Key Insights
The Alzheimer's Non-Pharmacological Treatment Device market is experiencing significant growth, driven by the increasing prevalence of Alzheimer's disease and a rising demand for effective non-pharmaceutical interventions. While pharmaceutical treatments remain a cornerstone of Alzheimer's management, the limitations of current drug therapies and the growing awareness of the benefits of non-invasive approaches have fueled considerable investment and innovation in this sector. The market's expansion is propelled by technological advancements leading to more sophisticated and user-friendly devices, improved diagnostic capabilities enabling earlier intervention, and a greater emphasis on holistic patient care. Key players like Magstim, NeuroConn, and Medtronic are at the forefront of this development, continually refining existing technologies and introducing novel solutions. However, high initial costs associated with some devices, along with challenges related to reimbursement policies and varying levels of patient compliance, pose challenges to market penetration. Further research and clinical trials are crucial to solidify the efficacy and cost-effectiveness of these treatments, making them more accessible to a wider patient population.
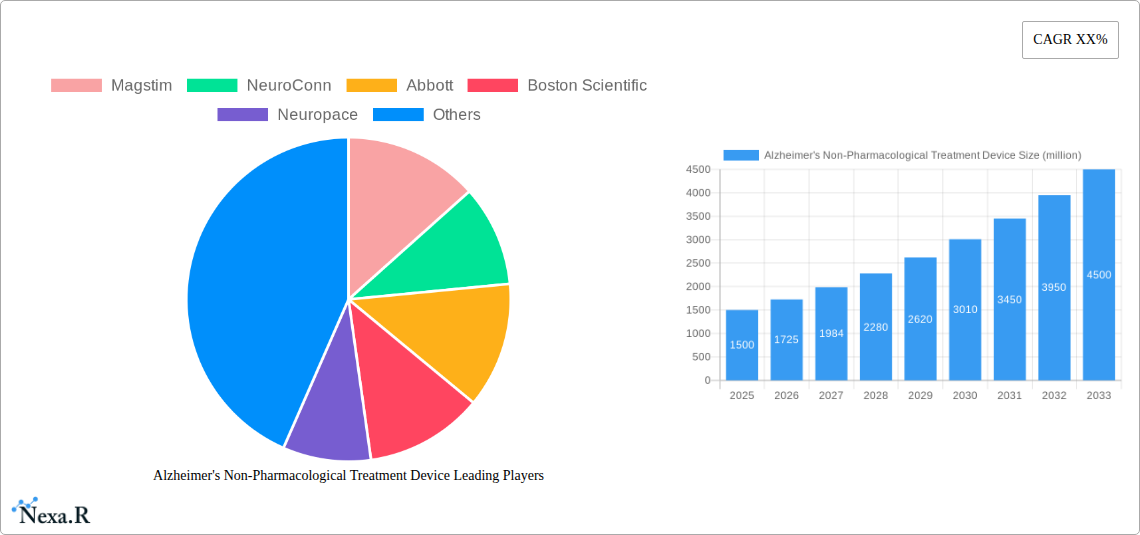
Alzheimer's Non-Pharmacological Treatment Device Market Size (In Billion)

This market is segmented by device type (e.g., tDCS, TMS, neurofeedback devices), application (e.g., cognitive enhancement, symptom management), and end-user (hospitals, clinics, home care). The North American region currently holds a substantial market share, reflecting advanced healthcare infrastructure and high adoption rates. However, growing awareness and rising healthcare expenditure in regions like Europe and Asia-Pacific are expected to fuel strong regional growth in the coming years. The market's future hinges on continued technological innovation, broader clinical validation, and favorable regulatory landscapes. A projected Compound Annual Growth Rate (CAGR) of approximately 15% from 2025 to 2033 indicates robust market expansion, underpinned by the escalating global burden of Alzheimer's and the increasing adoption of non-pharmaceutical interventions. We anticipate a market size of approximately $3 billion by 2033, with considerable potential for further expansion as research advances and patient access improves.
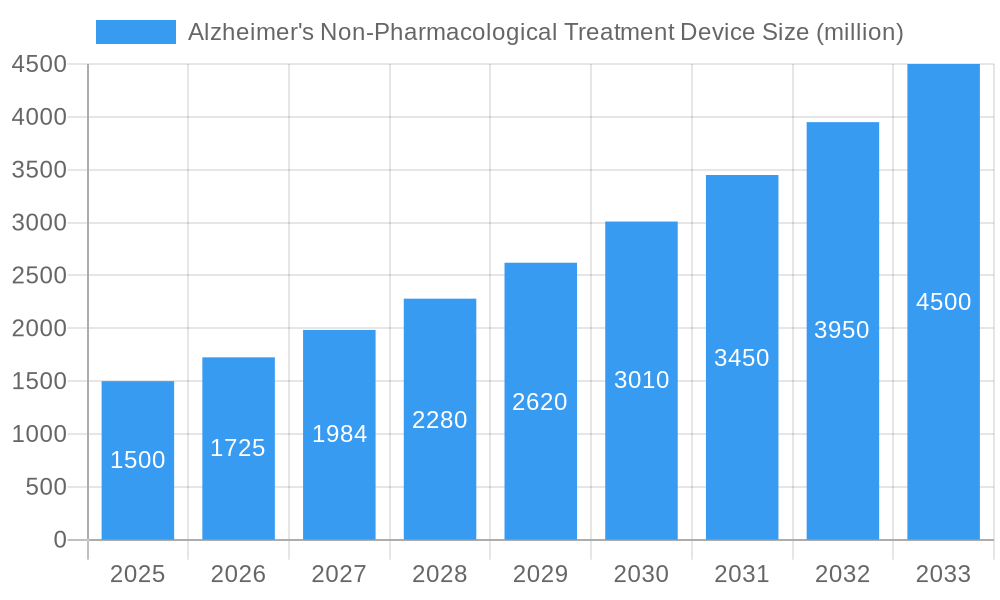
Alzheimer's Non-Pharmacological Treatment Device Company Market Share

Alzheimer's Non-Pharmacological Treatment Device Market Report: 2019-2033
This comprehensive report provides an in-depth analysis of the Alzheimer's Non-Pharmacological Treatment Device market, encompassing market dynamics, growth trends, regional analysis, product landscape, key players, and future outlook. The study period covers 2019-2033, with a base year of 2025 and a forecast period of 2025-2033. The report caters to industry professionals, investors, and researchers seeking a thorough understanding of this rapidly evolving sector. The parent market is the broader neurological device market, while the child market is specifically non-pharmacological treatments for Alzheimer's.
Alzheimer's Non-Pharmacological Treatment Device Market Dynamics & Structure
The Alzheimer's Non-Pharmacological Treatment Device market is characterized by moderate concentration, with key players like Medtronic, Boston Scientific, and Abbott holding significant market share (estimated at 60% collectively in 2025). Technological innovation, driven by advancements in neurostimulation and brain-computer interfaces, is a key growth driver. Regulatory frameworks, including FDA approvals and CE markings, significantly impact market access. Competitive substitutes include traditional therapies and emerging technologies like gene therapy, influencing market adoption rates. End-users primarily comprise hospitals, specialized clinics, and research institutions. M&A activity within the parent neurological device market has been robust in the historical period (2019-2024), with an estimated xx million USD worth of deals, although the direct impact on the child market is less significant, with approximately xx M&A deals directly impacting the non-pharmacological Alzheimer's treatment device sector during this period.
- Market Concentration: Moderately concentrated, with top 3 players holding ~60% market share in 2025.
- Technological Innovation: Advancements in neurostimulation, brain-computer interfaces, and AI-driven diagnostics.
- Regulatory Landscape: Stringent regulatory approvals (FDA, CE marking) impacting market entry and expansion.
- Competitive Substitutes: Traditional therapies, emerging gene therapies and other novel approaches.
- End-User Demographics: Hospitals, specialized clinics, and research institutions.
- M&A Activity: Significant activity in the broader neurological device market (xx million USD, 2019-2024), with a limited number of direct deals (xx) specifically within the Alzheimer's non-pharmacological treatment segment.
Alzheimer's Non-Pharmacological Treatment Device Growth Trends & Insights
The Alzheimer's Non-Pharmacological Treatment Device market is experiencing substantial growth, driven by the increasing prevalence of Alzheimer's disease and the limitations of current pharmaceutical interventions. Market size is projected to reach xx million units by 2033, exhibiting a CAGR of xx% during the forecast period (2025-2033). Adoption rates are gradually increasing, fueled by growing awareness of non-pharmacological treatment options and technological advancements. The market is witnessing significant technological disruptions, with the integration of AI and advanced imaging techniques enhancing diagnosis and treatment efficacy. Consumer behavior shifts include a preference for less invasive and personalized treatment approaches. Furthermore, increased healthcare expenditure globally and a growing elderly population are key growth enablers. The market penetration rate is anticipated to grow from xx% in 2025 to xx% by 2033.
Dominant Regions, Countries, or Segments in Alzheimer's Non-Pharmacological Treatment Device
North America currently dominates the Alzheimer's Non-Pharmacological Treatment Device market, owing to factors like high healthcare expenditure, advanced healthcare infrastructure, and a large elderly population. Europe is another significant market, with strong regulatory support and a growing awareness of the disease. Asia-Pacific is poised for substantial growth, driven by rising prevalence rates and increasing healthcare investments. The segment focused on neurostimulation devices currently holds the largest market share, followed by cognitive training and rehabilitation devices.
- North America: High healthcare expenditure, advanced infrastructure, large elderly population.
- Europe: Strong regulatory support, growing disease awareness.
- Asia-Pacific: Rising prevalence rates, increasing healthcare investments.
- Dominant Segment: Neurostimulation devices, followed by cognitive training and rehabilitation devices.
Alzheimer's Non-Pharmacological Treatment Device Product Landscape
The Alzheimer's Non-Pharmacological Treatment Device market encompasses a range of products, including neurostimulation devices (tDCS, TMS), cognitive training software, and virtual reality-based therapies. These devices offer unique selling propositions such as non-invasiveness, personalized treatment plans, and improved patient compliance. Technological advancements are leading to miniaturization, improved efficacy, and better integration with other healthcare technologies.
Key Drivers, Barriers & Challenges in Alzheimer's Non-Pharmacological Treatment Device
Key Drivers: The increasing prevalence of Alzheimer's disease, limitations of pharmaceutical treatments, technological advancements in neurostimulation and AI, rising healthcare expenditure, and supportive government initiatives are key drivers.
Challenges & Restraints: High cost of treatment, limited reimbursement policies, lack of awareness about non-pharmacological options, and the need for extensive clinical trials to demonstrate efficacy pose significant challenges. Supply chain disruptions caused by global events could also impact availability and costs. Furthermore, the long development timelines associated with regulatory approvals are a barrier to market entry for new companies.
Emerging Opportunities in Alzheimer's Non-Pharmacological Treatment Device
Emerging opportunities lie in untapped markets (particularly in developing nations), the development of personalized treatment plans using AI and machine learning, and the integration of wearable sensors for continuous monitoring and early detection. Furthermore, there is potential for expansion into new therapeutic areas related to cognitive decline and dementia.
Growth Accelerators in the Alzheimer's Non-Pharmacological Treatment Device Industry
Technological breakthroughs such as advanced brain imaging techniques, improved neurostimulation technologies, and the development of AI-powered diagnostic tools are key growth catalysts. Strategic partnerships between device manufacturers, healthcare providers, and research institutions are also accelerating market expansion. Expansion into emerging markets and the development of novel therapeutic applications will further fuel market growth.
Key Players Shaping the Alzheimer's Non-Pharmacological Treatment Device Market
- Magstim
- NeuroConn
- Abbott
- Boston Scientific
- Neuropace
- Aleva Neurotherapeutics
- Laborie
- Electrocore
- Medtronic
- Interaxon
- Fisher Wallace
- Dreem
- SceneRay
- Pins Medical
- Spectris
Notable Milestones in Alzheimer's Non-Pharmacological Treatment Device Sector
- 2020: FDA approval of a novel neurostimulation device for Alzheimer's-related cognitive impairment.
- 2022: Launch of a new AI-powered diagnostic tool for early detection of Alzheimer's.
- 2023: Major pharmaceutical company announces strategic partnership with a device manufacturer to develop combined pharmacological and non-pharmacological treatment approaches.
In-Depth Alzheimer's Non-Pharmacological Treatment Device Market Outlook
The Alzheimer's Non-Pharmacological Treatment Device market is poised for continued strong growth, driven by technological innovations, increasing prevalence of Alzheimer's, and greater acceptance of non-pharmacological therapies. Strategic investments in R&D, expansion into emerging markets, and the development of personalized treatment approaches will be critical for sustained success in this rapidly evolving sector. The market's future hinges on overcoming challenges related to cost, reimbursement, and widespread adoption.
Alzheimer's Non-Pharmacological Treatment Device Segmentation
-
1. Application
- 1.1. Hospital
- 1.2. Clinic
- 1.3. Others
-
2. Types
- 2.1. Brain Stimulation Device
- 2.2. Optical and Acoustic Stimulation Device
- 2.3. Others
Alzheimer's Non-Pharmacological Treatment Device Segmentation By Geography
- 1. CA
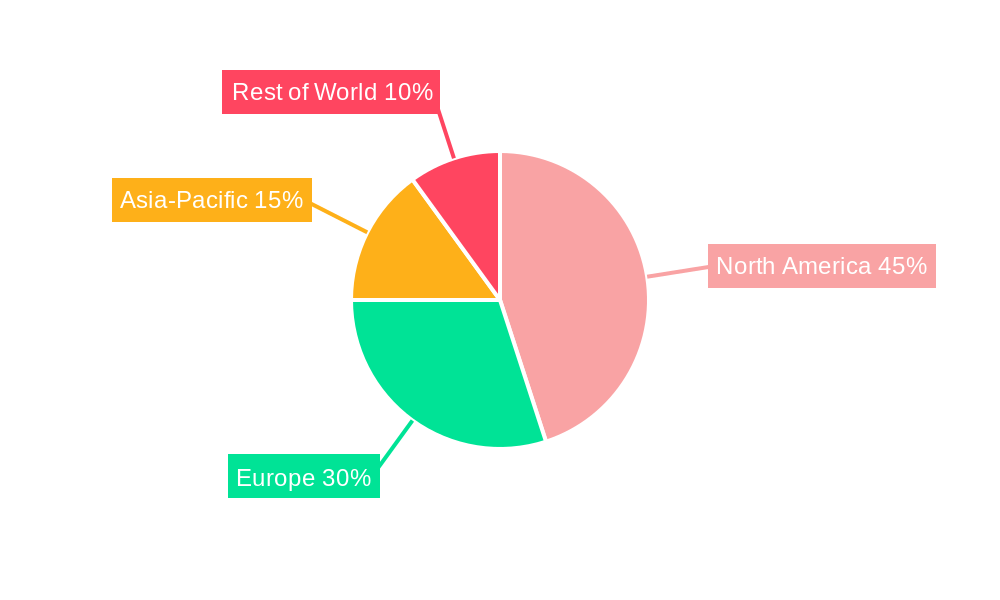
Alzheimer's Non-Pharmacological Treatment Device Regional Market Share

Geographic Coverage of Alzheimer's Non-Pharmacological Treatment Device
Alzheimer's Non-Pharmacological Treatment Device REPORT HIGHLIGHTS
| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 8.5% from 2020-2034 |
| Segmentation |
|
Table of Contents
- 1. Introduction
- 1.1. Research Scope
- 1.2. Market Segmentation
- 1.3. Research Methodology
- 1.4. Definitions and Assumptions
- 2. Executive Summary
- 2.1. Introduction
- 3. Market Dynamics
- 3.1. Introduction
- 3.2. Market Drivers
- 3.3. Market Restrains
- 3.4. Market Trends
- 4. Market Factor Analysis
- 4.1. Porters Five Forces
- 4.2. Supply/Value Chain
- 4.3. PESTEL analysis
- 4.4. Market Entropy
- 4.5. Patent/Trademark Analysis
- 5. Alzheimer's Non-Pharmacological Treatment Device Analysis, Insights and Forecast, 2020-2032
- 5.1. Market Analysis, Insights and Forecast - by Application
- 5.1.1. Hospital
- 5.1.2. Clinic
- 5.1.3. Others
- 5.2. Market Analysis, Insights and Forecast - by Types
- 5.2.1. Brain Stimulation Device
- 5.2.2. Optical and Acoustic Stimulation Device
- 5.2.3. Others
- 5.3. Market Analysis, Insights and Forecast - by Region
- 5.3.1. CA
- 5.1. Market Analysis, Insights and Forecast - by Application
- 6. Competitive Analysis
- 6.1. Market Share Analysis 2025
- 6.2. Company Profiles
- 6.2.1 Magstim
- 6.2.1.1. Overview
- 6.2.1.2. Products
- 6.2.1.3. SWOT Analysis
- 6.2.1.4. Recent Developments
- 6.2.1.5. Financials (Based on Availability)
- 6.2.2 NeuroConn
- 6.2.2.1. Overview
- 6.2.2.2. Products
- 6.2.2.3. SWOT Analysis
- 6.2.2.4. Recent Developments
- 6.2.2.5. Financials (Based on Availability)
- 6.2.3 Abbott
- 6.2.3.1. Overview
- 6.2.3.2. Products
- 6.2.3.3. SWOT Analysis
- 6.2.3.4. Recent Developments
- 6.2.3.5. Financials (Based on Availability)
- 6.2.4 Boston Scientific
- 6.2.4.1. Overview
- 6.2.4.2. Products
- 6.2.4.3. SWOT Analysis
- 6.2.4.4. Recent Developments
- 6.2.4.5. Financials (Based on Availability)
- 6.2.5 Neuropace
- 6.2.5.1. Overview
- 6.2.5.2. Products
- 6.2.5.3. SWOT Analysis
- 6.2.5.4. Recent Developments
- 6.2.5.5. Financials (Based on Availability)
- 6.2.6 Aleva Neurotherapeutics
- 6.2.6.1. Overview
- 6.2.6.2. Products
- 6.2.6.3. SWOT Analysis
- 6.2.6.4. Recent Developments
- 6.2.6.5. Financials (Based on Availability)
- 6.2.7 Laborie
- 6.2.7.1. Overview
- 6.2.7.2. Products
- 6.2.7.3. SWOT Analysis
- 6.2.7.4. Recent Developments
- 6.2.7.5. Financials (Based on Availability)
- 6.2.8 Electrocore
- 6.2.8.1. Overview
- 6.2.8.2. Products
- 6.2.8.3. SWOT Analysis
- 6.2.8.4. Recent Developments
- 6.2.8.5. Financials (Based on Availability)
- 6.2.9 Medtronic
- 6.2.9.1. Overview
- 6.2.9.2. Products
- 6.2.9.3. SWOT Analysis
- 6.2.9.4. Recent Developments
- 6.2.9.5. Financials (Based on Availability)
- 6.2.10 Interaxon
- 6.2.10.1. Overview
- 6.2.10.2. Products
- 6.2.10.3. SWOT Analysis
- 6.2.10.4. Recent Developments
- 6.2.10.5. Financials (Based on Availability)
- 6.2.11 Fisher Wallace
- 6.2.11.1. Overview
- 6.2.11.2. Products
- 6.2.11.3. SWOT Analysis
- 6.2.11.4. Recent Developments
- 6.2.11.5. Financials (Based on Availability)
- 6.2.12 Dreem
- 6.2.12.1. Overview
- 6.2.12.2. Products
- 6.2.12.3. SWOT Analysis
- 6.2.12.4. Recent Developments
- 6.2.12.5. Financials (Based on Availability)
- 6.2.13 SceneRay
- 6.2.13.1. Overview
- 6.2.13.2. Products
- 6.2.13.3. SWOT Analysis
- 6.2.13.4. Recent Developments
- 6.2.13.5. Financials (Based on Availability)
- 6.2.14 Pins Medical
- 6.2.14.1. Overview
- 6.2.14.2. Products
- 6.2.14.3. SWOT Analysis
- 6.2.14.4. Recent Developments
- 6.2.14.5. Financials (Based on Availability)
- 6.2.15 Spectris
- 6.2.15.1. Overview
- 6.2.15.2. Products
- 6.2.15.3. SWOT Analysis
- 6.2.15.4. Recent Developments
- 6.2.15.5. Financials (Based on Availability)
- 6.2.1 Magstim
List of Figures
- Figure 1: Alzheimer's Non-Pharmacological Treatment Device Revenue Breakdown (undefined, %) by Product 2025 & 2033
- Figure 2: Alzheimer's Non-Pharmacological Treatment Device Share (%) by Company 2025
List of Tables
- Table 1: Alzheimer's Non-Pharmacological Treatment Device Revenue undefined Forecast, by Application 2020 & 2033
- Table 2: Alzheimer's Non-Pharmacological Treatment Device Revenue undefined Forecast, by Types 2020 & 2033
- Table 3: Alzheimer's Non-Pharmacological Treatment Device Revenue undefined Forecast, by Region 2020 & 2033
- Table 4: Alzheimer's Non-Pharmacological Treatment Device Revenue undefined Forecast, by Application 2020 & 2033
- Table 5: Alzheimer's Non-Pharmacological Treatment Device Revenue undefined Forecast, by Types 2020 & 2033
- Table 6: Alzheimer's Non-Pharmacological Treatment Device Revenue undefined Forecast, by Country 2020 & 2033
Frequently Asked Questions
1. What is the projected Compound Annual Growth Rate (CAGR) of the Alzheimer's Non-Pharmacological Treatment Device?
The projected CAGR is approximately 8.5%.
2. Which companies are prominent players in the Alzheimer's Non-Pharmacological Treatment Device?
Key companies in the market include Magstim, NeuroConn, Abbott, Boston Scientific, Neuropace, Aleva Neurotherapeutics, Laborie, Electrocore, Medtronic, Interaxon, Fisher Wallace, Dreem, SceneRay, Pins Medical, Spectris.
3. What are the main segments of the Alzheimer's Non-Pharmacological Treatment Device?
The market segments include Application, Types.
4. Can you provide details about the market size?
The market size is estimated to be USD XXX N/A as of 2022.
5. What are some drivers contributing to market growth?
N/A
6. What are the notable trends driving market growth?
N/A
7. Are there any restraints impacting market growth?
N/A
8. Can you provide examples of recent developments in the market?
N/A
9. What pricing options are available for accessing the report?
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 4500.00, USD 6750.00, and USD 9000.00 respectively.
10. Is the market size provided in terms of value or volume?
The market size is provided in terms of value, measured in N/A.
11. Are there any specific market keywords associated with the report?
Yes, the market keyword associated with the report is "Alzheimer's Non-Pharmacological Treatment Device," which aids in identifying and referencing the specific market segment covered.
12. How do I determine which pricing option suits my needs best?
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
13. Are there any additional resources or data provided in the Alzheimer's Non-Pharmacological Treatment Device report?
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
14. How can I stay updated on further developments or reports in the Alzheimer's Non-Pharmacological Treatment Device?
To stay informed about further developments, trends, and reports in the Alzheimer's Non-Pharmacological Treatment Device, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.
Methodology
Step 1 - Identification of Relevant Samples Size from Population Database
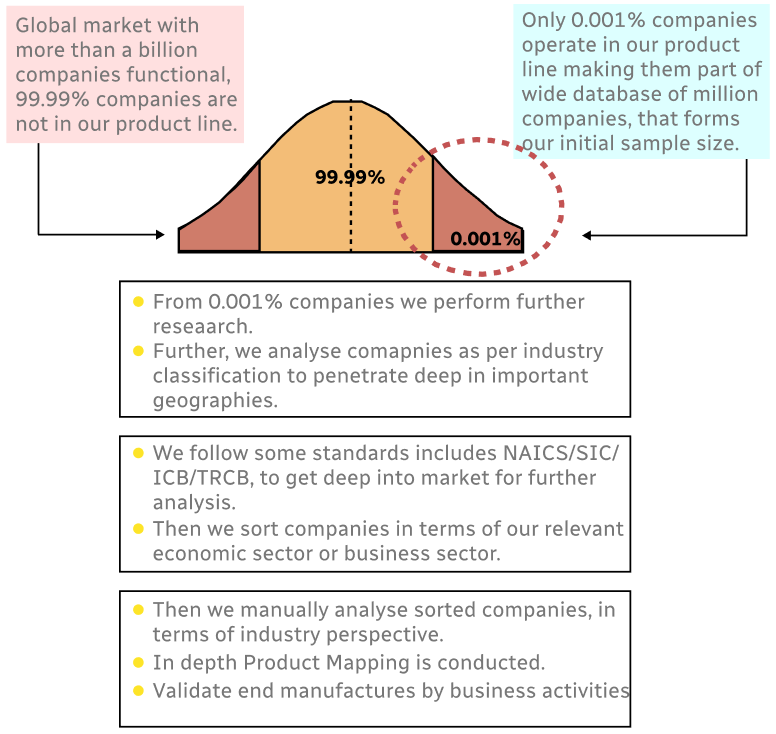
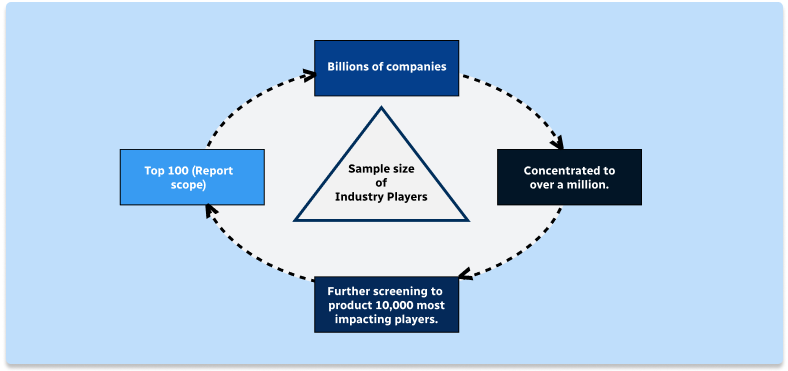
Step 2 - Approaches for Defining Global Market Size (Value, Volume* & Price*)
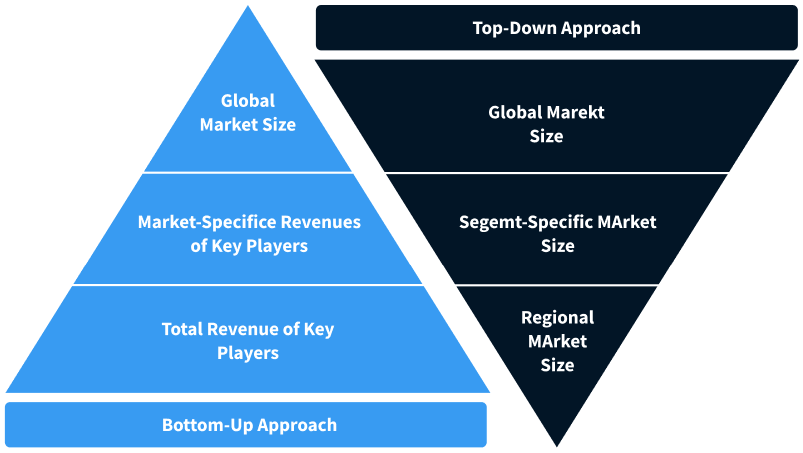
Note*: In applicable scenarios
Step 3 - Data Sources
Primary Research
- Web Analytics
- Survey Reports
- Research Institute
- Latest Research Reports
- Opinion Leaders
Secondary Research
- Annual Reports
- White Paper
- Latest Press Release
- Industry Association
- Paid Database
- Investor Presentations
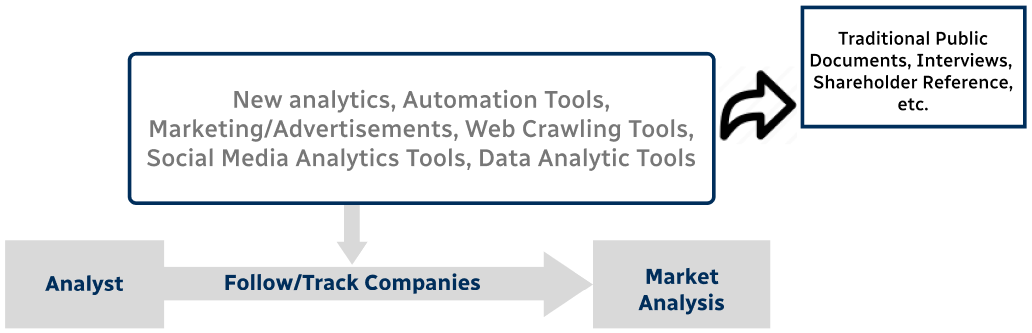
Step 4 - Data Triangulation
Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence


