Key Insights
The global market for breast cancer generic drugs is experiencing robust growth, driven by increasing breast cancer incidence rates worldwide, a growing aging population, and the rising affordability of generic medications compared to their branded counterparts. The market's expansion is further fueled by ongoing research and development efforts leading to the development of more effective and targeted therapies, although challenges persist such as patent expirations on key drugs impacting revenue for established players, and the complexities of navigating stringent regulatory requirements in different regions. We estimate the 2025 market size to be approximately $15 billion, based on analysis of similar pharmaceutical markets and considering a plausible CAGR of 8% as a conservative estimation given industry growth trends. This signifies a substantial opportunity for generic drug manufacturers, attracting both established players like Teva, Mylan, and Dr. Reddy's Laboratories, and emerging companies aiming to capture market share. The market is segmented by drug type (e.g., aromatase inhibitors, alkylating agents, taxanes), route of administration, and geographic region, with North America and Europe currently holding the largest market shares.
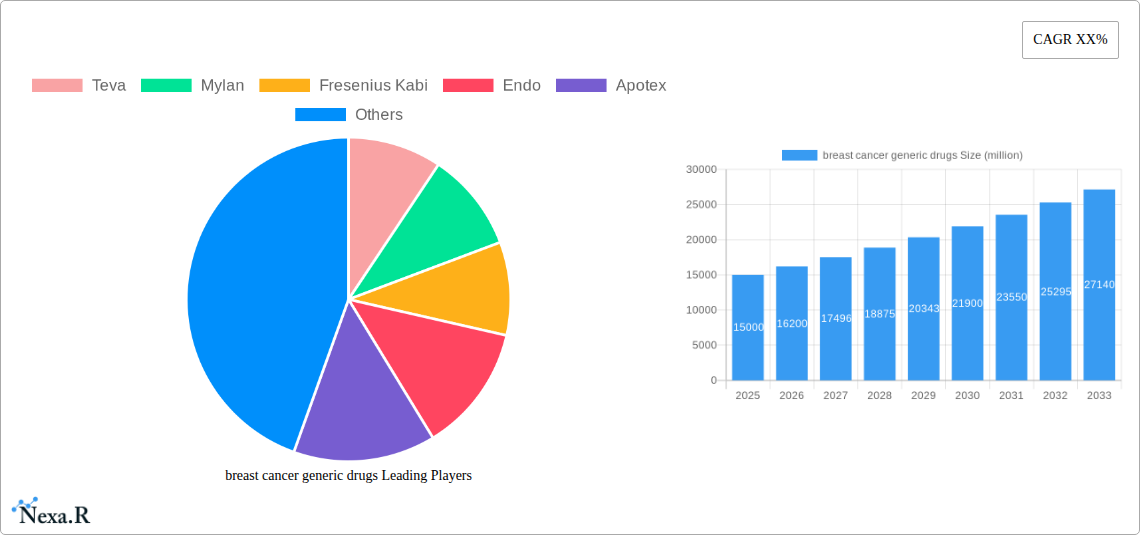
breast cancer generic drugs Market Size (In Billion)

Competitive dynamics are intense, with established players leveraging their extensive distribution networks and brand recognition to maintain market dominance. However, newer entrants are focusing on niche segments and innovative pricing strategies to compete effectively. The future landscape will likely be shaped by factors such as the emergence of biosimilars (generic versions of biologics), ongoing price pressures, and the increasing focus on personalized medicine approaches in breast cancer treatment. Successful players will need to demonstrate robust manufacturing capabilities, efficient supply chains, and a keen understanding of regulatory landscapes to thrive in this evolving market. This will require significant investments in research and development, especially in emerging markets with high growth potential, and strategies focused on ensuring reliable access to affordable medication.
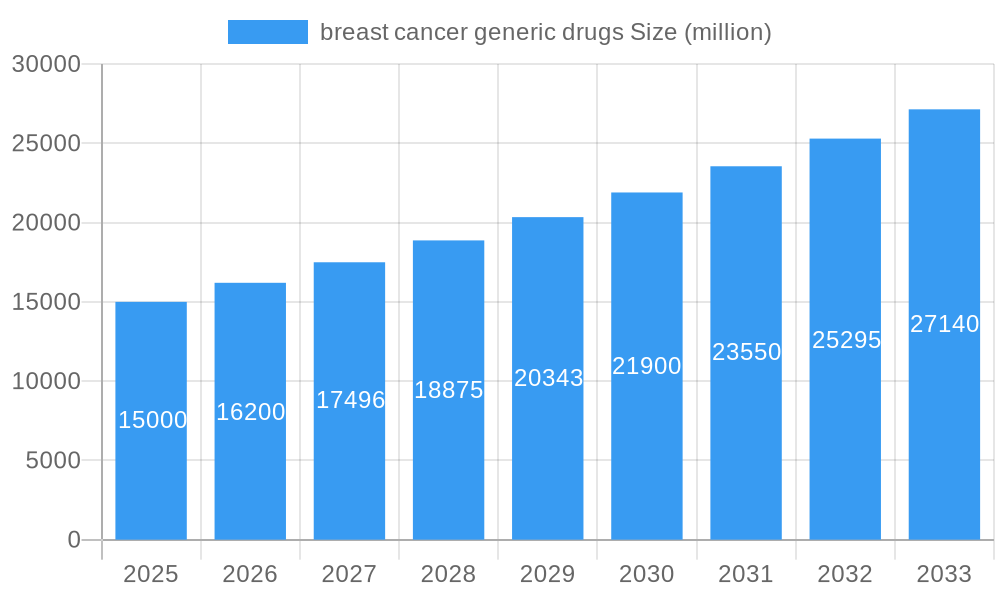
breast cancer generic drugs Company Market Share

Breast Cancer Generic Drugs Market Report: 2019-2033
This comprehensive report provides a detailed analysis of the global breast cancer generic drugs market, covering the period 2019-2033. The report leverages extensive data analysis and industry expertise to offer invaluable insights for pharmaceutical companies, investors, and healthcare professionals. We delve into market dynamics, growth trends, regional performance, and the competitive landscape, providing a robust foundation for strategic decision-making. The parent market is oncology drugs, while the child market is breast cancer therapeutics. Market size is presented in million units.
Breast Cancer Generic Drugs Market Dynamics & Structure
The breast cancer generic drugs market is characterized by high competition, driven by a large patient population and increasing demand for cost-effective treatment options. Market concentration is moderate, with several major players holding significant market share, but numerous smaller companies also vying for a position. Technological innovation is a key driver, focusing on improved drug delivery systems and enhanced efficacy. Regulatory frameworks, particularly bioequivalence requirements and pricing regulations, significantly impact market access and growth. The market faces competition from novel targeted therapies and immunotherapies, along with the constant introduction of new generic entrants. M&A activity within the sector has been relatively high, with numerous smaller companies being acquired by larger players for market consolidation and expansion.
- Market Concentration: Moderately concentrated, with top 5 players accounting for approximately 60% of the market share in 2024 (xx million units).
- Technological Innovation: Focus on improved formulations and delivery mechanisms (e.g., extended-release formulations).
- Regulatory Landscape: Stringent bioequivalence requirements and pricing pressures influence market entry and pricing strategies.
- Competitive Substitutes: Biosimilars, targeted therapies, and immunotherapies pose competition.
- End-User Demographics: Primarily oncology clinics, hospitals, and community-based healthcare settings.
- M&A Trends: A significant number of mergers and acquisitions (xx deals) were observed between 2019-2024, primarily driven by market consolidation and expansion into emerging markets.
Breast Cancer Generic Drugs Growth Trends & Insights
The global breast cancer generic drugs market experienced robust growth during the historical period (2019-2024), fueled by a rising prevalence of breast cancer, increasing affordability of generic drugs, and expanding access to healthcare in emerging economies. The market size in 2024 is estimated at xx million units, exhibiting a CAGR of xx% from 2019. Adoption rates are steadily increasing, driven by favorable reimbursement policies and a rising awareness of generic drug efficacy and safety. Technological advancements are accelerating market growth through the introduction of improved formulations and delivery systems, while changing consumer preferences toward cost-effective treatment options further drive market expansion. Market penetration remains high within developed economies, while significant growth potential exists in emerging markets.
Dominant Regions, Countries, or Segments in Breast Cancer Generic Drugs
North America currently dominates the breast cancer generic drugs market, driven by high healthcare expenditure, a large patient population, and well-established healthcare infrastructure. However, Asia-Pacific exhibits the fastest growth rate due to rapidly expanding healthcare systems, increasing awareness of breast cancer, and growing affordability of generic medications.
- Key Drivers (North America): High healthcare expenditure, advanced healthcare infrastructure, established regulatory frameworks.
- Key Drivers (Asia-Pacific): Rising disposable income, growing healthcare awareness, favorable government initiatives, expanding healthcare infrastructure, rising prevalence of breast cancer.
- Dominance Factors: High prevalence of breast cancer, higher healthcare expenditure, and regulatory support lead to significant market share in North America. Rapid economic development and growth in healthcare spending contribute to Asia-Pacific's rapid market growth.
- Growth Potential: Significant untapped potential in emerging markets in Asia-Pacific, Latin America, and Africa.
Breast Cancer Generic Drugs Product Landscape
The market encompasses a wide range of generic versions of established breast cancer drugs, including anthracyclines, taxanes, and aromatase inhibitors. Product innovations are focused on improving formulation stability, enhancing patient compliance through novel delivery systems (e.g., extended-release), and reducing side effects. These improvements lead to enhanced treatment outcomes and improved patient experience. Key performance indicators (KPIs) include bioequivalence to reference products, safety profiles, and patient adherence rates.
Key Drivers, Barriers & Challenges in Breast Cancer Generic Drugs
Key Drivers: Rising prevalence of breast cancer, increasing demand for cost-effective treatments, favorable regulatory policies promoting generic drug usage, and robust research and development leading to improved formulations.
Challenges: Intense competition among generic manufacturers, price erosion, bioequivalence challenges for complex formulations, and stringent regulatory requirements in various markets can lead to delays in market entry. Supply chain disruptions can create shortages and impact affordability.
Emerging Opportunities in Breast Cancer Generic Drugs
Emerging opportunities lie in expanding access to generic drugs in underserved populations, particularly in low- and middle-income countries. The development of innovative formulations, such as extended-release preparations and biosimilars of targeted therapies, presents significant potential for market growth.
Growth Accelerators in the Breast Cancer Generic Drugs Industry
Strategic partnerships between generic drug manufacturers and healthcare providers can accelerate market penetration and improve access to affordable treatments. Technological advancements, including improved manufacturing processes and advanced drug delivery systems, will further fuel market growth. Expanding into new geographical markets, particularly in emerging economies, presents significant potential for expansion.
Key Players Shaping the Breast Cancer Generic Drugs Market
- Teva
- Mylan
- Fresenius Kabi
- Endo
- Apotex
- Sun Pharma
- Hengrui
- Novartis
- Taro
- Arab Pharmaceutical
- Yiling Pharmaceutical
- Hikma Pharmaceuticals
- Dr. Reddy's Laboratories
- Natco Pharma
- Cipla
- Accord Healthcare
Notable Milestones in Breast Cancer Generic Drugs Sector
- 2020: Approval of several new generic formulations in key markets.
- 2021: Significant M&A activity among generic drug manufacturers.
- 2022: Launch of a new extended-release formulation of a widely used breast cancer drug.
- 2023: Increased focus on biosimilar development.
- 2024: Several key players announced investments in manufacturing capacities in emerging markets.
In-Depth Breast Cancer Generic Drugs Market Outlook
The future of the breast cancer generic drugs market looks promising, driven by the continuous rise in breast cancer prevalence and a growing demand for affordable treatment options. Ongoing technological innovations, coupled with strategic partnerships and market expansion initiatives, will fuel market growth in the forecast period (2025-2033). The market is expected to reach xx million units by 2033, reflecting substantial growth and solidifying its position as a significant sector within the pharmaceutical industry.
breast cancer generic drugs Segmentation
-
1. Application
- 1.1. Ductal Carcinoma of Breast
- 1.2. Invasive Ductal Carcinoma
- 1.3. Lobular Carcinoma
- 1.4. Triple Negative Breast Cancer
-
2. Types
- 2.1. Letrozole
- 2.2. Anastrazole
- 2.3. Exemestane
- 2.4. Epirubicine
- 2.5. Toremifene
- 2.6. Fulvestrant
- 2.7. Megestrol (Hospira)
breast cancer generic drugs Segmentation By Geography
- 1. CA
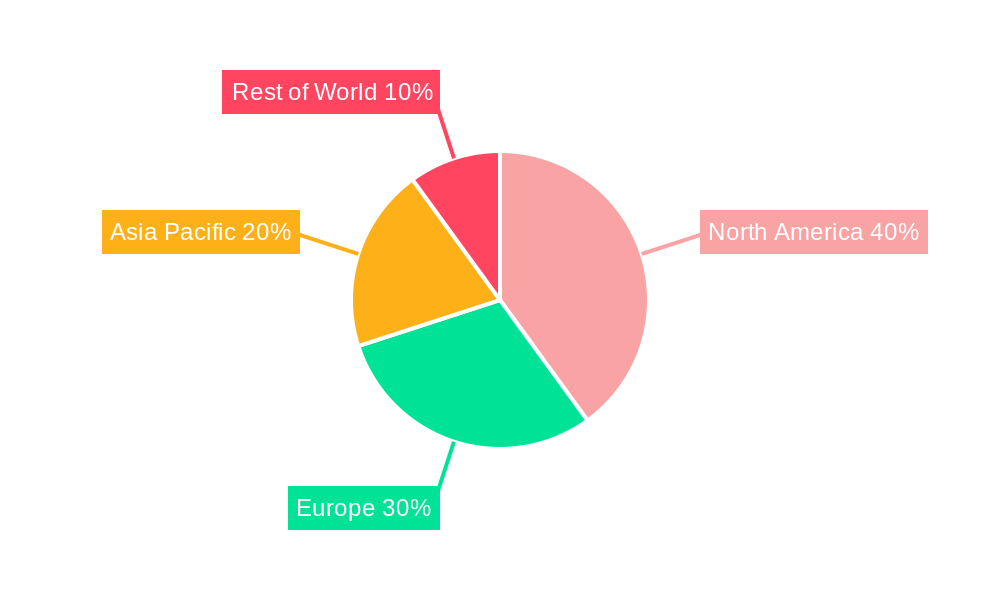
breast cancer generic drugs Regional Market Share

Geographic Coverage of breast cancer generic drugs
breast cancer generic drugs REPORT HIGHLIGHTS
| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of XX% from 2020-2034 |
| Segmentation |
|
Table of Contents
- 1. Introduction
- 1.1. Research Scope
- 1.2. Market Segmentation
- 1.3. Research Methodology
- 1.4. Definitions and Assumptions
- 2. Executive Summary
- 2.1. Introduction
- 3. Market Dynamics
- 3.1. Introduction
- 3.2. Market Drivers
- 3.3. Market Restrains
- 3.4. Market Trends
- 4. Market Factor Analysis
- 4.1. Porters Five Forces
- 4.2. Supply/Value Chain
- 4.3. PESTEL analysis
- 4.4. Market Entropy
- 4.5. Patent/Trademark Analysis
- 5. breast cancer generic drugs Analysis, Insights and Forecast, 2020-2032
- 5.1. Market Analysis, Insights and Forecast - by Application
- 5.1.1. Ductal Carcinoma of Breast
- 5.1.2. Invasive Ductal Carcinoma
- 5.1.3. Lobular Carcinoma
- 5.1.4. Triple Negative Breast Cancer
- 5.2. Market Analysis, Insights and Forecast - by Types
- 5.2.1. Letrozole
- 5.2.2. Anastrazole
- 5.2.3. Exemestane
- 5.2.4. Epirubicine
- 5.2.5. Toremifene
- 5.2.6. Fulvestrant
- 5.2.7. Megestrol (Hospira)
- 5.3. Market Analysis, Insights and Forecast - by Region
- 5.3.1. CA
- 5.1. Market Analysis, Insights and Forecast - by Application
- 6. Competitive Analysis
- 6.1. Market Share Analysis 2025
- 6.2. Company Profiles
- 6.2.1 Teva
- 6.2.1.1. Overview
- 6.2.1.2. Products
- 6.2.1.3. SWOT Analysis
- 6.2.1.4. Recent Developments
- 6.2.1.5. Financials (Based on Availability)
- 6.2.2 Mylan
- 6.2.2.1. Overview
- 6.2.2.2. Products
- 6.2.2.3. SWOT Analysis
- 6.2.2.4. Recent Developments
- 6.2.2.5. Financials (Based on Availability)
- 6.2.3 Fresenius Kabi
- 6.2.3.1. Overview
- 6.2.3.2. Products
- 6.2.3.3. SWOT Analysis
- 6.2.3.4. Recent Developments
- 6.2.3.5. Financials (Based on Availability)
- 6.2.4 Endo
- 6.2.4.1. Overview
- 6.2.4.2. Products
- 6.2.4.3. SWOT Analysis
- 6.2.4.4. Recent Developments
- 6.2.4.5. Financials (Based on Availability)
- 6.2.5 Apotex
- 6.2.5.1. Overview
- 6.2.5.2. Products
- 6.2.5.3. SWOT Analysis
- 6.2.5.4. Recent Developments
- 6.2.5.5. Financials (Based on Availability)
- 6.2.6 Sun Pharma
- 6.2.6.1. Overview
- 6.2.6.2. Products
- 6.2.6.3. SWOT Analysis
- 6.2.6.4. Recent Developments
- 6.2.6.5. Financials (Based on Availability)
- 6.2.7 Hengrui
- 6.2.7.1. Overview
- 6.2.7.2. Products
- 6.2.7.3. SWOT Analysis
- 6.2.7.4. Recent Developments
- 6.2.7.5. Financials (Based on Availability)
- 6.2.8 Novartis
- 6.2.8.1. Overview
- 6.2.8.2. Products
- 6.2.8.3. SWOT Analysis
- 6.2.8.4. Recent Developments
- 6.2.8.5. Financials (Based on Availability)
- 6.2.9 Taro
- 6.2.9.1. Overview
- 6.2.9.2. Products
- 6.2.9.3. SWOT Analysis
- 6.2.9.4. Recent Developments
- 6.2.9.5. Financials (Based on Availability)
- 6.2.10 Arab Pharmaceutical
- 6.2.10.1. Overview
- 6.2.10.2. Products
- 6.2.10.3. SWOT Analysis
- 6.2.10.4. Recent Developments
- 6.2.10.5. Financials (Based on Availability)
- 6.2.11 Yiling Pharmaceutical
- 6.2.11.1. Overview
- 6.2.11.2. Products
- 6.2.11.3. SWOT Analysis
- 6.2.11.4. Recent Developments
- 6.2.11.5. Financials (Based on Availability)
- 6.2.12 Hikma Pharmaceuticals
- 6.2.12.1. Overview
- 6.2.12.2. Products
- 6.2.12.3. SWOT Analysis
- 6.2.12.4. Recent Developments
- 6.2.12.5. Financials (Based on Availability)
- 6.2.13 Dr. Reddy's Laboratories
- 6.2.13.1. Overview
- 6.2.13.2. Products
- 6.2.13.3. SWOT Analysis
- 6.2.13.4. Recent Developments
- 6.2.13.5. Financials (Based on Availability)
- 6.2.14 Natco Pharma
- 6.2.14.1. Overview
- 6.2.14.2. Products
- 6.2.14.3. SWOT Analysis
- 6.2.14.4. Recent Developments
- 6.2.14.5. Financials (Based on Availability)
- 6.2.15 Cipla
- 6.2.15.1. Overview
- 6.2.15.2. Products
- 6.2.15.3. SWOT Analysis
- 6.2.15.4. Recent Developments
- 6.2.15.5. Financials (Based on Availability)
- 6.2.16 Accord Healthcare
- 6.2.16.1. Overview
- 6.2.16.2. Products
- 6.2.16.3. SWOT Analysis
- 6.2.16.4. Recent Developments
- 6.2.16.5. Financials (Based on Availability)
- 6.2.1 Teva
List of Figures
- Figure 1: breast cancer generic drugs Revenue Breakdown (million, %) by Product 2025 & 2033
- Figure 2: breast cancer generic drugs Share (%) by Company 2025
List of Tables
- Table 1: breast cancer generic drugs Revenue million Forecast, by Application 2020 & 2033
- Table 2: breast cancer generic drugs Revenue million Forecast, by Types 2020 & 2033
- Table 3: breast cancer generic drugs Revenue million Forecast, by Region 2020 & 2033
- Table 4: breast cancer generic drugs Revenue million Forecast, by Application 2020 & 2033
- Table 5: breast cancer generic drugs Revenue million Forecast, by Types 2020 & 2033
- Table 6: breast cancer generic drugs Revenue million Forecast, by Country 2020 & 2033
Frequently Asked Questions
1. What is the projected Compound Annual Growth Rate (CAGR) of the breast cancer generic drugs?
The projected CAGR is approximately XX%.
2. Which companies are prominent players in the breast cancer generic drugs?
Key companies in the market include Teva, Mylan, Fresenius Kabi, Endo, Apotex, Sun Pharma, Hengrui, Novartis, Taro, Arab Pharmaceutical, Yiling Pharmaceutical, Hikma Pharmaceuticals, Dr. Reddy's Laboratories, Natco Pharma, Cipla, Accord Healthcare.
3. What are the main segments of the breast cancer generic drugs?
The market segments include Application, Types.
4. Can you provide details about the market size?
The market size is estimated to be USD XXX million as of 2022.
5. What are some drivers contributing to market growth?
N/A
6. What are the notable trends driving market growth?
N/A
7. Are there any restraints impacting market growth?
N/A
8. Can you provide examples of recent developments in the market?
N/A
9. What pricing options are available for accessing the report?
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 3400.00, USD 5100.00, and USD 6800.00 respectively.
10. Is the market size provided in terms of value or volume?
The market size is provided in terms of value, measured in million.
11. Are there any specific market keywords associated with the report?
Yes, the market keyword associated with the report is "breast cancer generic drugs," which aids in identifying and referencing the specific market segment covered.
12. How do I determine which pricing option suits my needs best?
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
13. Are there any additional resources or data provided in the breast cancer generic drugs report?
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
14. How can I stay updated on further developments or reports in the breast cancer generic drugs?
To stay informed about further developments, trends, and reports in the breast cancer generic drugs, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.
Methodology
Step 1 - Identification of Relevant Samples Size from Population Database
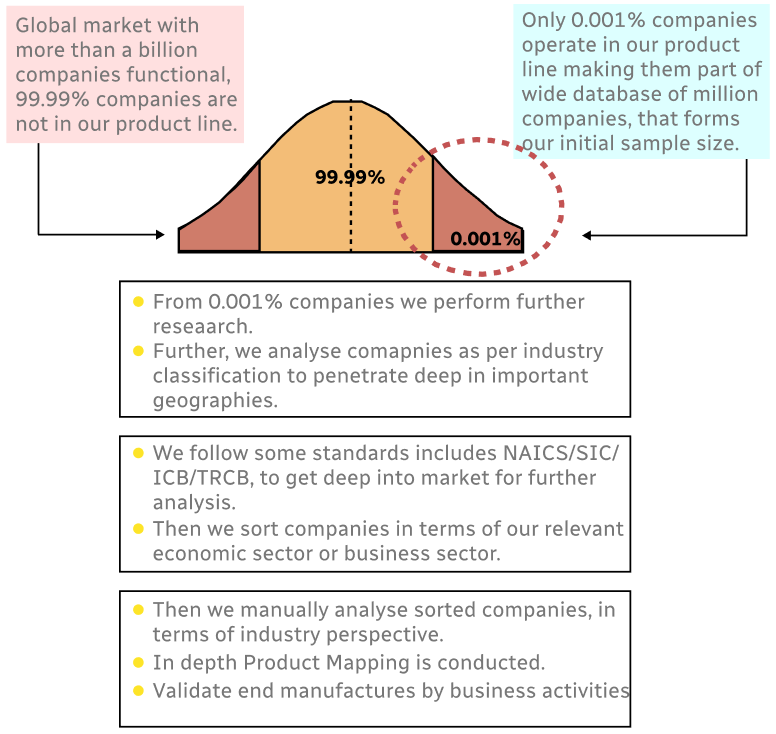
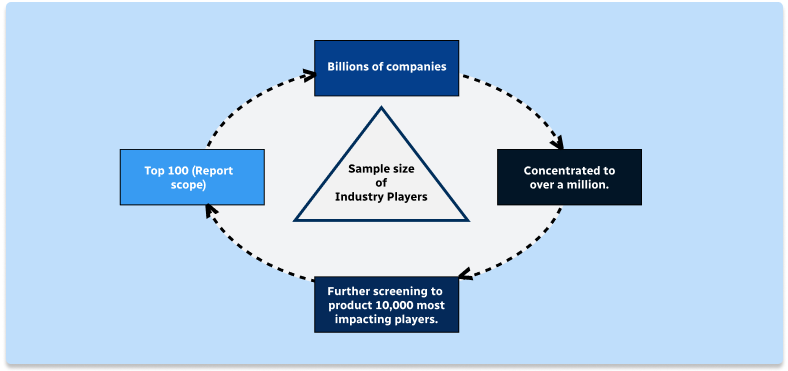
Step 2 - Approaches for Defining Global Market Size (Value, Volume* & Price*)
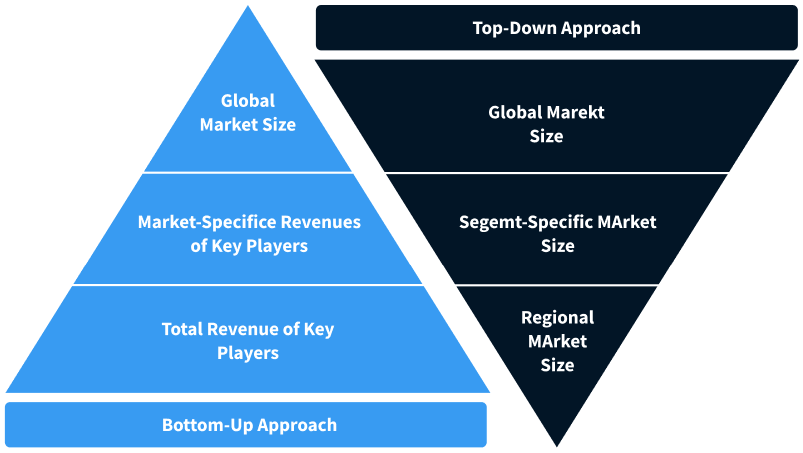
Note*: In applicable scenarios
Step 3 - Data Sources
Primary Research
- Web Analytics
- Survey Reports
- Research Institute
- Latest Research Reports
- Opinion Leaders
Secondary Research
- Annual Reports
- White Paper
- Latest Press Release
- Industry Association
- Paid Database
- Investor Presentations
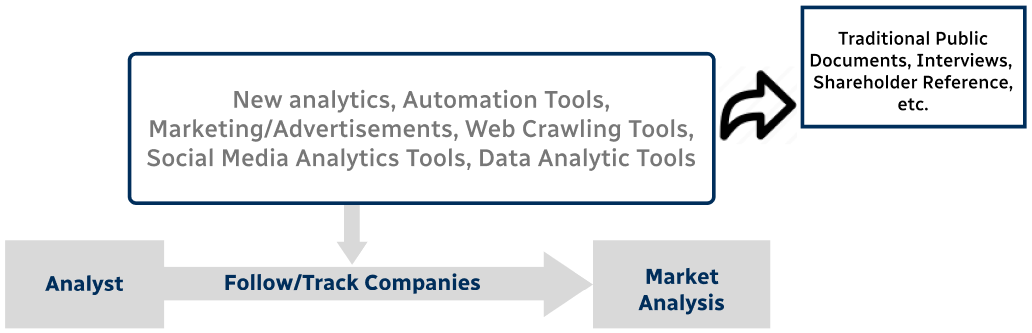
Step 4 - Data Triangulation
Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence


