Key Insights
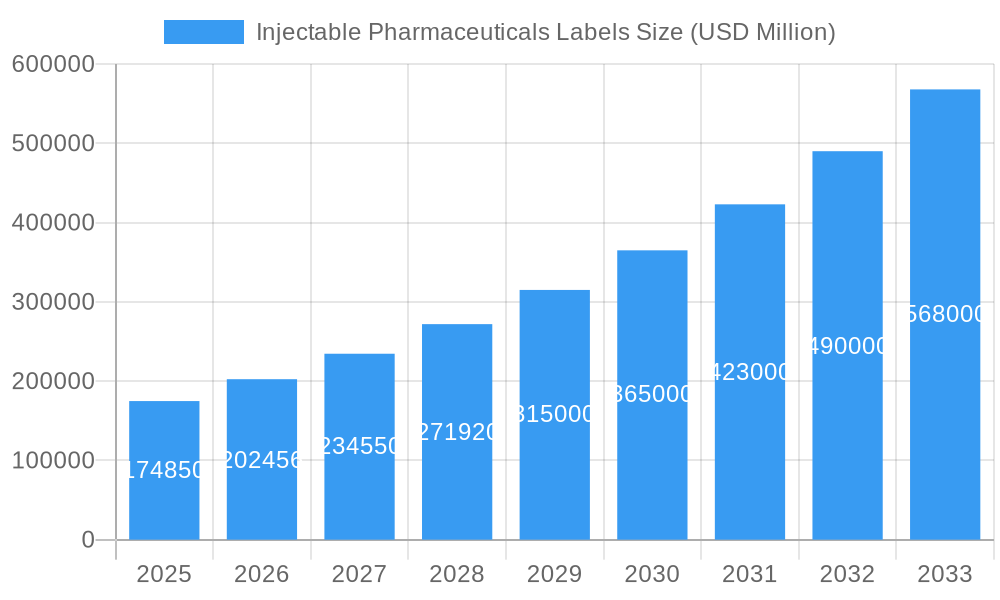
The global Injectable Pharmaceuticals Labels market is poised for significant expansion, projected to reach $174.85 billion by 2025. This robust growth is fueled by an estimated Compound Annual Growth Rate (CAGR) of 15.8% from 2019 to 2033. The burgeoning demand for injectable drugs, driven by advancements in biopharmaceuticals, a growing prevalence of chronic diseases, and an aging global population, is the primary catalyst for this market's upward trajectory. Furthermore, stringent regulatory requirements for pharmaceutical labeling, emphasizing product integrity, patient safety, and traceability, necessitate the adoption of advanced labeling solutions. The market is segmented into various applications, including Biomedical Laboratories, Medical Institutions, and Injectable Pharmaceuticals Manufacturers, each contributing to the overall demand. The "Others" segment likely encompasses specialized research facilities and diagnostic centers. Within the "Types" segment, Refrigerated Labels and Frozen Labels are crucial due to the temperature-sensitive nature of many injectable pharmaceuticals, ensuring product efficacy throughout the cold chain. Key trends include the adoption of smart labeling technologies, such as serialization and track-and-trace solutions, to combat counterfeiting and enhance supply chain visibility.
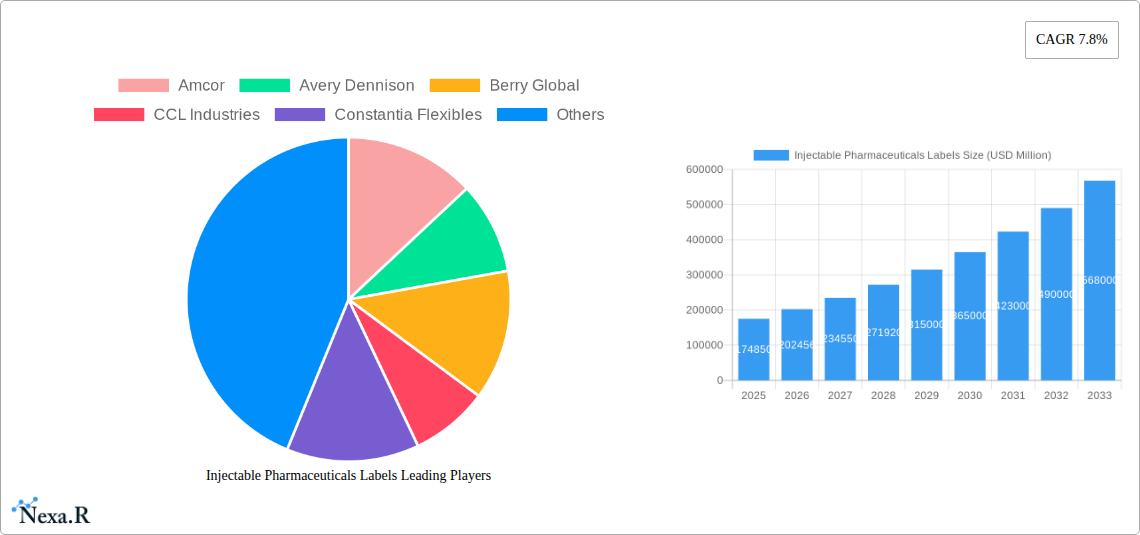
Injectable Pharmaceuticals Labels Market Size (In Billion)

The market is anticipated to experience sustained growth due to several compelling drivers. The increasing investment in pharmaceutical research and development, particularly in areas like biologics and vaccines, directly translates to a higher volume of injectable products requiring specialized labeling. Evolving healthcare infrastructure in emerging economies is also expanding access to injectable medications, thereby broadening the market reach. However, the market may encounter restraints such as the fluctuating costs of raw materials, including specialized adhesives and substrates, and the complexity of global supply chains that can impact production and distribution. Competition among prominent players like Amcor, Avery Dennison, and Berry Global is likely to intensify, driving innovation in label durability, readability, and tamper-evident features. The strategic geographical presence of these companies across North America, Europe, Asia Pacific, and other regions will play a crucial role in capturing market share and catering to diverse regional needs for compliant and effective injectable pharmaceutical labeling.
Injectable Pharmaceuticals Labels Company Market Share

This comprehensive report delves into the dynamic Injectable Pharmaceuticals Labels market, a critical component of the broader pharmaceutical packaging industry. Covering the historical period from 2019 to 2024 and projecting growth through 2033, with a base year of 2025, this analysis provides invaluable insights for stakeholders. The global market for injectable pharmaceuticals labels is expected to reach $xx billion in 2025, with a projected CAGR of xx% during the forecast period.
### Injectable Pharmaceuticals Labels Market Dynamics & Structure
The Injectable Pharmaceuticals Labels market is characterized by a moderately concentrated structure, with key players like Amcor, Avery Dennison, and UPM Raflatac holding significant market shares. Technological innovation is a primary driver, with advancements in substrate materials, printing technologies (including variable data printing and RFID integration), and adhesive formulations for extreme temperature storage (refrigerated and frozen labels) continuously shaping the competitive landscape. Stringent regulatory frameworks, particularly those governing pharmaceutical traceability, patient safety, and tamper-evidence, play a pivotal role in dictating label design and material specifications. Competitive product substitutes, such as direct printing on vials or advanced packaging solutions, pose a potential threat but are often outweighed by the cost-effectiveness and established protocols for specialized labels. End-user demographics are shifting with the rise of biologics and personalized medicine, demanding more sophisticated and highly informative labels. Mergers and acquisitions (M&A) remain an active strategy for consolidation and expansion, with recent years witnessing several notable transactions aimed at enhancing capabilities and market reach. The volume of M&A deals in the past three years is estimated at xx, impacting market share for acquired entities by an average of xx%. Barriers to innovation often stem from the rigorous validation processes required for pharmaceutical applications and the need for strict supply chain integrity.
### Injectable Pharmaceuticals Labels Growth Trends & Insights
The Injectable Pharmaceuticals Labels market is poised for significant expansion, driven by a confluence of factors. The increasing global demand for injectable drugs, fueled by an aging population, rising prevalence of chronic diseases, and advancements in therapeutic modalities like biologics and gene therapies, directly translates into a higher volume of pharmaceutical products requiring precise and durable labeling. The market size is projected to grow from an estimated $xx billion in 2025 to $xx billion by 2033, exhibiting a compound annual growth rate (CAGR) of xx%. Adoption rates for advanced labeling solutions, including cold chain compliant labels and those with enhanced security features, are steadily increasing as manufacturers prioritize product integrity and patient safety throughout the supply chain. Technological disruptions, such as the integration of Near Field Communication (NFC) and Radio-Frequency Identification (RFID) tags into labels for real-time tracking and authentication, are gaining traction, promising to revolutionize pharmaceutical logistics and combat counterfeiting. Consumer behavior shifts, though less direct in the pharmaceutical label market, are influenced by increased patient awareness regarding drug authenticity and handling instructions, prompting manufacturers to invest in clearer and more informative labeling. Market penetration of specialized labels, such as those designed for cryogenic storage or gamma irradiation sterilization, is also on the rise due to the expanding portfolio of sensitive injectable therapeutics. The overall market penetration of specialized injectable pharmaceutical labels is estimated to be around xx% in 2025, with significant room for growth.
### Dominant Regions, Countries, or Segments in Injectable Pharmaceuticals Labels
North America currently holds a dominant position in the Injectable Pharmaceuticals Labels market, driven by a robust pharmaceutical industry, significant investment in research and development, and stringent regulatory requirements that necessitate high-quality labeling solutions. The United States, in particular, represents a substantial market share of xx% within the region, attributed to its large manufacturing base for injectables, including vaccines and complex biologics, and its early adoption of serialization and track-and-trace technologies. Within the application segment, Injectable Pharmaceuticals Manufacturers constitute the largest share, accounting for an estimated xx% of the total market demand in 2025. This segment’s dominance is a direct reflection of the high volume of injectable drugs produced globally. The Refrigerated Labels segment is also experiencing substantial growth, mirroring the increasing reliance on temperature-sensitive biologics and vaccines, with an estimated market share of xx% in 2025.
### Injectable Pharmaceuticals Labels Product Landscape
The product landscape for Injectable Pharmaceuticals Labels is characterized by innovation focused on enhanced durability, compliance, and functionality. Manufacturers are developing specialized labels engineered to withstand extreme temperatures, including those required for refrigerated and frozen storage, as well as cryogenic conditions. High-performance adhesives are crucial for ensuring label adhesion on challenging surfaces like glass and plastic vials, even under fluctuating temperatures and humidity. Advancements in materials science have led to the development of tamper-evident features, chemical-resistant substrates, and sterile-grade materials that meet stringent pharmaceutical manufacturing standards. Furthermore, there is a growing integration of smart technologies, such as QR codes, bar codes, and even RFID chips, enabling enhanced traceability, data management, and patient information accessibility. The unique selling proposition of leading products lies in their ability to maintain label integrity and readability throughout the product's lifecycle, from manufacturing to administration, ensuring patient safety and regulatory compliance.
### Key Drivers, Barriers & Challenges in Injectable Pharmaceuticals Labels
The Injectable Pharmaceuticals Labels market is propelled by several key drivers. The escalating global demand for injectable pharmaceuticals, driven by an aging population and the rise of chronic and infectious diseases, directly fuels the need for labeling solutions. Advancements in biologics, vaccines, and gene therapies necessitate specialized labels capable of withstanding extreme temperatures and ensuring product integrity. Stringent regulatory mandates for serialization, track-and-trace capabilities, and counterfeit prevention are compelling manufacturers to adopt advanced labeling technologies. Economic growth in emerging markets is also opening new avenues for expansion.
However, the market faces significant barriers and challenges. The high cost associated with developing and validating specialized pharmaceutical-grade labels can be a restraint, particularly for smaller manufacturers. Rigorous regulatory approval processes for new label materials and technologies introduce lengthy lead times. Supply chain disruptions, as experienced globally in recent years, can impact the availability and cost of raw materials, leading to production delays. Intense competition among label manufacturers can also exert downward pressure on pricing, impacting profitability.
### Emerging Opportunities in Injectable Pharmaceuticals Labels
Emerging opportunities in the Injectable Pharmaceuticals Labels sector are primarily centered around the integration of smart technologies and the expansion into niche therapeutic areas. The increasing adoption of RFID and NFC tags within labels presents a significant opportunity for enhanced supply chain visibility, real-time temperature monitoring, and improved drug authentication, moving beyond basic traceability. The burgeoning field of personalized medicine and advanced therapies, such as cell and gene therapies, will demand highly specialized, often smaller-format labels with robust data management capabilities and ultra-low temperature resistance. Furthermore, the growing emphasis on sustainability is creating opportunities for eco-friendly label materials and manufacturing processes, appealing to environmentally conscious pharmaceutical companies. The need for robust cold chain management solutions for a wider range of biologics and vaccines in developing economies also represents a substantial growth frontier.
### Growth Accelerators in the Injectable Pharmaceuticals Labels Industry
The long-term growth of the Injectable Pharmaceuticals Labels industry is significantly accelerated by relentless technological innovation and strategic collaborations. Breakthroughs in material science, leading to more durable, flexible, and temperature-resistant substrates, coupled with advancements in printing technologies such as digital printing for variable data and on-demand production, are crucial. Strategic partnerships between label manufacturers, pharmaceutical companies, and technology providers are vital for developing integrated solutions that address evolving regulatory demands and supply chain complexities. The increasing focus on patient safety and drug authenticity is a powerful catalyst, driving demand for labels with advanced anti-counterfeiting features and verifiable data. Market expansion into underserved geographical regions with growing pharmaceutical manufacturing bases also acts as a significant growth accelerator.
### Key Players Shaping the Injectable Pharmaceuticals Labels Market
### Notable Milestones in Injectable Pharmaceuticals Labels Sector
### In-Depth Injectable Pharmaceuticals Labels Market Outlook
The future outlook for the Injectable Pharmaceuticals Labels market is exceptionally promising, driven by sustained global demand for injectable medicines and the continuous push for enhanced product integrity and patient safety. Growth accelerators, including technological innovation in smart labeling and advanced materials, coupled with strategic market expansion, will pave the way for significant value creation. Opportunities lie in catering to the evolving needs of biologics, vaccines, and personalized therapies, where specialized labeling solutions are paramount. The market is set to witness further consolidation and strategic alliances aimed at enhancing capabilities and global reach, ensuring a dynamic and robust future for this essential sector.
- Market Concentration: Moderate, with top 5 players holding an estimated xx% of the market share.
- Technological Drivers: Advancements in UV-curing inks, temperature-resistant adhesives, and smart labeling technologies.
- Regulatory Frameworks: FDA, EMA, and other regional regulatory bodies mandating serialization and track-and-trace capabilities.
- Competitive Substitutes: Direct vial printing, shrink sleeves, and advanced tamper-evident closures.
- End-User Demographics: Growing demand from biotech, specialty pharmaceuticals, and emerging markets.
- M&A Trends: Focus on expanding global footprint, acquiring specialized label technologies, and vertical integration.
- Regional Dominance Factors:
- North America: Strong pharmaceutical R&D, advanced healthcare infrastructure, stringent regulatory compliance (e.g., DSCSA in the US).
- Europe: Significant manufacturing hubs, established regulatory bodies (EMA), and increasing demand for high-value biologics.
- Country-Specific Drivers (USA):
- Leading pharmaceutical innovation and production of complex injectables.
- Mandatory serialization and aggregation requirements.
- High adoption of advanced cold chain labeling solutions.
- Segment Dominance (Application): Injectable Pharmaceuticals Manufacturers:
- Direct correlation with global injectable drug production volumes.
- Need for high-volume, cost-effective, and compliant labeling solutions.
- Segment Dominance (Type): Refrigerated Labels:
- Surge in demand for biologics, vaccines, and temperature-sensitive therapeutics.
- Advancements in adhesive and substrate technology for cold chain integrity.
- Key Drivers:
- Increasing global demand for injectable drugs.
- Growth in the biologics and vaccine markets.
- Stringent regulatory requirements for traceability and anti-counterfeiting.
- Technological advancements in label materials and printing.
- Key Barriers & Challenges:
- High R&D and validation costs for specialized labels.
- Lengthy regulatory approval timelines.
- Volatility in raw material prices and availability.
- Intensified competition and price pressures.
- Amcor
- Avery Dennison
- Berry Global
- CCL Industries
- Constantia Flexibles
- Flexo-Graphics
- GA International
- Hood Packaging
- Intertape Polymer Group
- Karlville Development
- Kiwi Labels
- Klckner Pentaplast
- SKK Global
- UPM Raflatac
- 2020: Increased adoption of UV-curing inks for enhanced durability and faster drying times.
- 2021: Significant advancements in cold chain adhesive technologies for prolonged temperature stability.
- 2022: Growing implementation of serialization and track-and-trace solutions driven by regulatory mandates.
- 2023: Emerging integration of QR codes and NFC tags for enhanced traceability and patient engagement.
- Early 2024: Increased focus on sustainable and eco-friendly labeling materials in response to industry demand.
Injectable Pharmaceuticals Labels Segmentation
-
1. Application
- 1.1. Biomedical Laboratory
- 1.2. Medical Institutions
- 1.3. Injectable Pharmaceuticals Manufacturers
- 1.4. Others
-
2. Types
- 2.1. Refrigerated Labels
- 2.2. Frozen Labels
Injectable Pharmaceuticals Labels Segmentation By Geography
-
1. North America
- 1.1. United States
- 1.2. Canada
- 1.3. Mexico
-
2. South America
- 2.1. Brazil
- 2.2. Argentina
- 2.3. Rest of South America
-
3. Europe
- 3.1. United Kingdom
- 3.2. Germany
- 3.3. France
- 3.4. Italy
- 3.5. Spain
- 3.6. Russia
- 3.7. Benelux
- 3.8. Nordics
- 3.9. Rest of Europe
-
4. Middle East & Africa
- 4.1. Turkey
- 4.2. Israel
- 4.3. GCC
- 4.4. North Africa
- 4.5. South Africa
- 4.6. Rest of Middle East & Africa
-
5. Asia Pacific
- 5.1. China
- 5.2. India
- 5.3. Japan
- 5.4. South Korea
- 5.5. ASEAN
- 5.6. Oceania
- 5.7. Rest of Asia Pacific
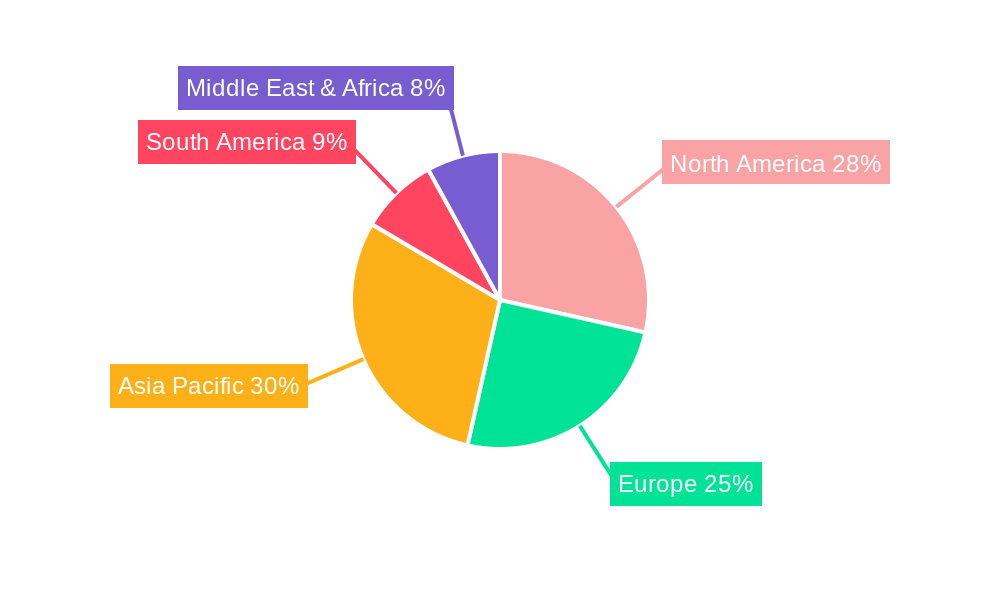
Injectable Pharmaceuticals Labels Regional Market Share

Geographic Coverage of Injectable Pharmaceuticals Labels
Injectable Pharmaceuticals Labels REPORT HIGHLIGHTS
| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 6.08% from 2020-2034 |
| Segmentation |
|
Table of Contents
- 1. Introduction
- 1.1. Research Scope
- 1.2. Market Segmentation
- 1.3. Research Methodology
- 1.4. Definitions and Assumptions
- 2. Executive Summary
- 2.1. Introduction
- 3. Market Dynamics
- 3.1. Introduction
- 3.2. Market Drivers
- 3.3. Market Restrains
- 3.4. Market Trends
- 4. Market Factor Analysis
- 4.1. Porters Five Forces
- 4.2. Supply/Value Chain
- 4.3. PESTEL analysis
- 4.4. Market Entropy
- 4.5. Patent/Trademark Analysis
- 5. Global Injectable Pharmaceuticals Labels Analysis, Insights and Forecast, 2020-2032
- 5.1. Market Analysis, Insights and Forecast - by Application
- 5.1.1. Biomedical Laboratory
- 5.1.2. Medical Institutions
- 5.1.3. Injectable Pharmaceuticals Manufacturers
- 5.1.4. Others
- 5.2. Market Analysis, Insights and Forecast - by Types
- 5.2.1. Refrigerated Labels
- 5.2.2. Frozen Labels
- 5.3. Market Analysis, Insights and Forecast - by Region
- 5.3.1. North America
- 5.3.2. South America
- 5.3.3. Europe
- 5.3.4. Middle East & Africa
- 5.3.5. Asia Pacific
- 5.1. Market Analysis, Insights and Forecast - by Application
- 6. North America Injectable Pharmaceuticals Labels Analysis, Insights and Forecast, 2020-2032
- 6.1. Market Analysis, Insights and Forecast - by Application
- 6.1.1. Biomedical Laboratory
- 6.1.2. Medical Institutions
- 6.1.3. Injectable Pharmaceuticals Manufacturers
- 6.1.4. Others
- 6.2. Market Analysis, Insights and Forecast - by Types
- 6.2.1. Refrigerated Labels
- 6.2.2. Frozen Labels
- 6.1. Market Analysis, Insights and Forecast - by Application
- 7. South America Injectable Pharmaceuticals Labels Analysis, Insights and Forecast, 2020-2032
- 7.1. Market Analysis, Insights and Forecast - by Application
- 7.1.1. Biomedical Laboratory
- 7.1.2. Medical Institutions
- 7.1.3. Injectable Pharmaceuticals Manufacturers
- 7.1.4. Others
- 7.2. Market Analysis, Insights and Forecast - by Types
- 7.2.1. Refrigerated Labels
- 7.2.2. Frozen Labels
- 7.1. Market Analysis, Insights and Forecast - by Application
- 8. Europe Injectable Pharmaceuticals Labels Analysis, Insights and Forecast, 2020-2032
- 8.1. Market Analysis, Insights and Forecast - by Application
- 8.1.1. Biomedical Laboratory
- 8.1.2. Medical Institutions
- 8.1.3. Injectable Pharmaceuticals Manufacturers
- 8.1.4. Others
- 8.2. Market Analysis, Insights and Forecast - by Types
- 8.2.1. Refrigerated Labels
- 8.2.2. Frozen Labels
- 8.1. Market Analysis, Insights and Forecast - by Application
- 9. Middle East & Africa Injectable Pharmaceuticals Labels Analysis, Insights and Forecast, 2020-2032
- 9.1. Market Analysis, Insights and Forecast - by Application
- 9.1.1. Biomedical Laboratory
- 9.1.2. Medical Institutions
- 9.1.3. Injectable Pharmaceuticals Manufacturers
- 9.1.4. Others
- 9.2. Market Analysis, Insights and Forecast - by Types
- 9.2.1. Refrigerated Labels
- 9.2.2. Frozen Labels
- 9.1. Market Analysis, Insights and Forecast - by Application
- 10. Asia Pacific Injectable Pharmaceuticals Labels Analysis, Insights and Forecast, 2020-2032
- 10.1. Market Analysis, Insights and Forecast - by Application
- 10.1.1. Biomedical Laboratory
- 10.1.2. Medical Institutions
- 10.1.3. Injectable Pharmaceuticals Manufacturers
- 10.1.4. Others
- 10.2. Market Analysis, Insights and Forecast - by Types
- 10.2.1. Refrigerated Labels
- 10.2.2. Frozen Labels
- 10.1. Market Analysis, Insights and Forecast - by Application
- 11. Competitive Analysis
- 11.1. Global Market Share Analysis 2025
- 11.2. Company Profiles
- 11.2.1 Amcor
- 11.2.1.1. Overview
- 11.2.1.2. Products
- 11.2.1.3. SWOT Analysis
- 11.2.1.4. Recent Developments
- 11.2.1.5. Financials (Based on Availability)
- 11.2.2 Avery Dennison
- 11.2.2.1. Overview
- 11.2.2.2. Products
- 11.2.2.3. SWOT Analysis
- 11.2.2.4. Recent Developments
- 11.2.2.5. Financials (Based on Availability)
- 11.2.3 Berry Global
- 11.2.3.1. Overview
- 11.2.3.2. Products
- 11.2.3.3. SWOT Analysis
- 11.2.3.4. Recent Developments
- 11.2.3.5. Financials (Based on Availability)
- 11.2.4 CCL Industries
- 11.2.4.1. Overview
- 11.2.4.2. Products
- 11.2.4.3. SWOT Analysis
- 11.2.4.4. Recent Developments
- 11.2.4.5. Financials (Based on Availability)
- 11.2.5 Constantia Flexibles
- 11.2.5.1. Overview
- 11.2.5.2. Products
- 11.2.5.3. SWOT Analysis
- 11.2.5.4. Recent Developments
- 11.2.5.5. Financials (Based on Availability)
- 11.2.6 Flexo-Graphics
- 11.2.6.1. Overview
- 11.2.6.2. Products
- 11.2.6.3. SWOT Analysis
- 11.2.6.4. Recent Developments
- 11.2.6.5. Financials (Based on Availability)
- 11.2.7 GA International
- 11.2.7.1. Overview
- 11.2.7.2. Products
- 11.2.7.3. SWOT Analysis
- 11.2.7.4. Recent Developments
- 11.2.7.5. Financials (Based on Availability)
- 11.2.8 Hood Packaging
- 11.2.8.1. Overview
- 11.2.8.2. Products
- 11.2.8.3. SWOT Analysis
- 11.2.8.4. Recent Developments
- 11.2.8.5. Financials (Based on Availability)
- 11.2.9 Intertape Polymer Group
- 11.2.9.1. Overview
- 11.2.9.2. Products
- 11.2.9.3. SWOT Analysis
- 11.2.9.4. Recent Developments
- 11.2.9.5. Financials (Based on Availability)
- 11.2.10 Karlville Development
- 11.2.10.1. Overview
- 11.2.10.2. Products
- 11.2.10.3. SWOT Analysis
- 11.2.10.4. Recent Developments
- 11.2.10.5. Financials (Based on Availability)
- 11.2.11 Kiwi Labels
- 11.2.11.1. Overview
- 11.2.11.2. Products
- 11.2.11.3. SWOT Analysis
- 11.2.11.4. Recent Developments
- 11.2.11.5. Financials (Based on Availability)
- 11.2.12 Klckner Pentaplast
- 11.2.12.1. Overview
- 11.2.12.2. Products
- 11.2.12.3. SWOT Analysis
- 11.2.12.4. Recent Developments
- 11.2.12.5. Financials (Based on Availability)
- 11.2.13 SKK Global
- 11.2.13.1. Overview
- 11.2.13.2. Products
- 11.2.13.3. SWOT Analysis
- 11.2.13.4. Recent Developments
- 11.2.13.5. Financials (Based on Availability)
- 11.2.14 UPM Raflatac
- 11.2.14.1. Overview
- 11.2.14.2. Products
- 11.2.14.3. SWOT Analysis
- 11.2.14.4. Recent Developments
- 11.2.14.5. Financials (Based on Availability)
- 11.2.1 Amcor
List of Figures
- Figure 1: Global Injectable Pharmaceuticals Labels Revenue Breakdown (undefined, %) by Region 2025 & 2033
- Figure 2: North America Injectable Pharmaceuticals Labels Revenue (undefined), by Application 2025 & 2033
- Figure 3: North America Injectable Pharmaceuticals Labels Revenue Share (%), by Application 2025 & 2033
- Figure 4: North America Injectable Pharmaceuticals Labels Revenue (undefined), by Types 2025 & 2033
- Figure 5: North America Injectable Pharmaceuticals Labels Revenue Share (%), by Types 2025 & 2033
- Figure 6: North America Injectable Pharmaceuticals Labels Revenue (undefined), by Country 2025 & 2033
- Figure 7: North America Injectable Pharmaceuticals Labels Revenue Share (%), by Country 2025 & 2033
- Figure 8: South America Injectable Pharmaceuticals Labels Revenue (undefined), by Application 2025 & 2033
- Figure 9: South America Injectable Pharmaceuticals Labels Revenue Share (%), by Application 2025 & 2033
- Figure 10: South America Injectable Pharmaceuticals Labels Revenue (undefined), by Types 2025 & 2033
- Figure 11: South America Injectable Pharmaceuticals Labels Revenue Share (%), by Types 2025 & 2033
- Figure 12: South America Injectable Pharmaceuticals Labels Revenue (undefined), by Country 2025 & 2033
- Figure 13: South America Injectable Pharmaceuticals Labels Revenue Share (%), by Country 2025 & 2033
- Figure 14: Europe Injectable Pharmaceuticals Labels Revenue (undefined), by Application 2025 & 2033
- Figure 15: Europe Injectable Pharmaceuticals Labels Revenue Share (%), by Application 2025 & 2033
- Figure 16: Europe Injectable Pharmaceuticals Labels Revenue (undefined), by Types 2025 & 2033
- Figure 17: Europe Injectable Pharmaceuticals Labels Revenue Share (%), by Types 2025 & 2033
- Figure 18: Europe Injectable Pharmaceuticals Labels Revenue (undefined), by Country 2025 & 2033
- Figure 19: Europe Injectable Pharmaceuticals Labels Revenue Share (%), by Country 2025 & 2033
- Figure 20: Middle East & Africa Injectable Pharmaceuticals Labels Revenue (undefined), by Application 2025 & 2033
- Figure 21: Middle East & Africa Injectable Pharmaceuticals Labels Revenue Share (%), by Application 2025 & 2033
- Figure 22: Middle East & Africa Injectable Pharmaceuticals Labels Revenue (undefined), by Types 2025 & 2033
- Figure 23: Middle East & Africa Injectable Pharmaceuticals Labels Revenue Share (%), by Types 2025 & 2033
- Figure 24: Middle East & Africa Injectable Pharmaceuticals Labels Revenue (undefined), by Country 2025 & 2033
- Figure 25: Middle East & Africa Injectable Pharmaceuticals Labels Revenue Share (%), by Country 2025 & 2033
- Figure 26: Asia Pacific Injectable Pharmaceuticals Labels Revenue (undefined), by Application 2025 & 2033
- Figure 27: Asia Pacific Injectable Pharmaceuticals Labels Revenue Share (%), by Application 2025 & 2033
- Figure 28: Asia Pacific Injectable Pharmaceuticals Labels Revenue (undefined), by Types 2025 & 2033
- Figure 29: Asia Pacific Injectable Pharmaceuticals Labels Revenue Share (%), by Types 2025 & 2033
- Figure 30: Asia Pacific Injectable Pharmaceuticals Labels Revenue (undefined), by Country 2025 & 2033
- Figure 31: Asia Pacific Injectable Pharmaceuticals Labels Revenue Share (%), by Country 2025 & 2033
List of Tables
- Table 1: Global Injectable Pharmaceuticals Labels Revenue undefined Forecast, by Application 2020 & 2033
- Table 2: Global Injectable Pharmaceuticals Labels Revenue undefined Forecast, by Types 2020 & 2033
- Table 3: Global Injectable Pharmaceuticals Labels Revenue undefined Forecast, by Region 2020 & 2033
- Table 4: Global Injectable Pharmaceuticals Labels Revenue undefined Forecast, by Application 2020 & 2033
- Table 5: Global Injectable Pharmaceuticals Labels Revenue undefined Forecast, by Types 2020 & 2033
- Table 6: Global Injectable Pharmaceuticals Labels Revenue undefined Forecast, by Country 2020 & 2033
- Table 7: United States Injectable Pharmaceuticals Labels Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 8: Canada Injectable Pharmaceuticals Labels Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 9: Mexico Injectable Pharmaceuticals Labels Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 10: Global Injectable Pharmaceuticals Labels Revenue undefined Forecast, by Application 2020 & 2033
- Table 11: Global Injectable Pharmaceuticals Labels Revenue undefined Forecast, by Types 2020 & 2033
- Table 12: Global Injectable Pharmaceuticals Labels Revenue undefined Forecast, by Country 2020 & 2033
- Table 13: Brazil Injectable Pharmaceuticals Labels Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 14: Argentina Injectable Pharmaceuticals Labels Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 15: Rest of South America Injectable Pharmaceuticals Labels Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 16: Global Injectable Pharmaceuticals Labels Revenue undefined Forecast, by Application 2020 & 2033
- Table 17: Global Injectable Pharmaceuticals Labels Revenue undefined Forecast, by Types 2020 & 2033
- Table 18: Global Injectable Pharmaceuticals Labels Revenue undefined Forecast, by Country 2020 & 2033
- Table 19: United Kingdom Injectable Pharmaceuticals Labels Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 20: Germany Injectable Pharmaceuticals Labels Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 21: France Injectable Pharmaceuticals Labels Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 22: Italy Injectable Pharmaceuticals Labels Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 23: Spain Injectable Pharmaceuticals Labels Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 24: Russia Injectable Pharmaceuticals Labels Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 25: Benelux Injectable Pharmaceuticals Labels Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 26: Nordics Injectable Pharmaceuticals Labels Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 27: Rest of Europe Injectable Pharmaceuticals Labels Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 28: Global Injectable Pharmaceuticals Labels Revenue undefined Forecast, by Application 2020 & 2033
- Table 29: Global Injectable Pharmaceuticals Labels Revenue undefined Forecast, by Types 2020 & 2033
- Table 30: Global Injectable Pharmaceuticals Labels Revenue undefined Forecast, by Country 2020 & 2033
- Table 31: Turkey Injectable Pharmaceuticals Labels Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 32: Israel Injectable Pharmaceuticals Labels Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 33: GCC Injectable Pharmaceuticals Labels Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 34: North Africa Injectable Pharmaceuticals Labels Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 35: South Africa Injectable Pharmaceuticals Labels Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 36: Rest of Middle East & Africa Injectable Pharmaceuticals Labels Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 37: Global Injectable Pharmaceuticals Labels Revenue undefined Forecast, by Application 2020 & 2033
- Table 38: Global Injectable Pharmaceuticals Labels Revenue undefined Forecast, by Types 2020 & 2033
- Table 39: Global Injectable Pharmaceuticals Labels Revenue undefined Forecast, by Country 2020 & 2033
- Table 40: China Injectable Pharmaceuticals Labels Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 41: India Injectable Pharmaceuticals Labels Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 42: Japan Injectable Pharmaceuticals Labels Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 43: South Korea Injectable Pharmaceuticals Labels Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 44: ASEAN Injectable Pharmaceuticals Labels Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 45: Oceania Injectable Pharmaceuticals Labels Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 46: Rest of Asia Pacific Injectable Pharmaceuticals Labels Revenue (undefined) Forecast, by Application 2020 & 2033
Frequently Asked Questions
1. What is the projected Compound Annual Growth Rate (CAGR) of the Injectable Pharmaceuticals Labels?
The projected CAGR is approximately 6.08%.
2. Which companies are prominent players in the Injectable Pharmaceuticals Labels?
Key companies in the market include Amcor, Avery Dennison, Berry Global, CCL Industries, Constantia Flexibles, Flexo-Graphics, GA International, Hood Packaging, Intertape Polymer Group, Karlville Development, Kiwi Labels, Klckner Pentaplast, SKK Global, UPM Raflatac.
3. What are the main segments of the Injectable Pharmaceuticals Labels?
The market segments include Application, Types.
4. Can you provide details about the market size?
The market size is estimated to be USD XXX N/A as of 2022.
5. What are some drivers contributing to market growth?
N/A
6. What are the notable trends driving market growth?
N/A
7. Are there any restraints impacting market growth?
N/A
8. Can you provide examples of recent developments in the market?
N/A
9. What pricing options are available for accessing the report?
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 4900.00, USD 7350.00, and USD 9800.00 respectively.
10. Is the market size provided in terms of value or volume?
The market size is provided in terms of value, measured in N/A.
11. Are there any specific market keywords associated with the report?
Yes, the market keyword associated with the report is "Injectable Pharmaceuticals Labels," which aids in identifying and referencing the specific market segment covered.
12. How do I determine which pricing option suits my needs best?
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
13. Are there any additional resources or data provided in the Injectable Pharmaceuticals Labels report?
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
14. How can I stay updated on further developments or reports in the Injectable Pharmaceuticals Labels?
To stay informed about further developments, trends, and reports in the Injectable Pharmaceuticals Labels, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.
Methodology
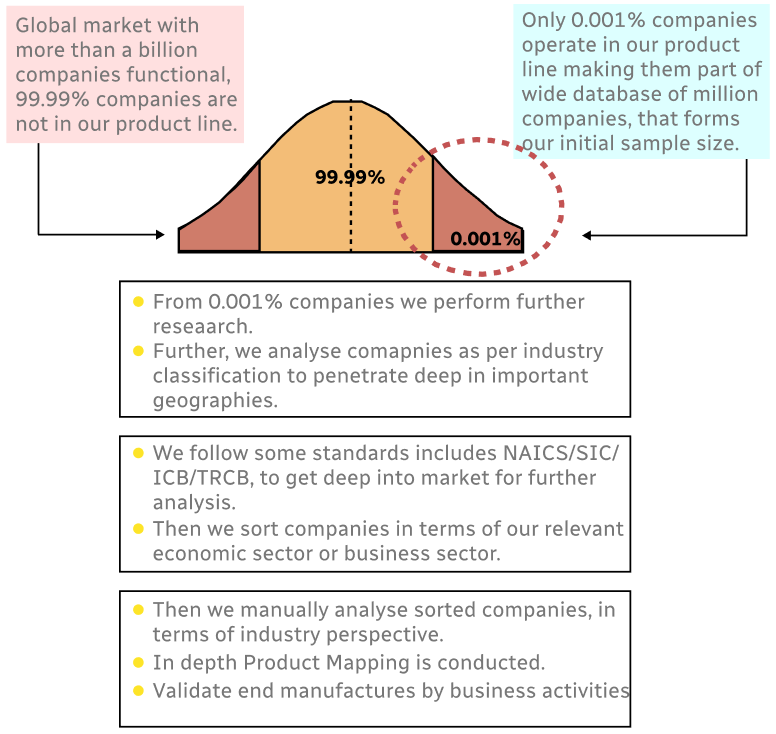
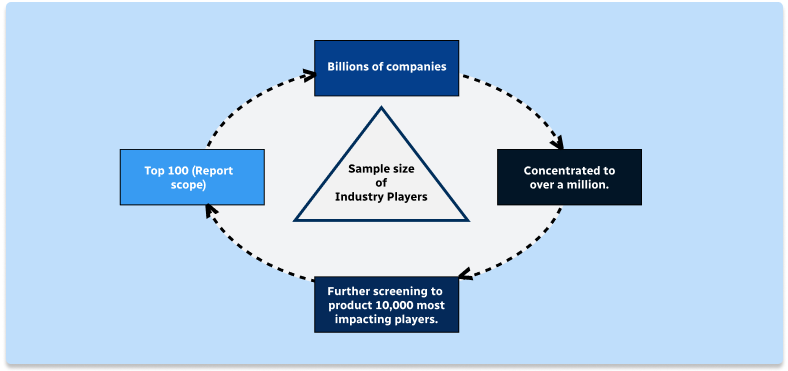
Step 1 - Identification of Relevant Samples Size from Population Database
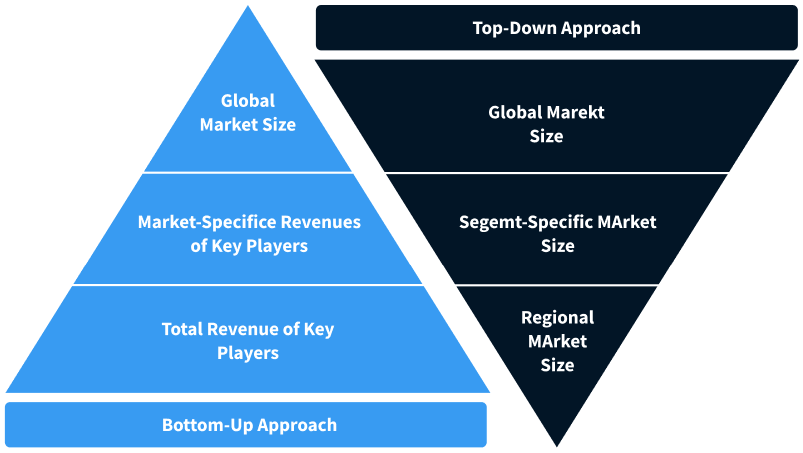
Step 2 - Approaches for Defining Global Market Size (Value, Volume* & Price*)
Note*: In applicable scenarios
Step 3 - Data Sources
Primary Research
- Web Analytics
- Survey Reports
- Research Institute
- Latest Research Reports
- Opinion Leaders
Secondary Research
- Annual Reports
- White Paper
- Latest Press Release
- Industry Association
- Paid Database
- Investor Presentations
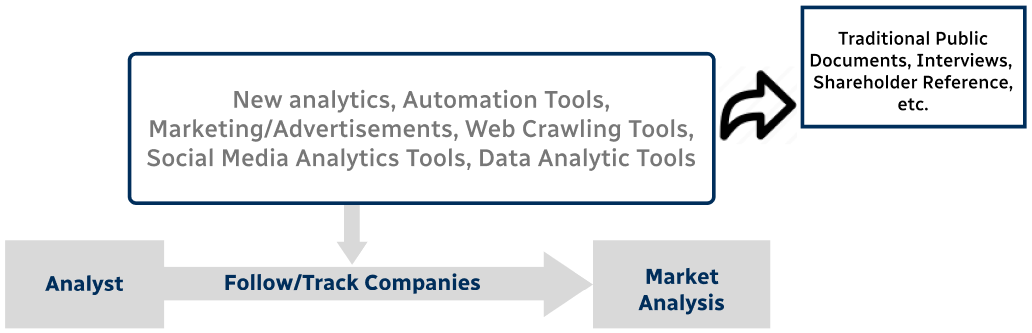
Step 4 - Data Triangulation
Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence


