2033
- Table 17: Global Handheld Vital Signs Monitor Revenue undefined Forecast, by Type 2020 & 2033
- Table 18: Global Handheld Vital Signs Monitor Revenue undefined Forecast, by Country 2020 & 2033
- Table 19: United Kingdom Handheld Vital Signs Monitor Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 20: Germany Handheld Vital Signs Monitor Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 21: France Handheld Vital Signs Monitor Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 22: Italy Handheld Vital Signs Monitor Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 23: Spain Handheld Vital Signs Monitor Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 24: Russia Handheld Vital Signs Monitor Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 25: Benelux Handheld Vital Signs Monitor Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 26: Nordics Handheld Vital Signs Monitor Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 27: Rest of Europe Handheld Vital Signs Monitor Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 28: Global Handheld Vital Signs Monitor Revenue undefined Forecast, by Application 2020 & 2033
- Table 29: Global Handheld Vital Signs Monitor Revenue undefined Forecast, by Type 2020 & 2033
- Table 30: Global Handheld Vital Signs Monitor Revenue undefined Forecast, by Country 2020 & 2033
- Table 31: Turkey Handheld Vital Signs Monitor Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 32: Israel Handheld Vital Signs Monitor Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 33: GCC Handheld Vital Signs Monitor Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 34: North Africa Handheld Vital Signs Monitor Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 35: South Africa Handheld Vital Signs Monitor Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 36: Rest of Middle East & Africa Handheld Vital Signs Monitor Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 37: Global Handheld Vital Signs Monitor Revenue undefined Forecast, by Application 2020 & 2033
- Table 38: Global Handheld Vital Signs Monitor Revenue undefined Forecast, by Type 2020 & 2033
- Table 39: Global Handheld Vital Signs Monitor Revenue undefined Forecast, by Country 2020 & 2033
- Table 40: China Handheld Vital Signs Monitor Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 41: India Handheld Vital Signs Monitor Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 42: Japan Handheld Vital Signs Monitor Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 43: South Korea Handheld Vital Signs Monitor Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 44: ASEAN Handheld Vital Signs Monitor Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 45: Oceania Handheld Vital Signs Monitor Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 46: Rest of Asia Pacific Handheld Vital Signs Monitor Revenue (undefined) Forecast, by Application 2020 & 2033
Frequently Asked Questions
1. What is the projected Compound Annual Growth Rate (CAGR) of the Handheld Vital Signs Monitor?
The projected CAGR is approximately 8.5%.
2. Which companies are prominent players in the Handheld Vital Signs Monitor?
Key companies in the market include Philips, General Electric, Hill-Rom, Spacelabs Healthcare, Nihon Kohden Corporation, CAS Medical Systems, Smiths Group plc, Infinium Medical, Mindray Medical International Limited, Biolight, Creative Medical, ContecMedical Systems, aXcent Medical, Comen.
3. What are the main segments of the Handheld Vital Signs Monitor?
The market segments include Application, Type.
4. Can you provide details about the market size?
The market size is estimated to be USD XXX N/A as of 2022.
5. What are some drivers contributing to market growth?
N/A
6. What are the notable trends driving market growth?
N/A
7. Are there any restraints impacting market growth?
N/A
8. Can you provide examples of recent developments in the market?
N/A
9. What pricing options are available for accessing the report?
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 2900.00, USD 4350.00, and USD 5800.00 respectively.
10. Is the market size provided in terms of value or volume?
The market size is provided in terms of value, measured in N/A.
11. Are there any specific market keywords associated with the report?
Yes, the market keyword associated with the report is "Handheld Vital Signs Monitor," which aids in identifying and referencing the specific market segment covered.
12. How do I determine which pricing option suits my needs best?
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
13. Are there any additional resources or data provided in the Handheld Vital Signs Monitor report?
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
14. How can I stay updated on further developments or reports in the Handheld Vital Signs Monitor?
To stay informed about further developments, trends, and reports in the Handheld Vital Signs Monitor, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.
Methodology
Step 1 - Identification of Relevant Samples Size from Population Database
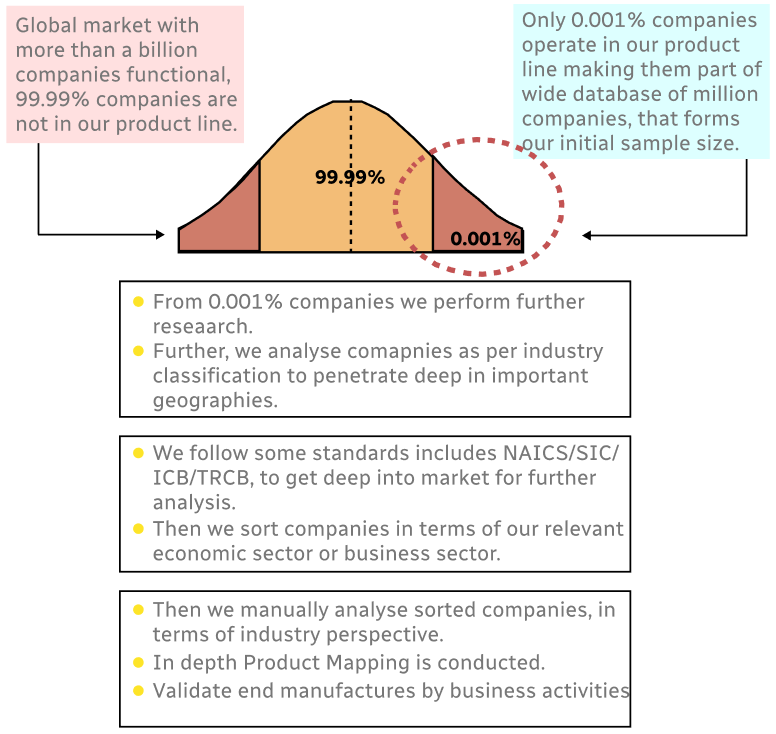
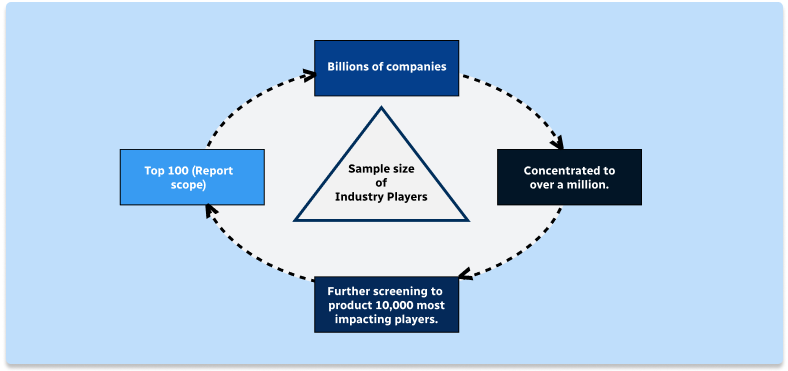
Step 2 - Approaches for Defining Global Market Size (Value, Volume* & Price*)
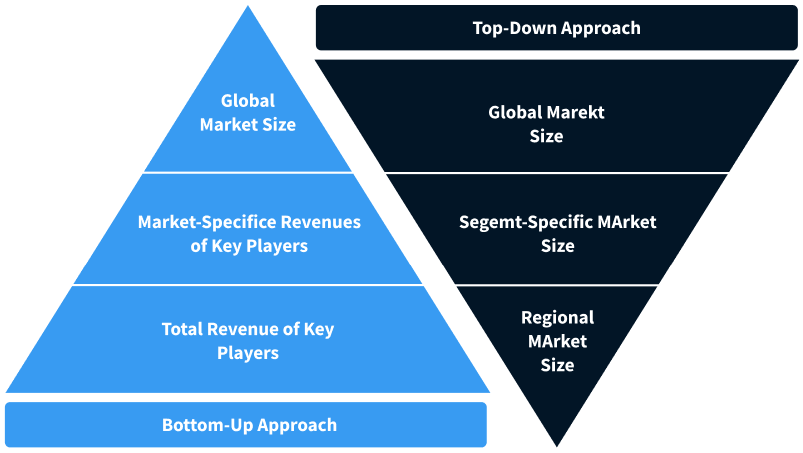
Note*: In applicable scenarios
Step 3 - Data Sources
Primary Research
- Web Analytics
- Survey Reports
- Research Institute
- Latest Research Reports
- Opinion Leaders
Secondary Research
- Annual Reports
- White Paper
- Latest Press Release
- Industry Association
- Paid Database
- Investor Presentations
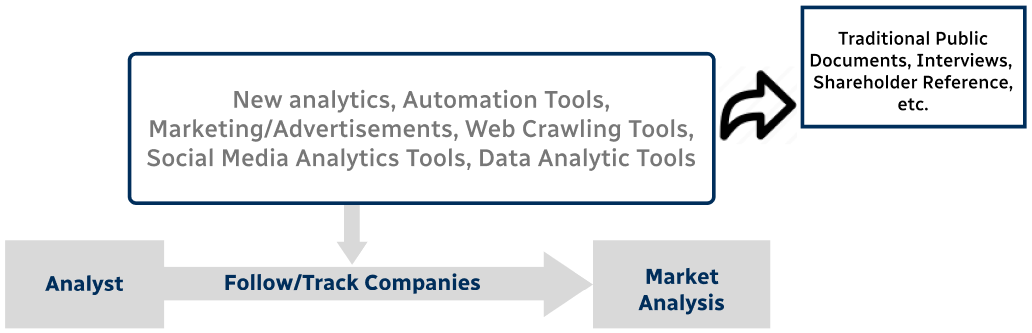
Step 4 - Data Triangulation
Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence


