Key Insights
The Biopharmaceutical Temperature Controlled Packaging market is poised for significant expansion, projected to reach a substantial $34.28 billion by 2025. This robust growth is driven by an impressive Compound Annual Growth Rate (CAGR) of 11.3% from 2019 to 2033, indicating a sustained and dynamic market trajectory. The increasing prevalence of temperature-sensitive biopharmaceuticals, including novel biologics, advanced therapies, and vaccines, necessitates sophisticated packaging solutions to maintain product integrity throughout the supply chain. Growing global healthcare expenditure, coupled with advancements in pharmaceutical manufacturing and a rising demand for specialized medicines, are key catalysts fueling this market expansion. Furthermore, the ongoing evolution of cold chain logistics, marked by technological innovations and the establishment of stringent regulatory frameworks, further underpins this optimistic market outlook.
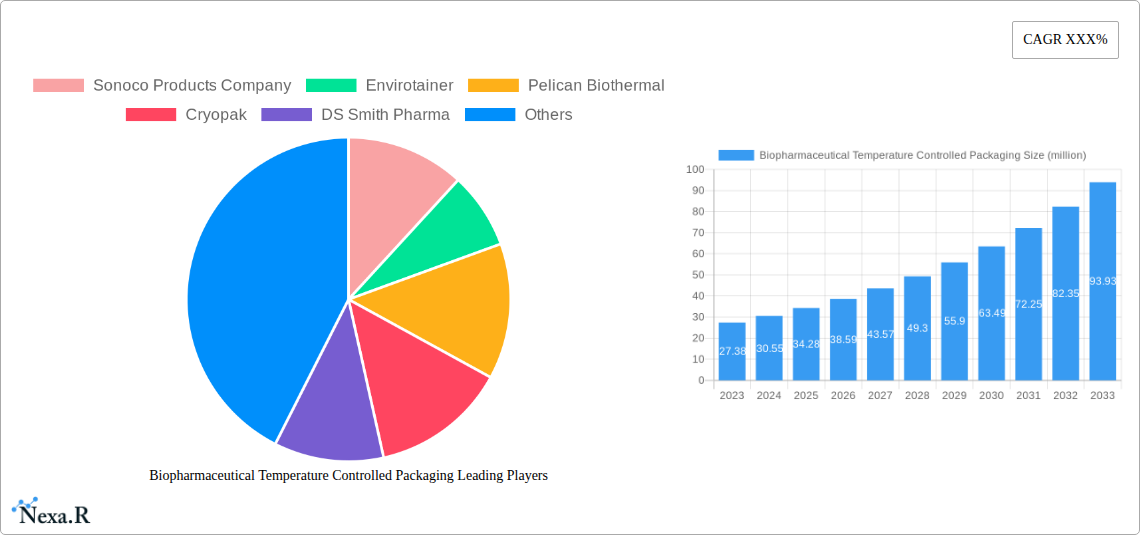
Biopharmaceutical Temperature Controlled Packaging Market Size (In Million)

The market is broadly segmented into reusable and single-use solutions, catering to diverse operational needs and cost considerations within the biopharmaceutical industry. Applications span critical areas such as temperature-sensitive pharmaceuticals, vaccines, and other specialized biological products. Geographically, North America and Europe currently dominate the market share, owing to their well-established pharmaceutical infrastructure, high adoption rates of advanced cold chain technologies, and significant research and development activities. However, the Asia Pacific region is anticipated to exhibit the fastest growth, propelled by a burgeoning biopharmaceutical sector, increasing investments in cold chain capabilities, and expanding healthcare access. Key market players are actively engaged in strategic partnerships, product innovations, and geographical expansions to capitalize on these burgeoning opportunities and maintain a competitive edge in this rapidly evolving landscape.
Biopharmaceutical Temperature Controlled Packaging Company Market Share

Biopharmaceutical Temperature Controlled Packaging Market Dynamics & Structure
The biopharmaceutical temperature-controlled packaging market, a critical component of the global pharmaceutical supply chain, is characterized by a moderately consolidated structure with key players investing heavily in technological innovation and expanding their global reach. The market's dynamism is fueled by increasing demand for temperature-sensitive biologics and vaccines, necessitating robust cold chain solutions. Regulatory frameworks, particularly those ensuring product integrity and patient safety, play a significant role in shaping market strategies. Competitive product substitutes, primarily evolving from advancements in insulation materials and active cooling technologies, are continuously pushing the boundaries of thermal performance and cost-effectiveness. End-user demographics are increasingly diverse, encompassing pharmaceutical manufacturers, contract development and manufacturing organizations (CDMOs), and specialized logistics providers. Merger and acquisition (M&A) trends are evident as companies seek to consolidate market share, gain access to new technologies, and enhance their service offerings.
- Market Concentration: The market is dominated by a few large players, but a significant number of smaller, specialized companies also contribute to the competitive landscape.
- Technological Innovation Drivers: Advancements in vacuum-insulated panels (VIPs), phase change materials (PCMs), and active refrigeration systems are key innovation drivers.
- Regulatory Frameworks: Stringent regulations from bodies like the FDA and EMA mandate precise temperature control and traceability throughout the cold chain.
- Competitive Product Substitutes: Innovations in sustainable packaging materials and the integration of IoT for real-time monitoring offer alternatives to traditional passive solutions.
- End-User Demographics: A growing segment of small and medium-sized biopharmaceutical companies necessitates flexible and scalable packaging solutions.
- M&A Trends: Recent M&A activities focus on acquiring specialized expertise in specific thermal technologies and expanding geographic footprints.
Biopharmaceutical Temperature Controlled Packaging Growth Trends & Insights
The global biopharmaceutical temperature-controlled packaging market is poised for substantial growth, driven by the escalating demand for life-saving biologics, vaccines, and advanced therapies that require stringent temperature maintenance throughout their journey from manufacturing to patient. The market size is projected to grow from approximately $XX billion in 2024 to an estimated $XX billion by 2033, exhibiting a compound annual growth rate (CAGR) of XX% during the forecast period of 2025–2033. This impressive expansion is underpinned by a confluence of factors including the accelerating development and commercialization of new biologic drugs, the increasing prevalence of chronic diseases, and the critical need for reliable cold chain logistics to ensure drug efficacy and patient safety. Furthermore, the ongoing global vaccination campaigns and the growing demand for advanced cell and gene therapies, which are highly sensitive to temperature fluctuations, are significant market accelerators.
Technological disruptions are continuously reshaping the market. The evolution from traditional single-use solutions to more sustainable and cost-effective reusable packaging systems, coupled with advancements in active temperature control technologies and smart monitoring solutions, is transforming supply chain operations. Consumer behavior shifts, driven by an increased awareness of drug integrity and a demand for transparency, are pushing stakeholders towards more sophisticated tracking and tracing capabilities. The adoption rates of advanced temperature-controlled packaging solutions are steadily increasing as companies recognize the inherent risks and financial implications of cold chain breaches. Market penetration is deepening across emerging economies as healthcare infrastructure improves and access to advanced pharmaceuticals expands.
The overarching trend is towards a more integrated and intelligent cold chain, where packaging solutions are not merely containers but active participants in maintaining product quality. This involves the integration of IoT sensors for real-time temperature monitoring, predictive analytics to anticipate potential issues, and the development of more durable and reusable packaging to reduce environmental impact and operational costs. The market is also witnessing a rise in demand for customized solutions tailored to specific drug profiles and logistical requirements, further driving innovation and specialization within the industry. The increasing complexity of biologics, requiring precise temperature ranges for stability, will continue to be a primary driver for advanced temperature-controlled packaging solutions.
Dominant Regions, Countries, or Segments in Biopharmaceutical Temperature Controlled Packaging
The Temperature-Sensitive Pharmaceuticals application segment is currently the dominant force driving growth within the global biopharmaceutical temperature-controlled packaging market. This dominance is directly attributable to the expanding pipeline and increasing commercialization of biologics, oncology drugs, and other complex pharmaceutical products that have narrow temperature stability ranges. The sheer volume and high value of these specialized therapeutics necessitate robust and reliable cold chain solutions to prevent degradation, maintain efficacy, and ensure patient safety. The growth potential within this segment remains exceptionally high due to ongoing research and development in novel biopharmaceutical products.
Key Drivers for Dominance:
- Market Share: Temperature-Sensitive Pharmaceuticals currently command an estimated XX% market share within the application segments, reflecting their substantial contribution to the overall market value and volume.
- Growth Potential: The segment is projected to experience a CAGR of XX% during the forecast period, outpacing other application areas due to the continuous introduction of new biologics and a growing global demand for advanced therapies.
- Economic Policies: Favorable reimbursement policies and government initiatives promoting access to advanced medicines in major economies like North America and Europe directly boost the demand for temperature-controlled packaging for these sensitive drugs.
- Infrastructure: The well-established pharmaceutical manufacturing and distribution infrastructure in regions like North America and Europe provides a fertile ground for the widespread adoption of specialized packaging for temperature-sensitive pharmaceuticals.
- Technological Integration: The integration of advanced thermal technologies and real-time monitoring solutions is particularly crucial and widely adopted for high-value temperature-sensitive pharmaceuticals, further solidifying this segment's dominance.
Dominant Regions & Countries:
North America, particularly the United States, stands out as the leading region in the biopharmaceutical temperature-controlled packaging market. This leadership is driven by its status as a global hub for pharmaceutical innovation, a large domestic market for biologics, and a well-developed logistics infrastructure. The country's stringent regulatory environment, while demanding, also fosters the adoption of high-quality temperature-controlled packaging solutions. Europe, with its significant pharmaceutical manufacturing base and a strong emphasis on patient safety, is another key contributor. The Asia-Pacific region is emerging as a significant growth driver, propelled by increasing healthcare spending, a burgeoning biopharmaceutical industry, and the expansion of cold chain logistics capabilities.
Biopharmaceutical Temperature Controlled Packaging Product Landscape
The product landscape for biopharmaceutical temperature-controlled packaging is characterized by continuous innovation in both passive and active solutions. Passive systems leverage advanced insulation materials like vacuum-insulated panels (VIPs) and phase change materials (PCMs) offering extended thermal protection for extended transit times. Active systems, incorporating electric refrigeration units, provide precise temperature control for highly sensitive and high-value therapeutics. The trend is towards integrated solutions that combine superior insulation with smart technologies, offering real-time temperature monitoring, data logging, and remote diagnostics. Unique selling propositions include enhanced thermal performance, compliance with international shipping regulations, and sustainable material options, meeting the evolving needs of the biopharmaceutical cold chain.
Key Drivers, Barriers & Challenges in Biopharmaceutical Temperature Controlled Packaging
Key Drivers:
The biopharmaceutical temperature-controlled packaging market is propelled by several critical drivers. The burgeoning biologics market, with its increasing complexity and temperature sensitivity, is a primary catalyst. Expanding global vaccination programs and the rapid development of cell and gene therapies further amplify the demand for specialized cold chain solutions. Technological advancements in insulation materials, active cooling systems, and IoT integration for real-time monitoring enhance product integrity and supply chain visibility. Furthermore, stringent regulatory mandates for maintaining product efficacy and patient safety are non-negotiable drivers for adopting high-performance packaging.
Barriers & Challenges:
Despite robust growth, the market faces significant challenges. The high cost associated with advanced temperature-controlled packaging solutions, especially for single-use options, can be a barrier for smaller companies or in cost-sensitive markets. The complexity of global shipping regulations and varying compliance requirements across different regions can create logistical hurdles. Supply chain disruptions, whether due to geopolitical events, natural disasters, or logistical bottlenecks, pose a constant threat to maintaining the cold chain integrity. Moreover, the need for specialized training and handling protocols for these sophisticated packaging systems can be a resource-intensive challenge for some organizations. The environmental impact of single-use packaging solutions also presents a growing concern, pushing for more sustainable alternatives.
Emerging Opportunities in Biopharmaceutical Temperature Controlled Packaging
Emerging opportunities in the biopharmaceutical temperature-controlled packaging sector lie in the development of more sustainable and eco-friendly packaging solutions, including recyclable and biodegradable materials. The increasing demand for last-mile delivery solutions, particularly for vaccines and critical medicines in remote or underserved areas, presents a significant growth avenue for smaller, agile packaging providers. The integration of advanced AI and machine learning for predictive analytics in cold chain management offers opportunities to optimize routes, anticipate potential temperature excursions, and minimize waste. Furthermore, the expanding market for personalized medicine and advanced therapies, which often have extremely sensitive temperature requirements, will drive the need for highly customized and sophisticated packaging solutions.
Growth Accelerators in the Biopharmaceutical Temperature Controlled Packaging Industry
Growth accelerators in the biopharmaceutical temperature-controlled packaging industry are fundamentally linked to breakthroughs in material science and technological integration. The development of next-generation insulation materials offering superior R-values and reduced thickness allows for larger payload volumes and lighter packaging, translating to lower shipping costs. The miniaturization and increased efficiency of active refrigeration units are making active temperature-controlled solutions more accessible and cost-effective for a wider range of applications. Strategic partnerships between packaging manufacturers, pharmaceutical companies, and logistics providers are crucial for co-creating optimized cold chain solutions, streamlining operations, and ensuring seamless end-to-end delivery. Market expansion into emerging economies, driven by improving healthcare infrastructure and increasing access to advanced pharmaceuticals, represents another significant growth accelerator.
Key Players Shaping the Biopharmaceutical Temperature Controlled Packaging Market
- Sonoco Products Company
- Envirotainer
- Pelican Biothermal
- Cryopak
- DS Smith Pharma
- Cold Chain Technologies
- Intelsius
- CSafe
- Softbox Systems
- Va-Q-tec AG
- Sofrigam
- American Aerogel Corporation
- EcoCool Gmbh
- Aeris Dynamics
- Dokasch
- Hazgo
- Beijing Roloo Technology CO.,Ltd
- Insulated Products Corporation
- Inmark Packaging
- Cold Chain Tools
- Exeltainer SL
- Inno Cool Pvt Ltd
- Cryo Store
Notable Milestones in Biopharmaceutical Temperature Controlled Packaging Sector
- 2021: Sonoco Products Company acquired the temperature-controlled packaging business of ThermoGenesis Corp., strengthening its position in the cold chain solutions market.
- 2022: Envirotainer launched its new generation of active container, the Envirotainer RAP e2, offering enhanced connectivity and temperature stability for air cargo.
- 2022: Pelican Biothermal introduced its new CRYO+ and XCRYO+ product lines, designed for ultra-low temperature shipping of vaccines and advanced therapies.
- 2023: Cold Chain Technologies partnered with pharmaceutical manufacturers to expand its reusable packaging solutions across various therapeutic areas.
- 2023: Va-Q-tec AG secured significant contracts for its high-performance vacuum insulation panels (VIPs) for use in pharmaceutical cold chain logistics.
- 2024: CSafe Global announced the expansion of its container repair and maintenance network to better serve global pharmaceutical clients.
In-Depth Biopharmaceutical Temperature Controlled Packaging Market Outlook
The future of the biopharmaceutical temperature-controlled packaging market is exceptionally bright, driven by an unyielding demand for temperature-sensitive biologics and an ever-expanding global healthcare landscape. Growth accelerators such as continuous advancements in insulation technologies, the increasing adoption of smart packaging with IoT capabilities for real-time monitoring, and the strategic expansion of reusable packaging solutions will continue to shape market dynamics. The market is expected to witness further consolidation as key players seek to enhance their service portfolios and geographic reach. The growing emphasis on sustainability will also drive innovation in eco-friendly packaging materials and circular economy models. Overall, the market outlook is characterized by robust growth, significant technological evolution, and a sustained focus on ensuring the integrity and efficacy of life-saving pharmaceuticals throughout the global supply chain.
Biopharmaceutical Temperature Controlled Packaging Segmentation
-
1. Application
- 1.1. Temperature-Sensitive Pharmaceuticals
- 1.2. Vaccines
- 1.3. Others
-
2. Type
- 2.1. Reusable Solutions
- 2.2. Single Use Solutions
Biopharmaceutical Temperature Controlled Packaging Segmentation By Geography
-
1. North America
- 1.1. United States
- 1.2. Canada
- 1.3. Mexico
-
2. South America
- 2.1. Brazil
- 2.2. Argentina
- 2.3. Rest of South America
-
3. Europe
- 3.1. United Kingdom
- 3.2. Germany
- 3.3. France
- 3.4. Italy
- 3.5. Spain
- 3.6. Russia
- 3.7. Benelux
- 3.8. Nordics
- 3.9. Rest of Europe
-
4. Middle East & Africa
- 4.1. Turkey
- 4.2. Israel
- 4.3. GCC
- 4.4. North Africa
- 4.5. South Africa
- 4.6. Rest of Middle East & Africa
-
5. Asia Pacific
- 5.1. China
- 5.2. India
- 5.3. Japan
- 5.4. South Korea
- 5.5. ASEAN
- 5.6. Oceania
- 5.7. Rest of Asia Pacific
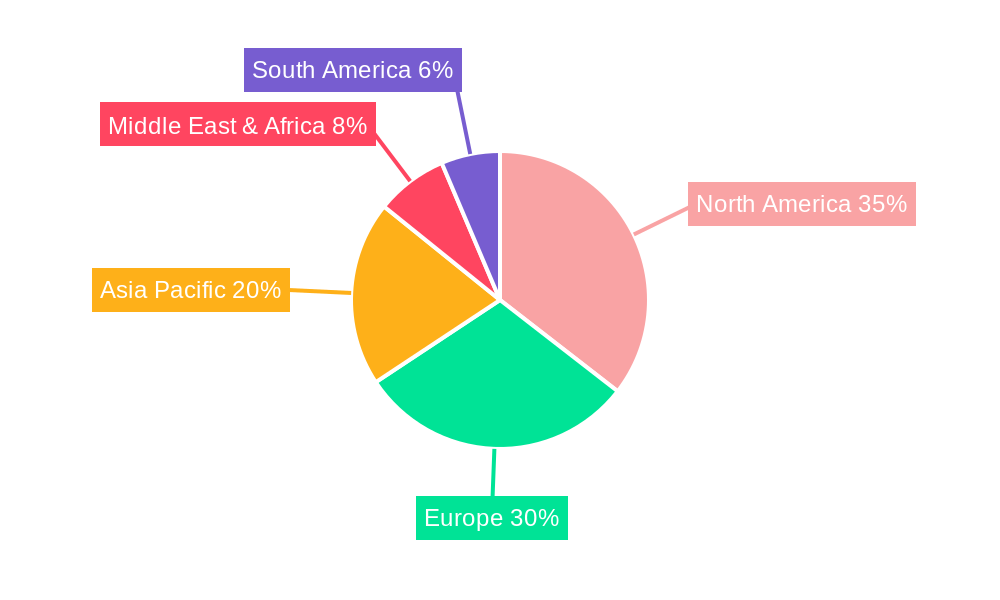
Biopharmaceutical Temperature Controlled Packaging Regional Market Share

Geographic Coverage of Biopharmaceutical Temperature Controlled Packaging
Biopharmaceutical Temperature Controlled Packaging REPORT HIGHLIGHTS
| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 7.4% from 2020-2034 |
| Segmentation |
|
Table of Contents
- 1. Introduction
- 1.1. Research Scope
- 1.2. Market Segmentation
- 1.3. Research Methodology
- 1.4. Definitions and Assumptions
- 2. Executive Summary
- 2.1. Introduction
- 3. Market Dynamics
- 3.1. Introduction
- 3.2. Market Drivers
- 3.3. Market Restrains
- 3.4. Market Trends
- 4. Market Factor Analysis
- 4.1. Porters Five Forces
- 4.2. Supply/Value Chain
- 4.3. PESTEL analysis
- 4.4. Market Entropy
- 4.5. Patent/Trademark Analysis
- 5. Global Biopharmaceutical Temperature Controlled Packaging Analysis, Insights and Forecast, 2020-2032
- 5.1. Market Analysis, Insights and Forecast - by Application
- 5.1.1. Temperature-Sensitive Pharmaceuticals
- 5.1.2. Vaccines
- 5.1.3. Others
- 5.2. Market Analysis, Insights and Forecast - by Type
- 5.2.1. Reusable Solutions
- 5.2.2. Single Use Solutions
- 5.3. Market Analysis, Insights and Forecast - by Region
- 5.3.1. North America
- 5.3.2. South America
- 5.3.3. Europe
- 5.3.4. Middle East & Africa
- 5.3.5. Asia Pacific
- 5.1. Market Analysis, Insights and Forecast - by Application
- 6. North America Biopharmaceutical Temperature Controlled Packaging Analysis, Insights and Forecast, 2020-2032
- 6.1. Market Analysis, Insights and Forecast - by Application
- 6.1.1. Temperature-Sensitive Pharmaceuticals
- 6.1.2. Vaccines
- 6.1.3. Others
- 6.2. Market Analysis, Insights and Forecast - by Type
- 6.2.1. Reusable Solutions
- 6.2.2. Single Use Solutions
- 6.1. Market Analysis, Insights and Forecast - by Application
- 7. South America Biopharmaceutical Temperature Controlled Packaging Analysis, Insights and Forecast, 2020-2032
- 7.1. Market Analysis, Insights and Forecast - by Application
- 7.1.1. Temperature-Sensitive Pharmaceuticals
- 7.1.2. Vaccines
- 7.1.3. Others
- 7.2. Market Analysis, Insights and Forecast - by Type
- 7.2.1. Reusable Solutions
- 7.2.2. Single Use Solutions
- 7.1. Market Analysis, Insights and Forecast - by Application
- 8. Europe Biopharmaceutical Temperature Controlled Packaging Analysis, Insights and Forecast, 2020-2032
- 8.1. Market Analysis, Insights and Forecast - by Application
- 8.1.1. Temperature-Sensitive Pharmaceuticals
- 8.1.2. Vaccines
- 8.1.3. Others
- 8.2. Market Analysis, Insights and Forecast - by Type
- 8.2.1. Reusable Solutions
- 8.2.2. Single Use Solutions
- 8.1. Market Analysis, Insights and Forecast - by Application
- 9. Middle East & Africa Biopharmaceutical Temperature Controlled Packaging Analysis, Insights and Forecast, 2020-2032
- 9.1. Market Analysis, Insights and Forecast - by Application
- 9.1.1. Temperature-Sensitive Pharmaceuticals
- 9.1.2. Vaccines
- 9.1.3. Others
- 9.2. Market Analysis, Insights and Forecast - by Type
- 9.2.1. Reusable Solutions
- 9.2.2. Single Use Solutions
- 9.1. Market Analysis, Insights and Forecast - by Application
- 10. Asia Pacific Biopharmaceutical Temperature Controlled Packaging Analysis, Insights and Forecast, 2020-2032
- 10.1. Market Analysis, Insights and Forecast - by Application
- 10.1.1. Temperature-Sensitive Pharmaceuticals
- 10.1.2. Vaccines
- 10.1.3. Others
- 10.2. Market Analysis, Insights and Forecast - by Type
- 10.2.1. Reusable Solutions
- 10.2.2. Single Use Solutions
- 10.1. Market Analysis, Insights and Forecast - by Application
- 11. Competitive Analysis
- 11.1. Global Market Share Analysis 2025
- 11.2. Company Profiles
- 11.2.1 Sonoco Products Company
- 11.2.1.1. Overview
- 11.2.1.2. Products
- 11.2.1.3. SWOT Analysis
- 11.2.1.4. Recent Developments
- 11.2.1.5. Financials (Based on Availability)
- 11.2.2 Envirotainer
- 11.2.2.1. Overview
- 11.2.2.2. Products
- 11.2.2.3. SWOT Analysis
- 11.2.2.4. Recent Developments
- 11.2.2.5. Financials (Based on Availability)
- 11.2.3 Pelican Biothermal
- 11.2.3.1. Overview
- 11.2.3.2. Products
- 11.2.3.3. SWOT Analysis
- 11.2.3.4. Recent Developments
- 11.2.3.5. Financials (Based on Availability)
- 11.2.4 Cryopak
- 11.2.4.1. Overview
- 11.2.4.2. Products
- 11.2.4.3. SWOT Analysis
- 11.2.4.4. Recent Developments
- 11.2.4.5. Financials (Based on Availability)
- 11.2.5 DS Smith Pharma
- 11.2.5.1. Overview
- 11.2.5.2. Products
- 11.2.5.3. SWOT Analysis
- 11.2.5.4. Recent Developments
- 11.2.5.5. Financials (Based on Availability)
- 11.2.6 Cold Chain Technologies
- 11.2.6.1. Overview
- 11.2.6.2. Products
- 11.2.6.3. SWOT Analysis
- 11.2.6.4. Recent Developments
- 11.2.6.5. Financials (Based on Availability)
- 11.2.7 Intelsius
- 11.2.7.1. Overview
- 11.2.7.2. Products
- 11.2.7.3. SWOT Analysis
- 11.2.7.4. Recent Developments
- 11.2.7.5. Financials (Based on Availability)
- 11.2.8 CSafe
- 11.2.8.1. Overview
- 11.2.8.2. Products
- 11.2.8.3. SWOT Analysis
- 11.2.8.4. Recent Developments
- 11.2.8.5. Financials (Based on Availability)
- 11.2.9 Softbox Systems
- 11.2.9.1. Overview
- 11.2.9.2. Products
- 11.2.9.3. SWOT Analysis
- 11.2.9.4. Recent Developments
- 11.2.9.5. Financials (Based on Availability)
- 11.2.10 Va-Q-tec AG
- 11.2.10.1. Overview
- 11.2.10.2. Products
- 11.2.10.3. SWOT Analysis
- 11.2.10.4. Recent Developments
- 11.2.10.5. Financials (Based on Availability)
- 11.2.11 Sofrigam
- 11.2.11.1. Overview
- 11.2.11.2. Products
- 11.2.11.3. SWOT Analysis
- 11.2.11.4. Recent Developments
- 11.2.11.5. Financials (Based on Availability)
- 11.2.12 American Aerogel Corporation
- 11.2.12.1. Overview
- 11.2.12.2. Products
- 11.2.12.3. SWOT Analysis
- 11.2.12.4. Recent Developments
- 11.2.12.5. Financials (Based on Availability)
- 11.2.13 EcoCool Gmbh
- 11.2.13.1. Overview
- 11.2.13.2. Products
- 11.2.13.3. SWOT Analysis
- 11.2.13.4. Recent Developments
- 11.2.13.5. Financials (Based on Availability)
- 11.2.14 Aeris Dynamics
- 11.2.14.1. Overview
- 11.2.14.2. Products
- 11.2.14.3. SWOT Analysis
- 11.2.14.4. Recent Developments
- 11.2.14.5. Financials (Based on Availability)
- 11.2.15 Dokasch
- 11.2.15.1. Overview
- 11.2.15.2. Products
- 11.2.15.3. SWOT Analysis
- 11.2.15.4. Recent Developments
- 11.2.15.5. Financials (Based on Availability)
- 11.2.16 Hazgo
- 11.2.16.1. Overview
- 11.2.16.2. Products
- 11.2.16.3. SWOT Analysis
- 11.2.16.4. Recent Developments
- 11.2.16.5. Financials (Based on Availability)
- 11.2.17 Beijing Roloo Technology CO.Ltd
- 11.2.17.1. Overview
- 11.2.17.2. Products
- 11.2.17.3. SWOT Analysis
- 11.2.17.4. Recent Developments
- 11.2.17.5. Financials (Based on Availability)
- 11.2.18 Insulated Products Corporation
- 11.2.18.1. Overview
- 11.2.18.2. Products
- 11.2.18.3. SWOT Analysis
- 11.2.18.4. Recent Developments
- 11.2.18.5. Financials (Based on Availability)
- 11.2.19 Inmark Packaging
- 11.2.19.1. Overview
- 11.2.19.2. Products
- 11.2.19.3. SWOT Analysis
- 11.2.19.4. Recent Developments
- 11.2.19.5. Financials (Based on Availability)
- 11.2.20 Cold Chain Tools
- 11.2.20.1. Overview
- 11.2.20.2. Products
- 11.2.20.3. SWOT Analysis
- 11.2.20.4. Recent Developments
- 11.2.20.5. Financials (Based on Availability)
- 11.2.21 Exeltainer SL
- 11.2.21.1. Overview
- 11.2.21.2. Products
- 11.2.21.3. SWOT Analysis
- 11.2.21.4. Recent Developments
- 11.2.21.5. Financials (Based on Availability)
- 11.2.22 Inno Cool Pvt Ltd
- 11.2.22.1. Overview
- 11.2.22.2. Products
- 11.2.22.3. SWOT Analysis
- 11.2.22.4. Recent Developments
- 11.2.22.5. Financials (Based on Availability)
- 11.2.23 Cryo Store
- 11.2.23.1. Overview
- 11.2.23.2. Products
- 11.2.23.3. SWOT Analysis
- 11.2.23.4. Recent Developments
- 11.2.23.5. Financials (Based on Availability)
- 11.2.1 Sonoco Products Company
List of Figures
- Figure 1: Global Biopharmaceutical Temperature Controlled Packaging Revenue Breakdown (undefined, %) by Region 2025 & 2033
- Figure 2: North America Biopharmaceutical Temperature Controlled Packaging Revenue (undefined), by Application 2025 & 2033
- Figure 3: North America Biopharmaceutical Temperature Controlled Packaging Revenue Share (%), by Application 2025 & 2033
- Figure 4: North America Biopharmaceutical Temperature Controlled Packaging Revenue (undefined), by Type 2025 & 2033
- Figure 5: North America Biopharmaceutical Temperature Controlled Packaging Revenue Share (%), by Type 2025 & 2033
- Figure 6: North America Biopharmaceutical Temperature Controlled Packaging Revenue (undefined), by Country 2025 & 2033
- Figure 7: North America Biopharmaceutical Temperature Controlled Packaging Revenue Share (%), by Country 2025 & 2033
- Figure 8: South America Biopharmaceutical Temperature Controlled Packaging Revenue (undefined), by Application 2025 & 2033
- Figure 9: South America Biopharmaceutical Temperature Controlled Packaging Revenue Share (%), by Application 2025 & 2033
- Figure 10: South America Biopharmaceutical Temperature Controlled Packaging Revenue (undefined), by Type 2025 & 2033
- Figure 11: South America Biopharmaceutical Temperature Controlled Packaging Revenue Share (%), by Type 2025 & 2033
- Figure 12: South America Biopharmaceutical Temperature Controlled Packaging Revenue (undefined), by Country 2025 & 2033
- Figure 13: South America Biopharmaceutical Temperature Controlled Packaging Revenue Share (%), by Country 2025 & 2033
- Figure 14: Europe Biopharmaceutical Temperature Controlled Packaging Revenue (undefined), by Application 2025 & 2033
- Figure 15: Europe Biopharmaceutical Temperature Controlled Packaging Revenue Share (%), by Application 2025 & 2033
- Figure 16: Europe Biopharmaceutical Temperature Controlled Packaging Revenue (undefined), by Type 2025 & 2033
- Figure 17: Europe Biopharmaceutical Temperature Controlled Packaging Revenue Share (%), by Type 2025 & 2033
- Figure 18: Europe Biopharmaceutical Temperature Controlled Packaging Revenue (undefined), by Country 2025 & 2033
- Figure 19: Europe Biopharmaceutical Temperature Controlled Packaging Revenue Share (%), by Country 2025 & 2033
- Figure 20: Middle East & Africa Biopharmaceutical Temperature Controlled Packaging Revenue (undefined), by Application 2025 & 2033
- Figure 21: Middle East & Africa Biopharmaceutical Temperature Controlled Packaging Revenue Share (%), by Application 2025 & 2033
- Figure 22: Middle East & Africa Biopharmaceutical Temperature Controlled Packaging Revenue (undefined), by Type 2025 & 2033
- Figure 23: Middle East & Africa Biopharmaceutical Temperature Controlled Packaging Revenue Share (%), by Type 2025 & 2033
- Figure 24: Middle East & Africa Biopharmaceutical Temperature Controlled Packaging Revenue (undefined), by Country 2025 & 2033
- Figure 25: Middle East & Africa Biopharmaceutical Temperature Controlled Packaging Revenue Share (%), by Country 2025 & 2033
- Figure 26: Asia Pacific Biopharmaceutical Temperature Controlled Packaging Revenue (undefined), by Application 2025 & 2033
- Figure 27: Asia Pacific Biopharmaceutical Temperature Controlled Packaging Revenue Share (%), by Application 2025 & 2033
- Figure 28: Asia Pacific Biopharmaceutical Temperature Controlled Packaging Revenue (undefined), by Type 2025 & 2033
- Figure 29: Asia Pacific Biopharmaceutical Temperature Controlled Packaging Revenue Share (%), by Type 2025 & 2033
- Figure 30: Asia Pacific Biopharmaceutical Temperature Controlled Packaging Revenue (undefined), by Country 2025 & 2033
- Figure 31: Asia Pacific Biopharmaceutical Temperature Controlled Packaging Revenue Share (%), by Country 2025 & 2033
List of Tables
- Table 1: Global Biopharmaceutical Temperature Controlled Packaging Revenue undefined Forecast, by Application 2020 & 2033
- Table 2: Global Biopharmaceutical Temperature Controlled Packaging Revenue undefined Forecast, by Type 2020 & 2033
- Table 3: Global Biopharmaceutical Temperature Controlled Packaging Revenue undefined Forecast, by Region 2020 & 2033
- Table 4: Global Biopharmaceutical Temperature Controlled Packaging Revenue undefined Forecast, by Application 2020 & 2033
- Table 5: Global Biopharmaceutical Temperature Controlled Packaging Revenue undefined Forecast, by Type 2020 & 2033
- Table 6: Global Biopharmaceutical Temperature Controlled Packaging Revenue undefined Forecast, by Country 2020 & 2033
- Table 7: United States Biopharmaceutical Temperature Controlled Packaging Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 8: Canada Biopharmaceutical Temperature Controlled Packaging Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 9: Mexico Biopharmaceutical Temperature Controlled Packaging Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 10: Global Biopharmaceutical Temperature Controlled Packaging Revenue undefined Forecast, by Application 2020 & 2033
- Table 11: Global Biopharmaceutical Temperature Controlled Packaging Revenue undefined Forecast, by Type 2020 & 2033
- Table 12: Global Biopharmaceutical Temperature Controlled Packaging Revenue undefined Forecast, by Country 2020 & 2033
- Table 13: Brazil Biopharmaceutical Temperature Controlled Packaging Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 14: Argentina Biopharmaceutical Temperature Controlled Packaging Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 15: Rest of South America Biopharmaceutical Temperature Controlled Packaging Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 16: Global Biopharmaceutical Temperature Controlled Packaging Revenue undefined Forecast, by Application 2020 & 2033
- Table 17: Global Biopharmaceutical Temperature Controlled Packaging Revenue undefined Forecast, by Type 2020 & 2033
- Table 18: Global Biopharmaceutical Temperature Controlled Packaging Revenue undefined Forecast, by Country 2020 & 2033
- Table 19: United Kingdom Biopharmaceutical Temperature Controlled Packaging Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 20: Germany Biopharmaceutical Temperature Controlled Packaging Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 21: France Biopharmaceutical Temperature Controlled Packaging Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 22: Italy Biopharmaceutical Temperature Controlled Packaging Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 23: Spain Biopharmaceutical Temperature Controlled Packaging Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 24: Russia Biopharmaceutical Temperature Controlled Packaging Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 25: Benelux Biopharmaceutical Temperature Controlled Packaging Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 26: Nordics Biopharmaceutical Temperature Controlled Packaging Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 27: Rest of Europe Biopharmaceutical Temperature Controlled Packaging Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 28: Global Biopharmaceutical Temperature Controlled Packaging Revenue undefined Forecast, by Application 2020 & 2033
- Table 29: Global Biopharmaceutical Temperature Controlled Packaging Revenue undefined Forecast, by Type 2020 & 2033
- Table 30: Global Biopharmaceutical Temperature Controlled Packaging Revenue undefined Forecast, by Country 2020 & 2033
- Table 31: Turkey Biopharmaceutical Temperature Controlled Packaging Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 32: Israel Biopharmaceutical Temperature Controlled Packaging Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 33: GCC Biopharmaceutical Temperature Controlled Packaging Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 34: North Africa Biopharmaceutical Temperature Controlled Packaging Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 35: South Africa Biopharmaceutical Temperature Controlled Packaging Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 36: Rest of Middle East & Africa Biopharmaceutical Temperature Controlled Packaging Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 37: Global Biopharmaceutical Temperature Controlled Packaging Revenue undefined Forecast, by Application 2020 & 2033
- Table 38: Global Biopharmaceutical Temperature Controlled Packaging Revenue undefined Forecast, by Type 2020 & 2033
- Table 39: Global Biopharmaceutical Temperature Controlled Packaging Revenue undefined Forecast, by Country 2020 & 2033
- Table 40: China Biopharmaceutical Temperature Controlled Packaging Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 41: India Biopharmaceutical Temperature Controlled Packaging Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 42: Japan Biopharmaceutical Temperature Controlled Packaging Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 43: South Korea Biopharmaceutical Temperature Controlled Packaging Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 44: ASEAN Biopharmaceutical Temperature Controlled Packaging Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 45: Oceania Biopharmaceutical Temperature Controlled Packaging Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 46: Rest of Asia Pacific Biopharmaceutical Temperature Controlled Packaging Revenue (undefined) Forecast, by Application 2020 & 2033
Frequently Asked Questions
1. What is the projected Compound Annual Growth Rate (CAGR) of the Biopharmaceutical Temperature Controlled Packaging?
The projected CAGR is approximately 7.4%.
2. Which companies are prominent players in the Biopharmaceutical Temperature Controlled Packaging?
Key companies in the market include Sonoco Products Company, Envirotainer, Pelican Biothermal, Cryopak, DS Smith Pharma, Cold Chain Technologies, Intelsius, CSafe, Softbox Systems, Va-Q-tec AG, Sofrigam, American Aerogel Corporation, EcoCool Gmbh, Aeris Dynamics, Dokasch, Hazgo, Beijing Roloo Technology CO.,Ltd, Insulated Products Corporation, Inmark Packaging, Cold Chain Tools, Exeltainer SL, Inno Cool Pvt Ltd, Cryo Store.
3. What are the main segments of the Biopharmaceutical Temperature Controlled Packaging?
The market segments include Application, Type.
4. Can you provide details about the market size?
The market size is estimated to be USD XXX N/A as of 2022.
5. What are some drivers contributing to market growth?
N/A
6. What are the notable trends driving market growth?
N/A
7. Are there any restraints impacting market growth?
N/A
8. Can you provide examples of recent developments in the market?
N/A
9. What pricing options are available for accessing the report?
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 4250.00, USD 6375.00, and USD 8500.00 respectively.
10. Is the market size provided in terms of value or volume?
The market size is provided in terms of value, measured in N/A.
11. Are there any specific market keywords associated with the report?
Yes, the market keyword associated with the report is "Biopharmaceutical Temperature Controlled Packaging," which aids in identifying and referencing the specific market segment covered.
12. How do I determine which pricing option suits my needs best?
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
13. Are there any additional resources or data provided in the Biopharmaceutical Temperature Controlled Packaging report?
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
14. How can I stay updated on further developments or reports in the Biopharmaceutical Temperature Controlled Packaging?
To stay informed about further developments, trends, and reports in the Biopharmaceutical Temperature Controlled Packaging, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.
Methodology
Step 1 - Identification of Relevant Samples Size from Population Database
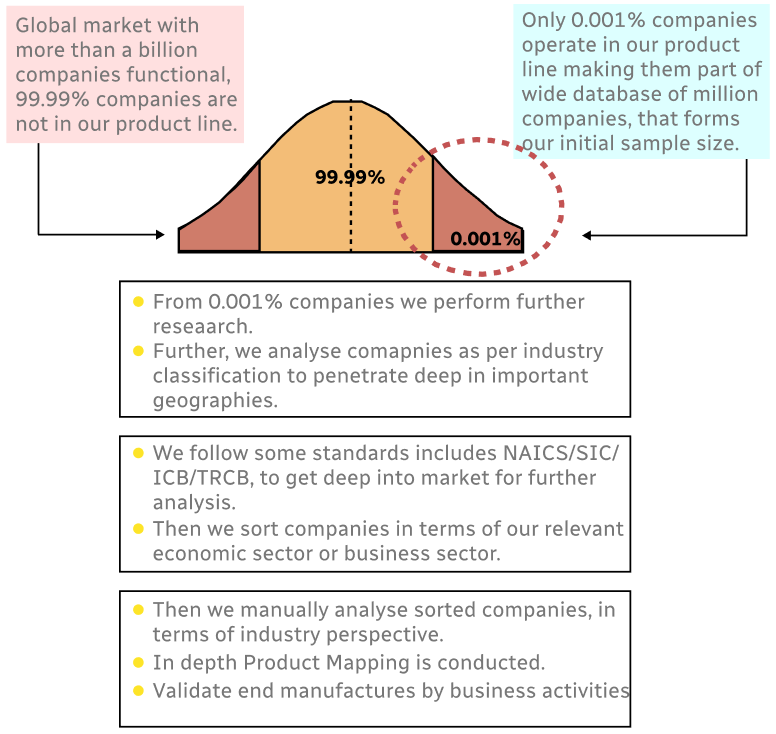
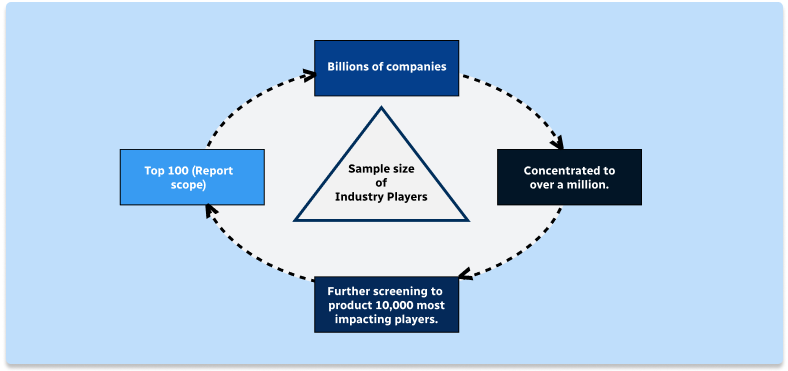
Step 2 - Approaches for Defining Global Market Size (Value, Volume* & Price*)
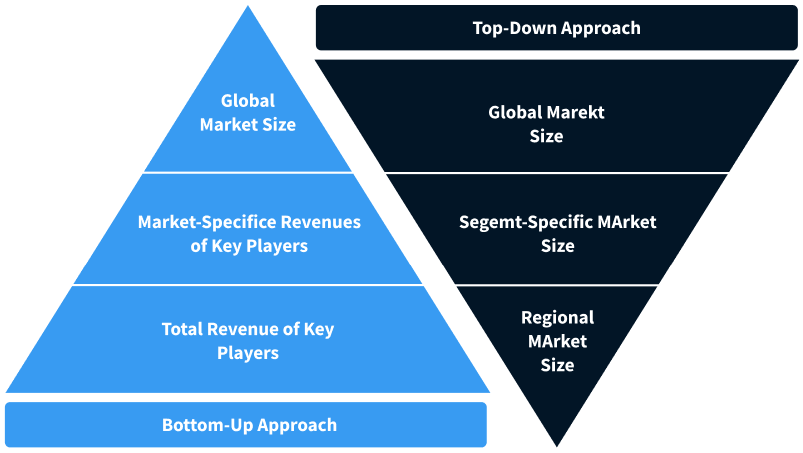
Note*: In applicable scenarios
Step 3 - Data Sources
Primary Research
- Web Analytics
- Survey Reports
- Research Institute
- Latest Research Reports
- Opinion Leaders
Secondary Research
- Annual Reports
- White Paper
- Latest Press Release
- Industry Association
- Paid Database
- Investor Presentations
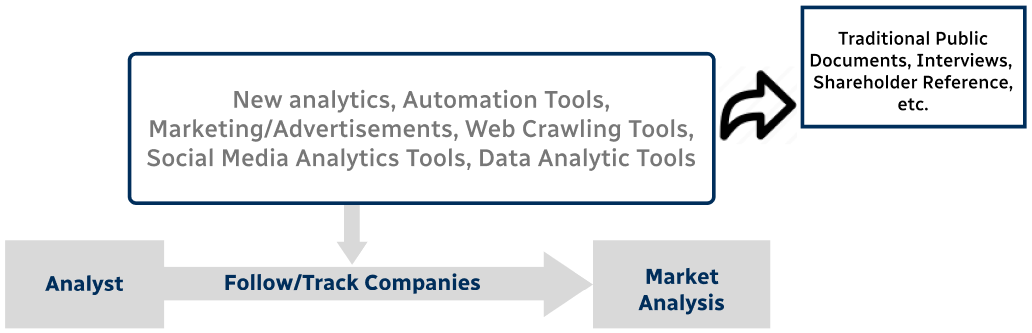
Step 4 - Data Triangulation
Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence


